Abstract
Cellular insensitivity to vinca alkaloids is suggested to be primarily due to drug efflux by P-glycoprotein (P-gp). The anti-epileptic phenytoin (DPH), which does not bind to P-gp, can selectively enhance vincristine (VCR) cytotoxicity in wild-type (WT) or multidrug-resistant (MDR) cells. We now demonstrate that the protein phosphatase inhibitor okadaic acid (OKA) can mimic the effect of DPH by selectively enhancing cytotoxicity of vinblastine (VBL), but not taxol and doxorubicin, in human leukaemia HL-60 cells. Both DPH and OKA potentiate the anti-mitotic effects of VBL by enhanced damage to the mitotic spindle, resulting in prolonged growth arrest. Also, unlike VBL alone, in human leukaemia or non-small-cell lung carcinoma cells treated with VBL plus DPH, recovery from damage to the mitotic spindle is compromised in drug-free medium and cell death by apoptosis in interphase ensues. Since protein phosphatases are involved with the regulation of metaphase to anaphase transit of cells during the mitotic cycle, enhanced VBL cytotoxicity in the presence of DPH or OKA may involve effects during metaphase on the mitotic spindle tubulin leading to growth arrest and apoptosis in interphase. These novel results suggest that DPH or OKA could be powerful tools to study cellular effects of vinca alkaloids and possibly for the development of novel therapeutic strategies.
Full text
PDF
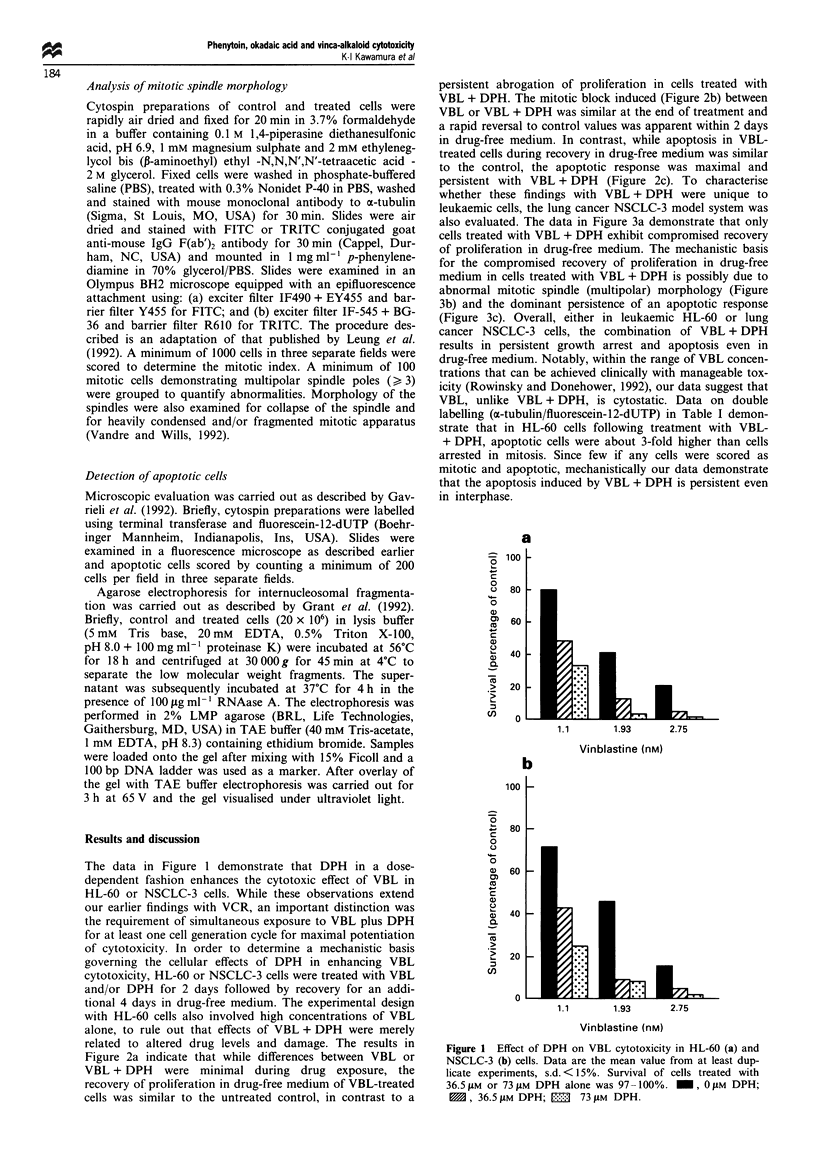
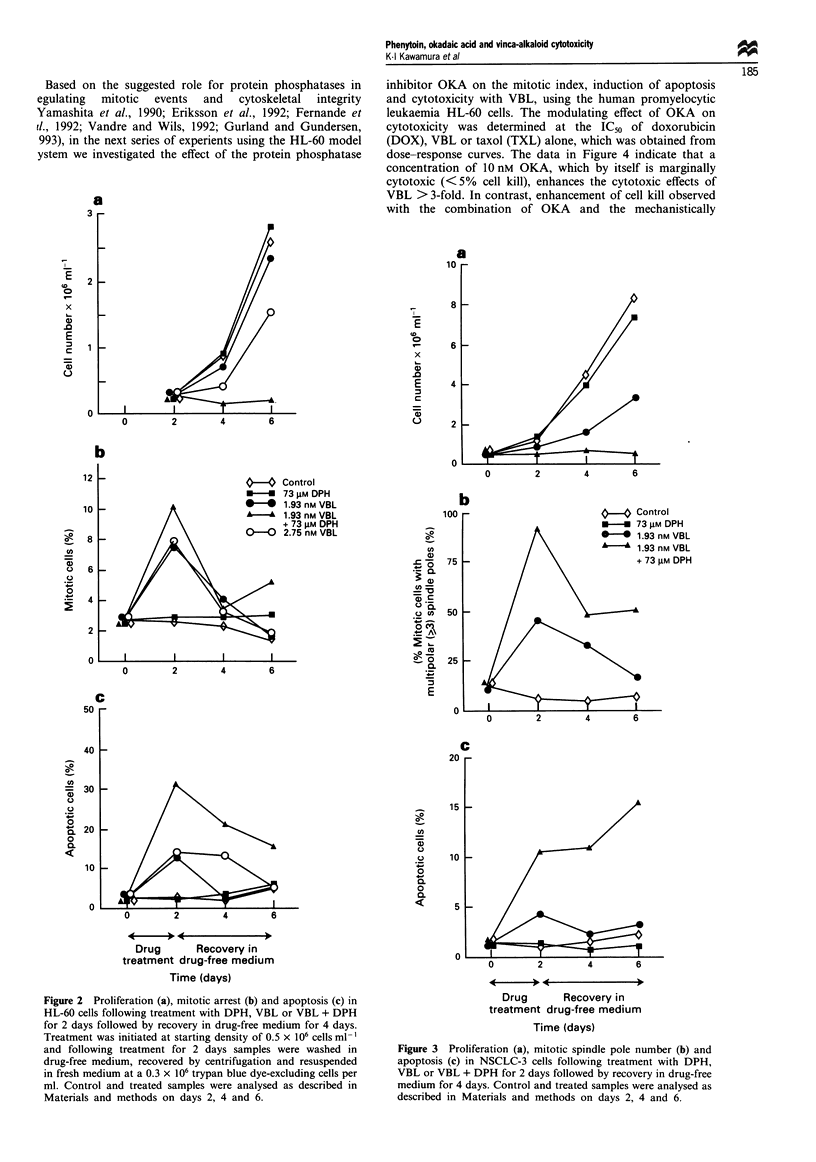
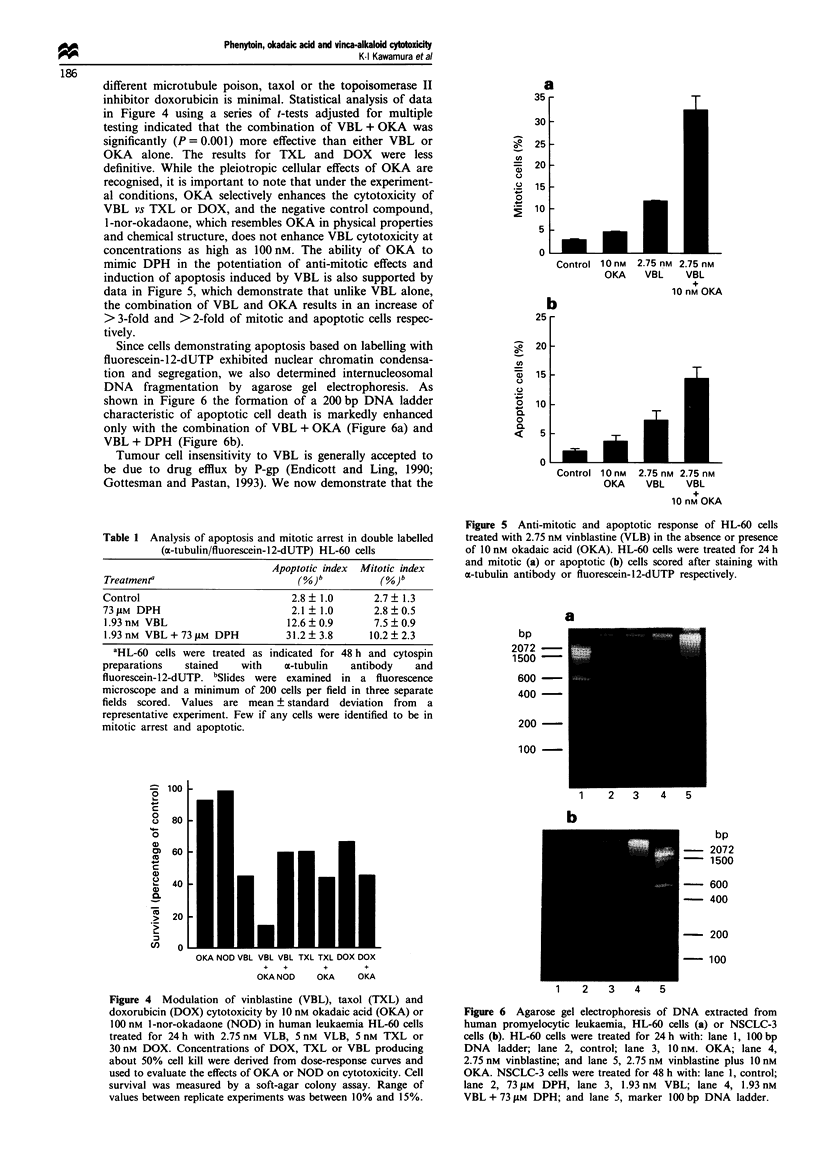


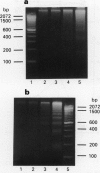
Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Chambers T. C., Raynor R. L., Kuo J. F. Multidrug-resistant human KB carcinoma cells are highly resistant to the protein phosphatase inhibitors okadaic acid and calyculin A. Analysis of potential mechanisms involved in toxin resistance. Int J Cancer. 1993 Jan 21;53(2):323–327. doi: 10.1002/ijc.2910530225. [DOI] [PubMed] [Google Scholar]
