Abstract
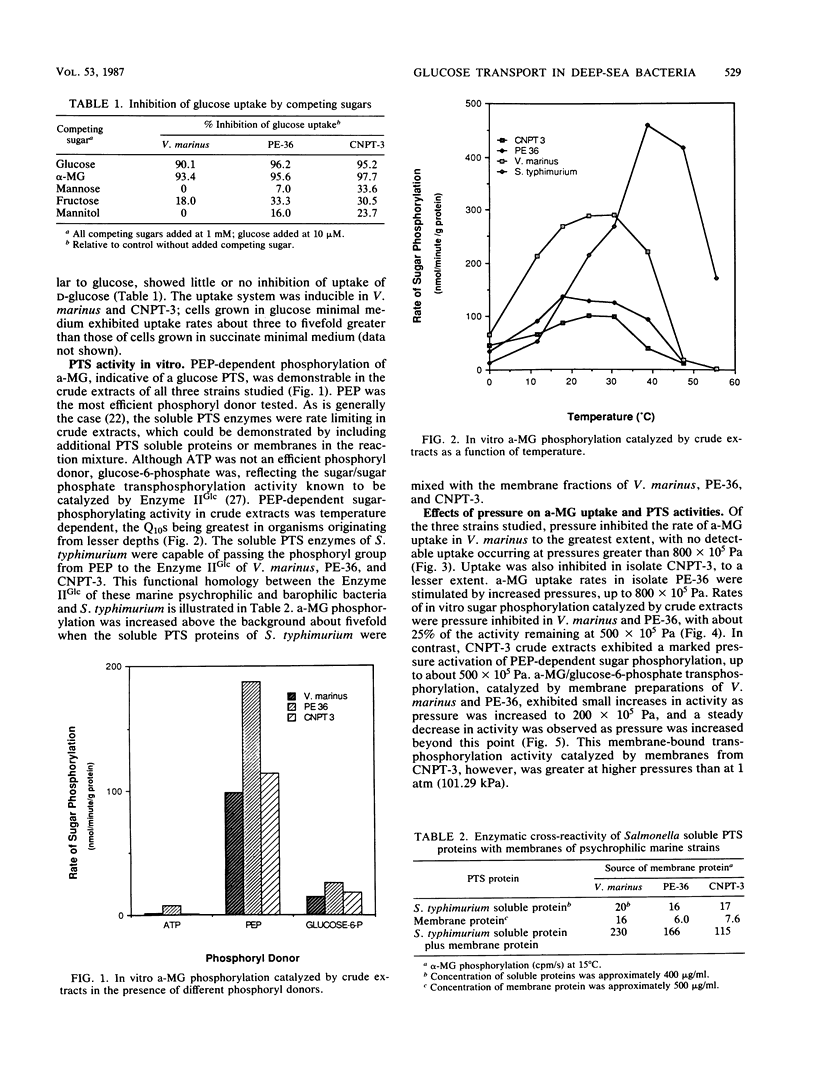
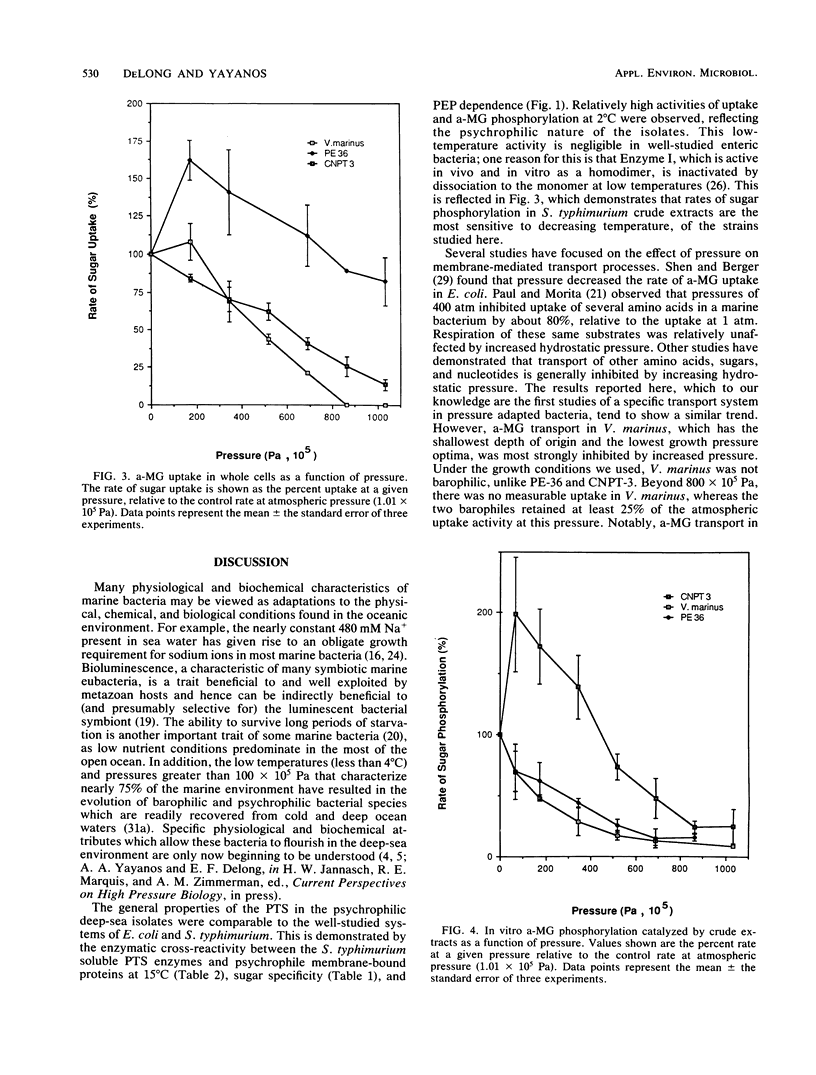
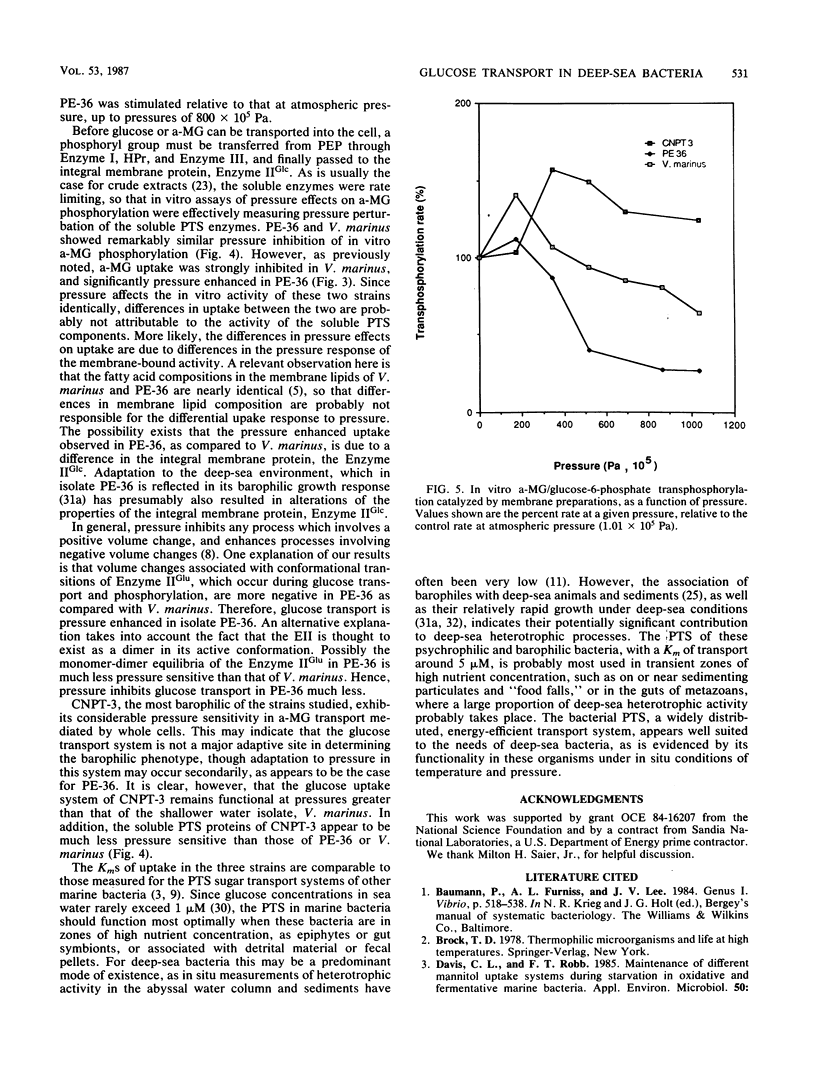
Many deep-sea bacteria are specifically adapted to flourish under the high hydrostatic pressures which exist in their natural environment. For better understanding of the physiology and biochemistry of these microorganisms, properties of the glucose transport systems in two barophilic isolates (PE-36, CNPT-3) and one psychrophilic marine bacterium (Vibrio marinus MP1) were studied. These bacteria use a phosphoenol-pyruvate:sugar phosphotransferase system (PTS) for glucose transport, similar to that found in many members of the Vibrionaceae and Enterobacteriaceae. The system was highly specific for glucose and its nonmetabolizable analog, methyl alpha-glucoside (a-MG), and exhibited little affinity for other sugars tested. The temperature optimum for glucose phosphorylation in vitro was approximately 20°C. Membrane-bound PTS components of deep-sea bacteria were capable of enzymatically cross-reacting with the soluble PTS enzymes of Salmonella typhimurium, indicating functional similarities between the PTS systems of these organisms. In CNPT-3 and V. marinus, increased pressure had an inhibitory effect on a-MG uptake, to the greatest extent in V. marinus. Relative to atmospheric pressure, increased pressure stimulated sugar uptake in the barophilic isolate PE-36 considerably. Increased hydrostatic pressure inhibited in vitro phosphoenolpyruvate-dependent a-MG phosphorylation catalyzed by crude extracts of V. marinus and PE-36 but enhanced this activity in crude extracts of the barophile CNPT-3. Both of the pressure-adapted barophilic bacteria were capable of a-MG uptake at higher pressures than was the nonbarophilic psychrophile, V. marinus.