Abstract
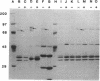
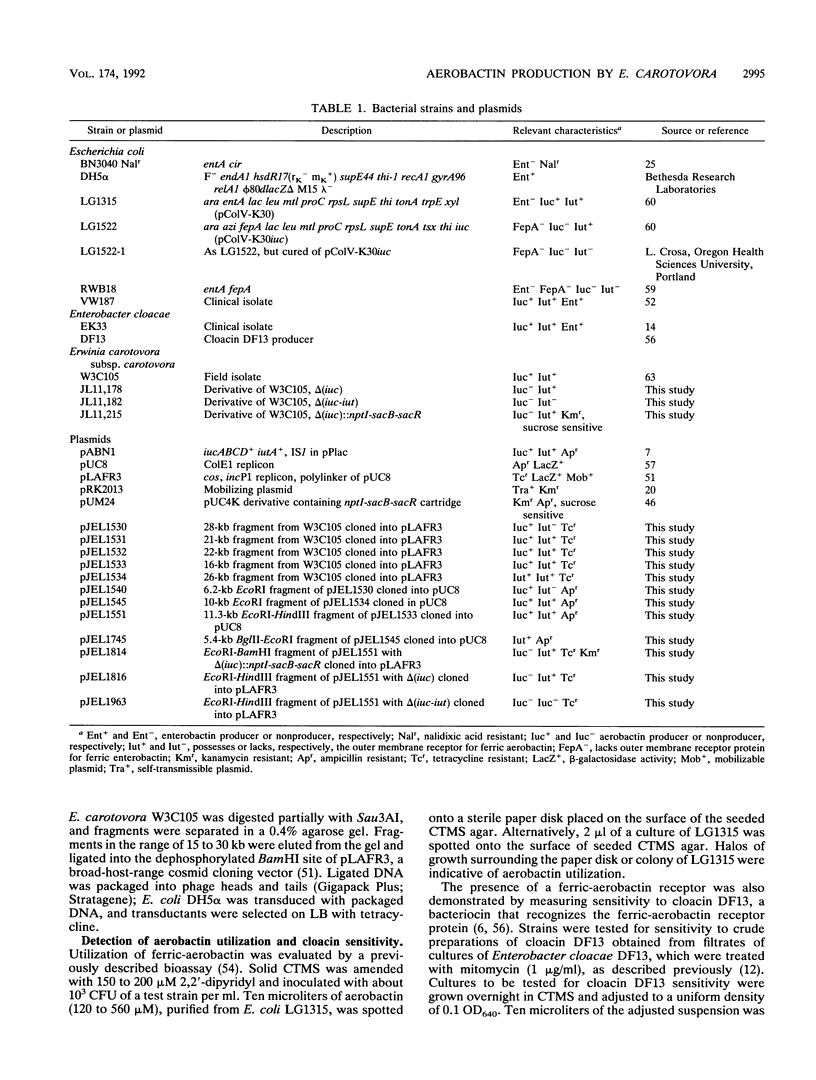
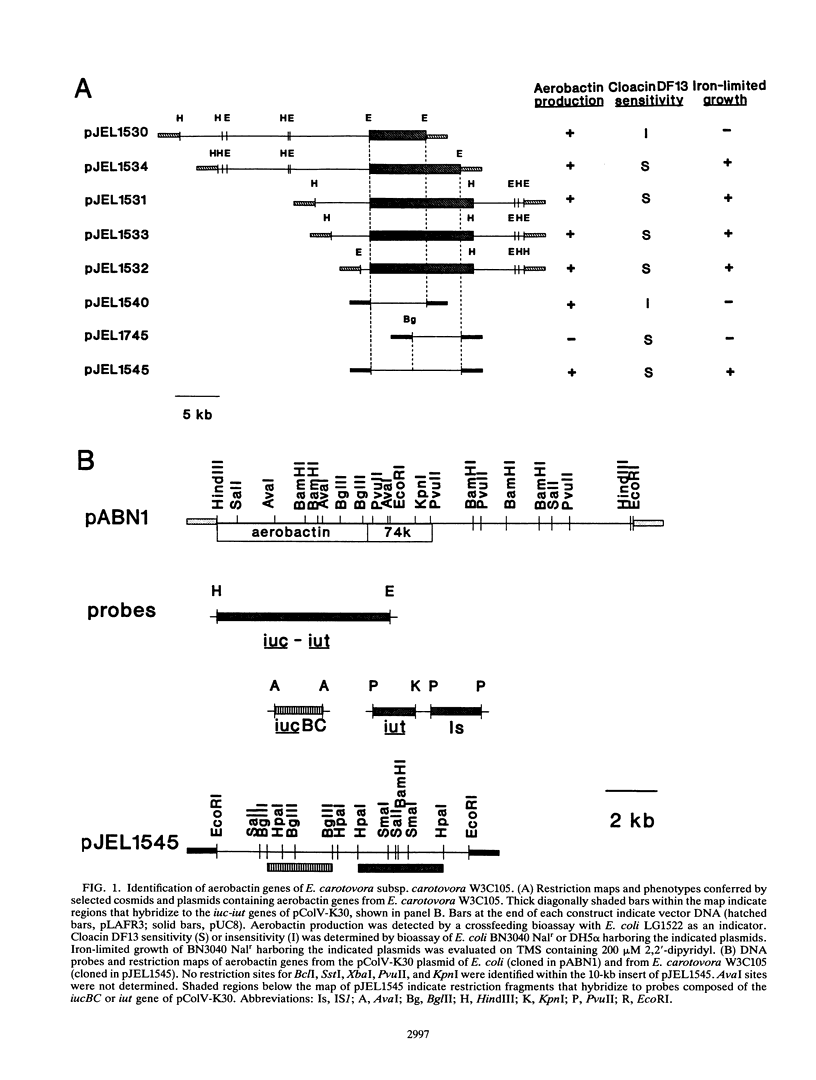
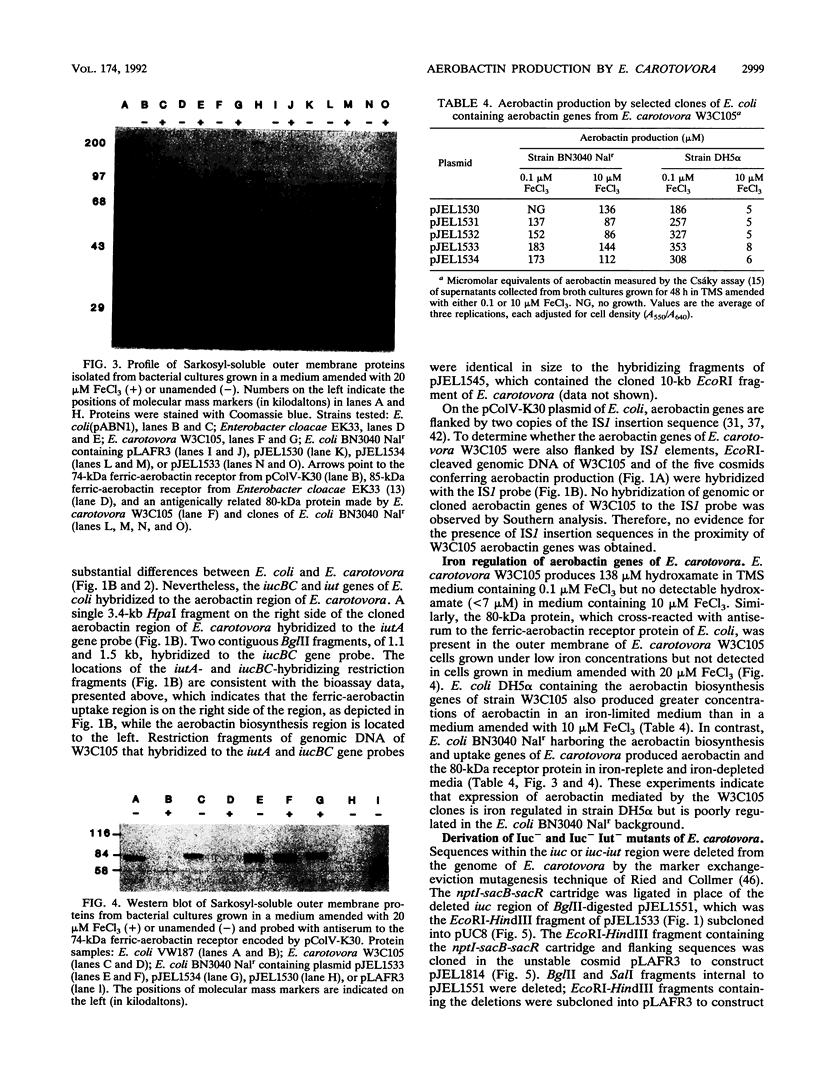
The phytopathogenic bacterium Erwinia carotovora subsp. carotovora W3C105 produced the hydroxamate siderophore aerobactin under iron-limiting conditions. A survey of 22 diverse strains of E. carotovora revealed that strain W3C105 alone produced aerobactin. The ferric-aerobactin receptor of strain W3C105 was an 80-kDa protein, identified by immunoblots of Sarkosyl-soluble proteins obtained from E. carotovora cells grown in iron-depleted medium and probed with antiserum raised against the 74-kDa ferric-aerobactin receptor encoded by the pColV-K30 plasmid of Escherichia coli. Genes determining aerobactin biosynthesis and uptake were localized to an 11.3-kb EcoRI-HindIII chromosomal fragment of strain W3C105. A 10-kb subclone of the fragment conferred on E. coli DH5 alpha both aerobactin biosynthesis and uptake, determined by cloacin DF13 sensitivity, the presence of the 80-kDa receptor protein, and iron-independent growth of E. coli clones. The aerobactin biosynthesis genes of E. carotovora W3C105 hybridized to those of the pColV-K30 plasmid of E. coli, but the restriction patterns of the aerobactin regions of E. coli and E. carotovora differed. Although the aerobactin region of enteric bacteria is commonly flanked by IS1-like sequences, IS1 sequences were not detected in the genomic DNA or the cloned aerobactin region of E. carotovora. E. coli DH5 alpha cells harboring cloned aerobactin biosynthesis genes from E. carotovora W3C105 produced greater quantities of aerobactin and the 80-kDa ferric-aerobactin receptor when grown in iron-limited than in iron-replete medium. Strain W3C105 grew on an iron-limited medium, whereas derivatives that lacked a functional aerobactin iron acquisition system did not grow on the medium. These results provide evidence for the occurrence and heterogeneity of aerobactin as a high-affinity iron uptake system of both clinical and phytopathogenic species of the Enterobacteriaceae. Although future studies may reveal a role for aerobactin in the virulence or ecology of strain W3C105, a functional aerobactin iron acquisition system is not necessary for the pathogenicity of E. carotovora.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bagg A., Neilands J. B. Ferric uptake regulation protein acts as a repressor, employing iron (II) as a cofactor to bind the operator of an iron transport operon in Escherichia coli. Biochemistry. 1987 Aug 25;26(17):5471–5477. doi: 10.1021/bi00391a039. [DOI] [PubMed] [Google Scholar]