- Collins S. J. The HL-60 promyelocytic leukemia cell line: proliferation, differentiation, and cellular oncogene expression. Blood. 1987 Nov;70(5):1233–1244. [PubMed] [Google Scholar]
- Endicott J. A., Ling V. The biochemistry of P-glycoprotein-mediated multidrug resistance. Annu Rev Biochem. 1989;58:137–171. doi: 10.1146/annurev.bi.58.070189.001033. [DOI] [PubMed] [Google Scholar]
- Eriksson J. E., Brautigan D. L., Vallee R., Olmsted J., Fujiki H., Goldman R. D. Cytoskeletal integrity in interphase cells requires protein phosphatase activity. Proc Natl Acad Sci U S A. 1992 Nov 15;89(22):11093–11097. doi: 10.1073/pnas.89.22.11093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernandez A., Brautigan D. L., Lamb N. J. Protein phosphatase type 1 in mammalian cell mitosis: chromosomal localization and involvement in mitotic exit. J Cell Biol. 1992 Mar;116(6):1421–1430. doi: 10.1083/jcb.116.6.1421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ganapathi R., Hercbergs A., Grabowski D., Ford J. Selective enhancement of vincristine cytotoxicity in multidrug-resistant tumor cells by dilantin (phenytoin) Cancer Res. 1993 Jul 15;53(14):3262–3265. [PubMed] [Google Scholar]
- Gavrieli Y., Sherman Y., Ben-Sasson S. A. Identification of programmed cell death in situ via specific labeling of nuclear DNA fragmentation. J Cell Biol. 1992 Nov;119(3):493–501. doi: 10.1083/jcb.119.3.493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Georges E., Sharom F. J., Ling V. Multidrug resistance and chemosensitization: therapeutic implications for cancer chemotherapy. Adv Pharmacol. 1990;21:185–220. doi: 10.1016/s1054-3589(08)60343-9. [DOI] [PubMed] [Google Scholar]
