Abstract
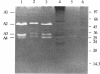
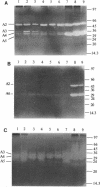
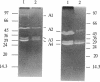
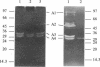
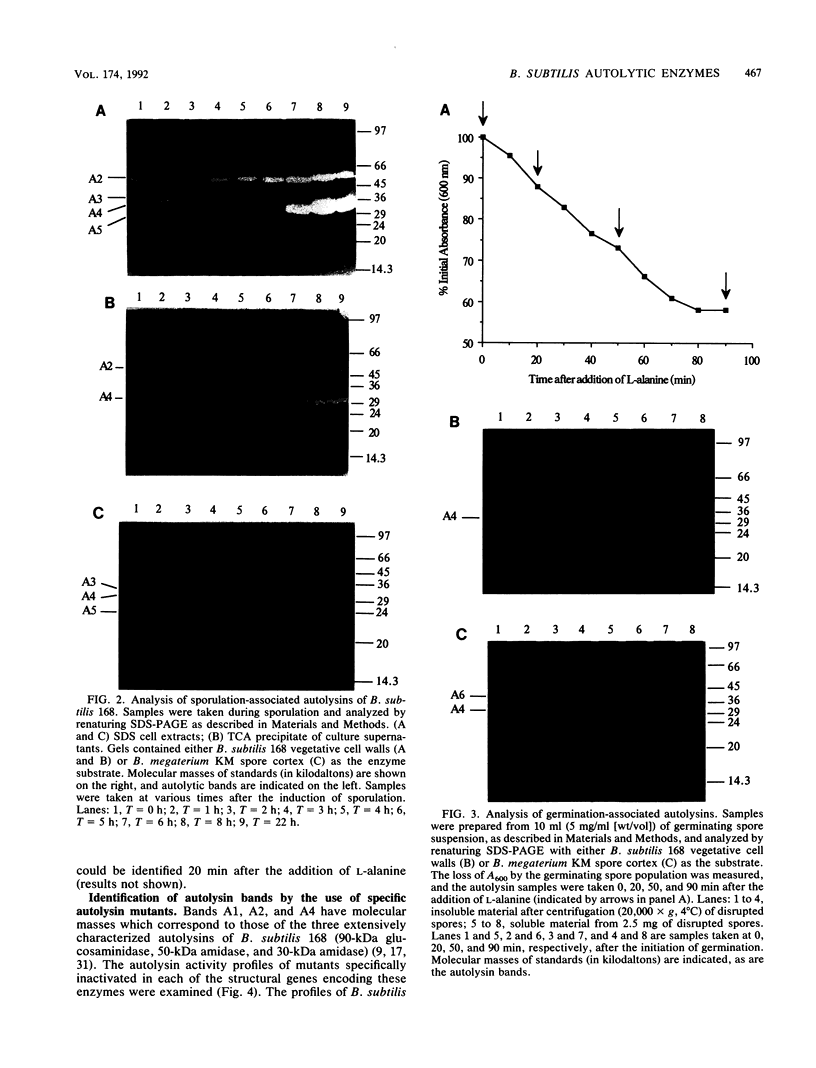
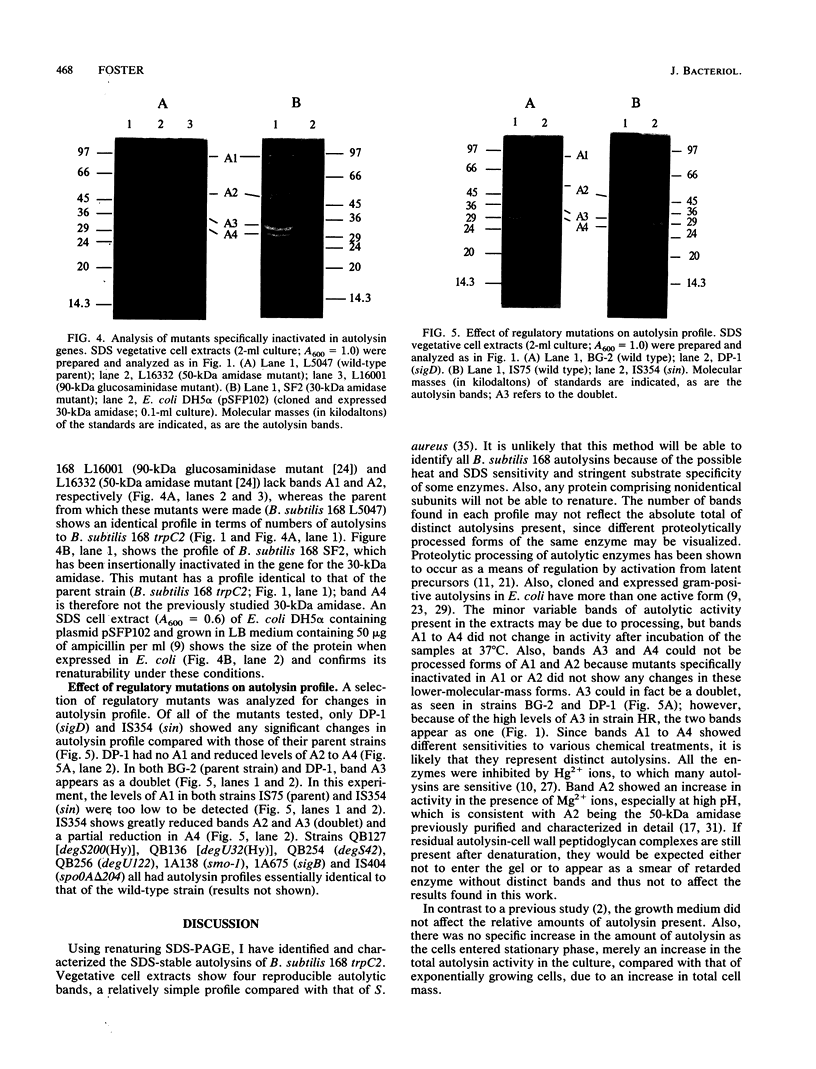
The autolysins of Bacillus subtilis 168 were analyzed by sodium dodecyl sulfate (SDS)-polyacrylamide gel electrophoresis with substrate-containing gels. Four bands of vegetative autolytic activity of 90, 50, 34, and 30 kDa (bands A1 to A4) were detected in SDS and LiCl extracts and in native cell walls by using B. subtilis 168 vegetative cell walls as the substrate incorporated in the gel. The four enzyme activities showed different substrate specificities and sensitivities to various chemical treatments. The autolysin profile was not medium dependent and remained constant during vegetative growth. During sporulation, band A4 greatly increased in activity just prior to mother-cell lysis. No germination-associated changes in the profile were observed, although a soluble 41-kDa endospore-associated cortex-lytic enzyme was found. By using insertionally inactivated mutants, bands A1 and A2 were positively identified as the previously characterized 90-kDa glucosaminidase and 50-kDa amidase, respectively. The common filamentous phenotype of various regulatory mutants could not be correlated to specific changes in the autolysin profile.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anagnostopoulos C., Spizizen J. REQUIREMENTS FOR TRANSFORMATION IN BACILLUS SUBTILIS. J Bacteriol. 1961 May;81(5):741–746. doi: 10.1128/jb.81.5.741-746.1961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doyle R. J., Koch A. L. The functions of autolysins in the growth and division of Bacillus subtilis. Crit Rev Microbiol. 1987;15(2):169–222. doi: 10.3109/10408418709104457. [DOI] [PubMed] [Google Scholar]
- Fein J. E., Rogers H. J. Autolytic enzyme-deficient mutants of Bacillus subtilis 168. J Bacteriol. 1976 Sep;127(3):1427–1442. doi: 10.1128/jb.127.3.1427-1442.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Foster S. J. Cloning, expression, sequence analysis and biochemical characterization of an autolytic amidase of Bacillus subtilis 168 trpC2. J Gen Microbiol. 1991 Aug;137(8):1987–1998. doi: 10.1099/00221287-137-8-1987. [DOI] [PubMed] [Google Scholar]
- Foster S. J., Johnstone K. Germination-specific cortex-lytic enzyme is activated during triggering of Bacillus megaterium KM spore germination. Mol Microbiol. 1988 Nov;2(6):727–733. doi: 10.1111/j.1365-2958.1988.tb00083.x. [DOI] [PubMed] [Google Scholar]