- Bell S. J., Friedman S. A., Leong J. Antibiotic action of N-methylthioformohydroxamate metal complexes. Antimicrob Agents Chemother. 1979 Mar;15(3):384–391. doi: 10.1128/aac.15.3.384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bindereif A., Braun V., Hantke K. The cloacin receptor of ColV-bearing Escherichia coli is part of the Fe3+-aerobactin transport system. J Bacteriol. 1982 Jun;150(3):1472–1475. doi: 10.1128/jb.150.3.1472-1475.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bindereif A., Neilands J. B. Aerobactin genes in clinical isolates of Escherichia coli. J Bacteriol. 1985 Feb;161(2):727–735. doi: 10.1128/jb.161.2.727-735.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bindereif A., Neilands J. B. Cloning of the aerobactin-mediated iron assimilation system of plasmid ColV. J Bacteriol. 1983 Feb;153(2):1111–1113. doi: 10.1128/jb.153.2.1111-1113.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bindereif A., Neilands J. B. Promoter mapping and transcriptional regulation of the iron assimilation system of plasmid ColV-K30 in Escherichia coli K-12. J Bacteriol. 1985 Jun;162(3):1039–1046. doi: 10.1128/jb.162.3.1039-1046.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braun V., Burkhardt R. Regulation of the ColV plasmid-determined iron (III)-aerobactin transport system in Escherichia coli. J Bacteriol. 1982 Oct;152(1):223–231. doi: 10.1128/jb.152.1.223-231.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carbonetti N. H., Williams P. H. A cluster of five genes specifying the aerobactin iron uptake system of plasmid ColV-K30. Infect Immun. 1984 Oct;46(1):7–12. doi: 10.1128/iai.46.1.7-12.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crosa L. M., Wolf M. K., Actis L. A., Sanders-Loehr J., Crosa J. H. New aerobactin-mediated iron uptake system in a septicemia-causing strain of Enterobacter cloacae. J Bacteriol. 1988 Dec;170(12):5539–5544. doi: 10.1128/jb.170.12.5539-5544.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Enard C., Diolez A., Expert D. Systemic virulence of Erwinia chrysanthemi 3937 requires a functional iron assimilation system. J Bacteriol. 1988 Jun;170(6):2419–2426. doi: 10.1128/jb.170.6.2419-2426.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Figurski D. H., Helinski D. R. Replication of an origin-containing derivative of plasmid RK2 dependent on a plasmid function provided in trans. Proc Natl Acad Sci U S A. 1979 Apr;76(4):1648–1652. doi: 10.1073/pnas.76.4.1648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Figurski D. H., Meyer R. J., Helinski D. R. Suppression of Co1E1 replication properties by the Inc P-1 plasmid RK2 in hybrid plasmids constructed in vitro. J Mol Biol. 1979 Sep 25;133(3):295–318. doi: 10.1016/0022-2836(79)90395-4. [DOI] [PubMed] [Google Scholar]