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- DeLong E. F., Yayanos A. A. Adaptation of the membrane lipids of a deep-sea bacterium to changes in hydrostatic pressure. Science. 1985 May 31;228(4703):1101–1103. doi: 10.1126/science.3992247. [DOI] [PubMed] [Google Scholar]
- Delong E. F., Yayanos A. A. Biochemical function and ecological significance of novel bacterial lipids in deep-sea procaryotes. Appl Environ Microbiol. 1986 Apr;51(4):730–737. doi: 10.1128/aem.51.4.730-737.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deming J. W., Hada H., Colwell R. R., Luehrsen K. R., Fox G. E. The ribonucleotide sequence of 5s rRNA from two strains of deep-sea barophilic bacteria. J Gen Microbiol. 1984 Aug;130(8):1911–1920. doi: 10.1099/00221287-130-8-1911. [DOI] [PubMed] [Google Scholar]
- Hodson R. E., Azam F. Occurrence and Characterization of a Phosphoenolpyruvate: Glucose Phosphotransferase System in a Marine Bacterium, Serratia marinorubra. Appl Environ Microbiol. 1979 Dec;38(6):1086–1091. doi: 10.1128/aem.38.6.1086-1091.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jannasch H. W., Taylor C. D. Deep-sea microbiology. Annu Rev Microbiol. 1984;38:487–514. doi: 10.1146/annurev.mi.38.100184.002415. [DOI] [PubMed] [Google Scholar]
- Kubota Y., Iuchi S., Fujisawa A., Tanaka S. Separation of four components of the phosphoenolpyruvate: glucose phosphotransferase system in Vibrio parahaemolyticus. Microbiol Immunol. 1979;23(3):131–146. doi: 10.1111/j.1348-0421.1979.tb00450.x. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- MACLEOD R. A. THE QUESTION OF THE EXISTENCE OF SPECIFIC MARINE BACTERIA. Bacteriol Rev. 1965 Mar;29:9–24. [PMC free article] [PubMed] [Google Scholar]