- Foster S. J., Johnstone K. Purification and properties of a germination-specific cortex-lytic enzyme from spores of Bacillus megaterium KM. Biochem J. 1987 Mar 1;242(2):573–579. doi: 10.1042/bj2420573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gaur N. K., Dubnau E., Smith I. Characterization of a cloned Bacillus subtilis gene that inhibits sporulation in multiple copies. J Bacteriol. 1986 Nov;168(2):860–869. doi: 10.1128/jb.168.2.860-869.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guinand M., Michel G., Balassa G. Lytic enzymes in sporulating Bacillus subtilis. Biochem Biophys Res Commun. 1976 Feb 23;68(4):1287–1293. doi: 10.1016/0006-291x(76)90336-3. [DOI] [PubMed] [Google Scholar]
- Hanahan D. Studies on transformation of Escherichia coli with plasmids. J Mol Biol. 1983 Jun 5;166(4):557–580. doi: 10.1016/s0022-2836(83)80284-8. [DOI] [PubMed] [Google Scholar]
- Helmann J. D., Márquez L. M., Chamberlin M. J. Cloning, sequencing, and disruption of the Bacillus subtilis sigma 28 gene. J Bacteriol. 1988 Apr;170(4):1568–1574. doi: 10.1128/jb.170.4.1568-1574.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herbold D. R., Glaser L. Bacillus subtilis N-acetylmuramic acid L-alanine amidase. J Biol Chem. 1975 Mar 10;250(5):1676–1682. [PubMed] [Google Scholar]
- Höltje J. V., Tuomanen E. I. The murein hydrolases of Escherichia coli: properties, functions and impact on the course of infections in vivo. J Gen Microbiol. 1991 Mar;137(3):441–454. doi: 10.1099/00221287-137-3-441. [DOI] [PubMed] [Google Scholar]
- Jayaswal R. K., Lee Y. I., Wilkinson B. J. Cloning and expression of a Staphylococcus aureus gene encoding a peptidoglycan hydrolase activity. J Bacteriol. 1990 Oct;172(10):5783–5788. doi: 10.1128/jb.172.10.5783-5788.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawamura T., Shockman G. D. Purification and some properties of the endogenous, autolytic N-acetylmuramoylhydrolase of Streptococcus faecium, a bacterial glycoenzyme. J Biol Chem. 1983 Aug 10;258(15):9514–9521. [PubMed] [Google Scholar]
- Kuroda A., Sekiguchi J. Cloning, sequencing and genetic mapping of a Bacillus subtilis cell wall hydrolase gene. J Gen Microbiol. 1990 Nov;136(11):2209–2216. doi: 10.1099/00221287-136-11-2209. [DOI] [PubMed] [Google Scholar]
- Leclerc D., Asselin A. Detection of bacterial cell wall hydrolases after denaturing polyacrylamide gel electrophoresis. Can J Microbiol. 1989 Aug;35(8):749–753. doi: 10.1139/m89-125. [DOI] [PubMed] [Google Scholar]
- Msadek T., Kunst F., Henner D., Klier A., Rapoport G., Dedonder R. Signal transduction pathway controlling synthesis of a class of degradative enzymes in Bacillus subtilis: expression of the regulatory genes and analysis of mutations in degS and degU. J Bacteriol. 1990 Feb;172(2):824–834. doi: 10.1128/jb.172.2.824-834.