- Gibson F., Magrath D. I. The isolation and characterization of a hydroxamic acid (aerobactin) formed by Aerobacter aerogenes 62-I. Biochim Biophys Acta. 1969 Nov 18;192(2):175–184. doi: 10.1016/0304-4165(69)90353-5. [DOI] [PubMed] [Google Scholar]
- Hinton J. C., Perombelon M. C., Salmond G. P. Efficient transformation of Erwinia carotovora subsp. carotovora and E. carotovora subsp. atroseptica. J Bacteriol. 1985 Feb;161(2):786–788. doi: 10.1128/jb.161.2.786-788.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hollifield W. C., Jr, Fiss E. H., Neilands J. B. Modification of a ferric enterobactin receptor protein from the outer membrane of Escherichia coli. Biochem Biophys Res Commun. 1978 Jul 28;83(2):739–746. doi: 10.1016/0006-291x(78)91051-3. [DOI] [PubMed] [Google Scholar]
- Krone W. J., Oudega B., Stegehuis F., de Graaf F. K. Cloning and expression of the cloacin DF13/aerobactin receptor of Escherichia coli (ColV-K30). J Bacteriol. 1983 Feb;153(2):716–721. doi: 10.1128/jb.153.2.716-721.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawlor K. M., Payne S. M. Aerobactin genes in Shigella spp. J Bacteriol. 1984 Oct;160(1):266–272. doi: 10.1128/jb.160.1.266-272.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leong S. A., Neilands J. B. Siderophore production by phytopathogenic microbial species. Arch Biochem Biophys. 1982 Oct 15;218(2):351–359. doi: 10.1016/0003-9861(82)90356-3. [DOI] [PubMed] [Google Scholar]
- Marolda C. L., Valvano M. A., Lawlor K. M., Payne S. M., Crosa J. H. Flanking and internal regions of chromosomal genes mediating aerobactin iron uptake systems in enteroinvasive Escherichia coli and Shigella flexneri. J Gen Microbiol. 1987 Aug;133(8):2269–2278. doi: 10.1099/00221287-133-8-2269. [DOI] [PubMed] [Google Scholar]
- McDougall S., Neilands J. B. Plasmid- and chromosome-coded aerobactin synthesis in enteric bacteria: insertion sequences flank operon in plasmid-mediated systems. J Bacteriol. 1984 Jul;159(1):300–305. doi: 10.1128/jb.159.1.300-305.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neilands J. B. Microbial iron compounds. Annu Rev Biochem. 1981;50:715–731. doi: 10.1146/annurev.bi.50.070181.003435. [DOI] [PubMed] [Google Scholar]
- Ohrloff C., Olson R., Apple D., Carey J. Angeborene Hornhauttrübung durch Verdickung der Bowman'schen Membran. Fortschr Ophthalmol. 1984;81(5):429–432. [PubMed] [Google Scholar]