- Gottesman M. M., Pastan I. Biochemistry of multidrug resistance mediated by the multidrug transporter. Annu Rev Biochem. 1993;62:385–427. doi: 10.1146/annurev.bi.62.070193.002125. [DOI] [PubMed] [Google Scholar]
- Grant S., Jarvis W. D., Swerdlow P. S., Turner A. J., Traylor R. S., Wallace H. J., Lin P. S., Pettit G. R., Gewirtz D. A. Potentiation of the activity of 1-beta-D-arabinofuranosylcytosine by the protein kinase C activator bryostatin 1 in HL-60 cells: association with enhanced fragmentation of mature DNA. Cancer Res. 1992 Nov 15;52(22):6270–6278. [PubMed] [Google Scholar]
- Gurland G., Gundersen G. G. Protein phosphatase inhibitors induce the selective breakdown of stable microtubules in fibroblasts and epithelial cells. Proc Natl Acad Sci U S A. 1993 Oct 1;90(19):8827–8831. doi: 10.1073/pnas.90.19.8827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kerr J. F., Winterford C. M., Harmon B. V. Apoptosis. Its significance in cancer and cancer therapy. Cancer. 1994 Apr 15;73(8):2013–2026. doi: 10.1002/1097-0142(19940415)73:8<2013::aid-cncr2820730802>3.0.co;2-j. [DOI] [PubMed] [Google Scholar]
- Leung M. F., Sokoloski J. A., Sartorelli A. C. Changes in microtubules, microtubule-associated proteins, and intermediate filaments during the differentiation of HL-60 leukemia cells. Cancer Res. 1992 Feb 15;52(4):949–954. [PubMed] [Google Scholar]
- MacKinney A. A., Vyas R. S., Walker D. Hydantoin drugs inhibit polymerization of pure microtubular protein. J Pharmacol Exp Ther. 1978 Jan;204(1):189–194. [PubMed] [Google Scholar]
- MacKinney A. A., Vyas R., Mueller C., Gorder C. A comparison of potency of hydantoins in metaphase arrest and inhibition of microtubular polymerization. Mol Pharmacol. 1980 Mar;17(2):275–278. [PubMed] [Google Scholar]