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Márquez L. M., Helmann J. D., Ferrari E., Parker H. M., Ordal G. W., Chamberlin M. J. Studies of sigma D-dependent functions in Bacillus subtilis. J Bacteriol. 1990 Jun;172(6):3435–3443. doi: 10.1128/jb.172.6.3435-3443.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peetz R. H., Kenny G. E. Prevention of autolysis in suspensions of Neisseria gonorrhoeae by mercuric ions. J Bacteriol. 1978 Jul;135(1):283–285. doi: 10.1128/jb.135.1.283-285.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Potvin C., Leclerc D., Tremblay G., Asselin A., Bellemare G. Cloning, sequencing and expression of a Bacillus bacteriolytic enzyme in Escherichia coli. Mol Gen Genet. 1988 Oct;214(2):241–248. doi: 10.1007/BF00337717. [DOI] [PubMed] [Google Scholar]
- Rogers H. J., Forsberg C. W. Role of autolysins in the killing of bacteria by some bactericidal antibiotics. J Bacteriol. 1971 Dec;108(3):1235–1243. doi: 10.1128/jb.108.3.1235-1243.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rogers H. J., Taylor C., Rayter S., Ward J. B. Purification and properties of autolytic endo-beta-N-acetylglucosaminidase and the N-acetylmuramyl-L-alanine amidase from Bacillus subtilis strain 168. J Gen Microbiol. 1984 Sep;130(9):2395–2402. doi: 10.1099/00221287-130-9-2395. [DOI] [PubMed] [Google Scholar]
- Sekiguchi J., Ezaki B., Kodama K., Akamatsu T. Molecular cloning of a gene affecting the autolysin level and flagellation in Bacillus subtilis. J Gen Microbiol. 1988 Jun;134(6):1611–1621. doi: 10.1099/00221287-134-6-1611. [DOI] [PubMed] [Google Scholar]
- Sterlini J. M., Mandelstam J. Commitment to sporulation in Bacillus subtilis and its relationship to development of actinomycin resistance. Biochem J. 1969 Jun;113(1):29–37. doi: 10.1042/bj1130029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stewart G. S., Johnstone K., Hagelberg E., Ellar D. J. Commitment of bacterial spores to germinate. A measure of the trigger reaction. Biochem J. 1981 Jul 15;198(1):101–106. doi: 10.1042/bj1980101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugai M., Akiyama T., Komatsuzawa H., Miyake Y., Suginaka H. Characterization of sodium dodecyl sulfate-stable Staphylococcus aureus bacteriolytic enzymes by polyacrylamide gel electrophoresis. J Bacteriol. 1990 Nov;172(11):6494–6498. doi: 10.1128/jb.172.11.6494-6498.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Venkatasubramanian P., Johnstone K. Biochemical analysis of the Bacillus subtilis 1604 spore germination response. J Gen Microbiol. 1989 Oct;135(10):2723–2733. doi: 10.1099/00221287-135-10-2723. [DOI] [PubMed] [Google Scholar]
- Vitkovic L. Cell wall turnover in lyt mutants of Bacillus subtilis. Ann Inst Pasteur Microbiol. 1985 Jan-Feb;136A(1):67–72. doi: 10.1016/s0769-2609(85)80024-7. [DOI] [PubMed] [Google Scholar]