- Macdonald A. G. The effects of pressure on the molecular structure and physiological functions of cell membranes. Philos Trans R Soc Lond B Biol Sci. 1984 Jan 7;304(1118):47–68. doi: 10.1098/rstb.1984.0008. [DOI] [PubMed] [Google Scholar]
- Morita R. Y. Psychrophilic bacteria. Bacteriol Rev. 1975 Jun;39(2):144–167. doi: 10.1128/br.39.2.144-167.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nealson K. H., Hastings J. W. Bacterial bioluminescence: its control and ecological significance. Microbiol Rev. 1979 Dec;43(4):496–518. doi: 10.1128/mr.43.4.496-518.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Novitsky J. A., Morita R. Y. Survival of a psychrophilic marine Vibrio under long-term nutrient starvation. Appl Environ Microbiol. 1977 Mar;33(3):635–641. doi: 10.1128/aem.33.3.635-641.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paul K. L., Morita R. Y. Effects of hydrostatic pressure and temperature on the uptake and respiration of amino acids by a facultatively psychrophilic marine bacterium. J Bacteriol. 1971 Nov;108(2):835–843. doi: 10.1128/jb.108.2.835-843.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Postma P. W., Lengeler J. W. Phosphoenolpyruvate:carbohydrate phosphotransferase system of bacteria. Microbiol Rev. 1985 Sep;49(3):232–269. doi: 10.1128/mr.49.3.232-269.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Postma P. W., Roseman S. The bacterial phosphoenolpyruvate: sugar phosphotransferase system. Biochim Biophys Acta. 1976 Dec 14;457(3-4):213–257. doi: 10.1016/0304-4157(76)90001-0. [DOI] [PubMed] [Google Scholar]