- Oren M. Relationship of p53 to the control of apoptotic cell death. Semin Cancer Biol. 1994 Jun;5(3):221–227. [PubMed] [Google Scholar]
- Vandré D. D., Wills V. L. Inhibition of mitosis by okadaic acid: possible involvement of a protein phosphatase 2A in the transition from metaphase to anaphase. J Cell Sci. 1992 Jan;101(Pt 1):79–91. doi: 10.1242/jcs.101.1.79. [DOI] [PubMed] [Google Scholar]
- Wells N. J., Addison C. M., Fry A. M., Ganapathi R., Hickson I. D. Serine 1524 is a major site of phosphorylation on human topoisomerase II alpha protein in vivo and is a substrate for casein kinase II in vitro. J Biol Chem. 1994 Nov 25;269(47):29746–29751. [PubMed] [Google Scholar]
- Wolf D., Rotter V. Major deletions in the gene encoding the p53 tumor antigen cause lack of p53 expression in HL-60 cells. Proc Natl Acad Sci U S A. 1985 Feb;82(3):790–794. doi: 10.1073/pnas.82.3.790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamashita K., Yasuda H., Pines J., Yasumoto K., Nishitani H., Ohtsubo M., Hunter T., Sugimura T., Nishimoto T. Okadaic acid, a potent inhibitor of type 1 and type 2A protein phosphatases, activates cdc2/H1 kinase and transiently induces a premature mitosis-like state in BHK21 cells. EMBO J. 1990 Dec;9(13):4331–4338. doi: 10.1002/j.1460-2075.1990.tb07882.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng B., Chambers T. C., Raynor R. L., Markham P. N., Gebel H. M., Vogler W. R., Kuo J. F. Human leukemia K562 cell mutant (K562/OA200) selected for resistance to okadaic acid (protein phosphatase inhibitor) lacks protein kinase C-epsilon, exhibits multidrug resistance phenotype, and expresses drug pump P-glycoprotein. J Biol Chem. 1994 Apr 22;269(16):12332–12338. [PubMed] [Google Scholar]