- Payne S. M. Iron and virulence in the family Enterobacteriaceae. Crit Rev Microbiol. 1988;16(2):81–111. doi: 10.3109/10408418809104468. [DOI] [PubMed] [Google Scholar]
- Payne S. M. Synthesis and utilization of siderophores by Shigella flexneri. J Bacteriol. 1980 Sep;143(3):1420–1424. doi: 10.1128/jb.143.3.1420-1424.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perez-Casal J. F., Crosa J. H. Aerobactin iron uptake sequences in plasmid ColV-K30 are flanked by inverted IS1-like elements and replication regions. J Bacteriol. 1984 Oct;160(1):256–265. doi: 10.1128/jb.160.1.256-265.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Persmark M., Expert D., Neilands J. B. Isolation, characterization, and synthesis of chrysobactin, a compound with siderophore activity from Erwinia chrysanthemi. J Biol Chem. 1989 Feb 25;264(6):3187–3193. [PubMed] [Google Scholar]
- Ried J. L., Collmer A. An nptI-sacB-sacR cartridge for constructing directed, unmarked mutations in gram-negative bacteria by marker exchange-eviction mutagenesis. Gene. 1987;57(2-3):239–246. doi: 10.1016/0378-1119(87)90127-2. [DOI] [PubMed] [Google Scholar]
- Rioux C., Jordan D. C., Rattray J. B. Colorimetric determination of catechol siderophores in microbial cultures. Anal Biochem. 1983 Aug;133(1):163–169. doi: 10.1016/0003-2697(83)90238-5. [DOI] [PubMed] [Google Scholar]
- SIMON E. H., TESSMAN I. THYMIDINE-REQUIRING MUTANTS OF PHAGE T4. Proc Natl Acad Sci U S A. 1963 Sep;50:526–532. doi: 10.1073/pnas.50.3.526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwyn B., Neilands J. B. Universal chemical assay for the detection and determination of siderophores. Anal Biochem. 1987 Jan;160(1):47–56. doi: 10.1016/0003-2697(87)90612-9. [DOI] [PubMed] [Google Scholar]
- Staskawicz B., Dahlbeck D., Keen N., Napoli C. Molecular characterization of cloned avirulence genes from race 0 and race 1 of Pseudomonas syringae pv. glycinea. J Bacteriol. 1987 Dec;169(12):5789–5794. doi: 10.1128/jb.169.12.5789-5794.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valvano M. A., Crosa J. H. Aerobactin iron transport genes commonly encoded by certain ColV plasmids occur in the chromosome of a human invasive strain of Escherichia coli K1. Infect Immun. 1984 Oct;46(1):159–167. doi: 10.1128/iai.46.1.159-167.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valvano M. A., Crosa J. H. Molecular cloning, expression, and regulation in Escherichia coli K-12 of a chromosome-mediated aerobactin iron transport system from a human invasive isolate of E. coli K1. J Bacteriol. 