- Reichelt J. L., Baumann P. Effect of sodium chloride on growth of heterotrophic marine bacteria. Arch Microbiol. 1974 May 20;97(4):329–345. doi: 10.1007/BF00403071. [DOI] [PubMed] [Google Scholar]
- Saier M. H., Jr, Feucht B. U., Mora W. K. Sugar phosphate: sugar transphosphorylation and exchange group translocation catalyzed by the enzyme 11 complexes of the bacterial phosphoenolpyruvate: sugar phosphotransferase system. J Biol Chem. 1977 Dec 25;252(24):8899–8907. [PubMed] [Google Scholar]
- Waygood E. B., Meadow N. D., Roseman S. Modified assay procedures for the phosphotransferase system in enteric bacteria. Anal Biochem. 1979 May;95(1):293–304. doi: 10.1016/0003-2697(79)90219-7. [DOI] [PubMed] [Google Scholar]
- Yayanos A. A., Dietz A. S. Thermal Inactivation of a Deep-Sea Barophilic Bacterium, Isolate CNPT-3. Appl Environ Microbiol. 1982 Jun;43(6):1481–1489. doi: 10.1128/aem.43.6.1481-1489.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yayanos A. A., Dietz A. S., VAN Boxtel R. Isolation of a deep-sea barophilic bacterium and some of its growth characteristics. Science. 1979 Aug 24;205(4408):808–810. doi: 10.1126/science.205.4408.808. [DOI] [PubMed] [Google Scholar]
- Yayanos A. A. Evolutional and ecological implications of the properties of deep-sea barophilic bacteria. Proc Natl Acad Sci U S A. 1986 Dec;83(24):9542–9546. doi: 10.1073/pnas.83.24.9542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zobell C. E., Johnson F. H. THE INFLUENCE OF HYDROSTATIC PRESSURE ON THE GROWTH AND VIABILITY OF TERRESTRIAL AND MARINE BACTERIA. J Bacteriol. 1949 Feb;57(2):179–189. doi: 10.1128/jb.57.2.179-189.1949. [DOI] [PMC free article] [PubMed] [Google Scholar]