1988 Dec;170(12):5529–5538. doi: 10.1128/jb.170.12.5529-5538.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valvano M. A., Silver R. P., Crosa J. H. Occurrence of chromosome- or plasmid-mediated aerobactin iron transport systems and hemolysin production among clonal groups of human invasive strains of Escherichia coli K1. Infect Immun. 1986 Apr;52(1):192–199. doi: 10.1128/iai.52.1.192-199.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Tiel-Menkveld G. J., Mentjox-Vervuurt J. M., Oudega B., de Graaf F. K. Siderophore production by Enterobacter cloacae and a common receptor protein for the uptake of aerobactin and cloacin DF13. J Bacteriol. 1982 May;150(2):490–497. doi: 10.1128/jb.150.2.490-497.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vanneste J. L., Paulin J. P., Expert D. Bacteriophage Mu as a genetic tool to study Erwinia amylovora pathogenicity and hypersensitive reaction on tobacco. J Bacteriol. 1990 Feb;172(2):932–941. doi: 10.1128/jb.172.2.932-941.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vieira J., Messing J. The pUC plasmids, an M13mp7-derived system for insertion mutagenesis and sequencing with synthetic universal primers. Gene. 1982 Oct;19(3):259–268. doi: 10.1016/0378-1119(82)90015-4. [DOI] [PubMed] [Google Scholar]
- Waters V. L., Crosa J. H. Divergence of the aerobactin iron uptake systems encoded by plasmids pColV-K30 in Escherichia coli K-12 and pSMN1 in Aerobacter aerogenes 62-1. J Bacteriol. 1988 Nov;170(11):5153–5160. doi: 10.1128/jb.170.11.5153-5160.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wayne R., Frick K., Neilands J. B. Siderophore protection against colicins M, B, V, and Ia in Escherichia coli. J Bacteriol. 1976 Apr;126(1):7–12. doi: 10.1128/jb.126.1.7-12.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams P. H. Novel iron uptake system specified by ColV plasmids: an important component in the virulence of invasive strains of Escherichia coli. Infect Immun. 1979 Dec;26(3):925–932. doi: 10.1128/iai.26.3.925-932.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams P. H., Warner P. J. ColV plasmid-mediated, colicin V-independent iron uptake system of invasive strains of Escherichia coli. Infect Immun. 1980 Aug;29(2):411–416. doi: 10.1128/iai.29.2.411-416.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Lorenzo V., Wee S., Herrero M., Neilands J. B. Operator sequences of the aerobactin operon of plasmid ColV-K30 binding the ferric uptake regulation (fur) repressor. J Bacteriol. 1987 Jun;169(6):2624–2630. doi: 10.1128/jb.169.6.2624-2630.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]