Abstract
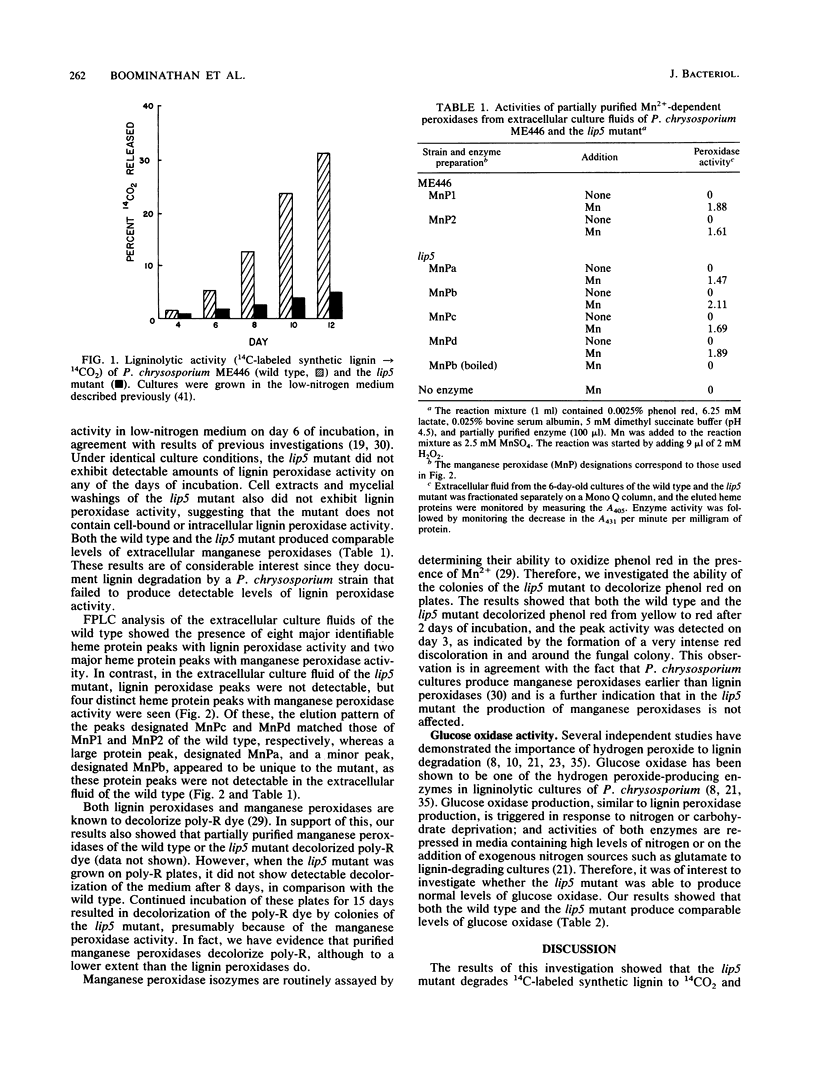
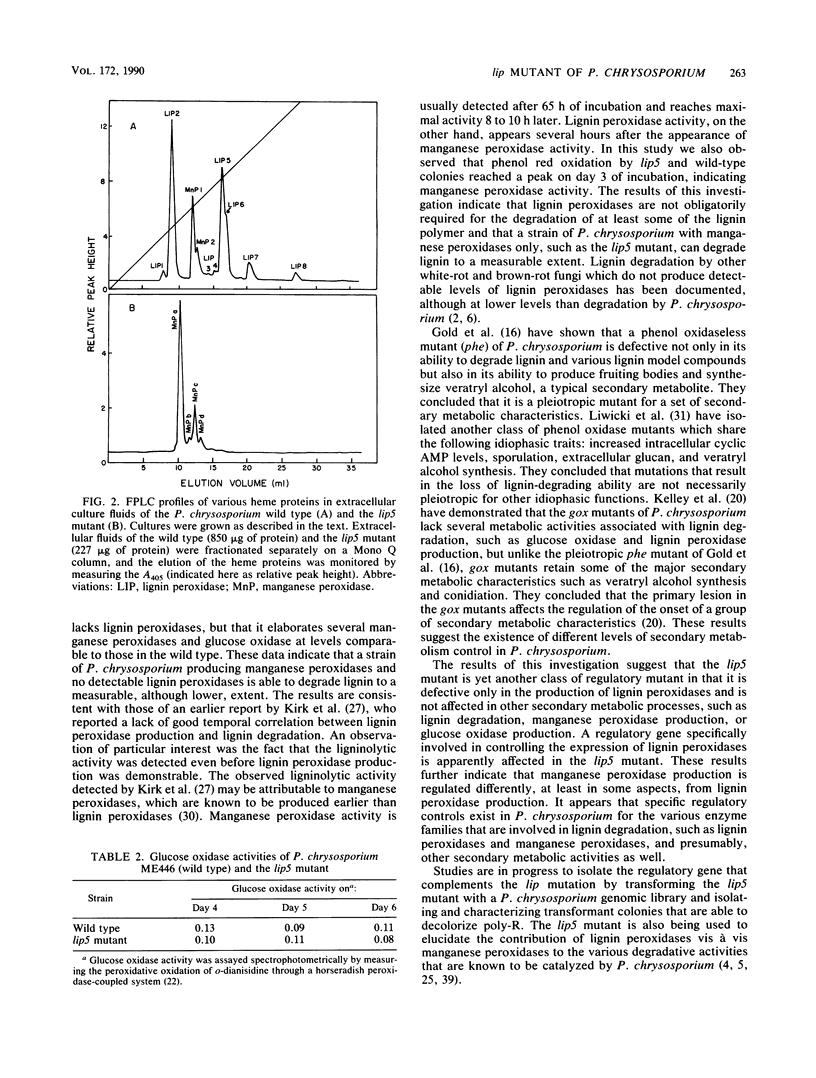
Phanerochaete chrysosporium produces two classes of extracellular heme proteins, designated lignin peroxidases and manganese peroxidases, that play a key role in lignin degradation. In this study we isolated and characterized a lignin peroxidase-negative mutant (lip mutant) that showed 16% of the ligninolytic activity (14C-labeled synthetic lignin----14CO2) exhibited by the wild type. The lip mutant did not produce detectable levels of lignin peroxidase, whereas the wild type, under identical conditions, produced 96 U of lignin peroxidase per liter. Both the wild type and the mutant produced comparable levels of manganese peroxidase and glucose oxidase, a key H2O2-generating secondary metabolic enzyme in P. chrysosporium. Fast protein liquid chromatographic analysis of the concentrated extracellular fluid of the lip mutant confirmed that it produced only heme proteins with manganese peroxidase activity but no detectable lignin peroxidase activity, whereas both lignin peroxidase and manganese peroxidase activities were produced by the wild type. The lip mutant appears to be a regulatory mutant that is defective in the production of all the lignin peroxidases.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bourbonnais R., Paice M. G. Veratryl alcohol oxidases from the lignin-degrading basidiomycete Pleurotus sajor-caju. Biochem J. 1988 Oct 15;255(2):445–450. doi: 10.1042/bj2550445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown A., Sims P. F., Raeder U., Broda P. Multiple ligninase-related genes from Phanerochaete chrysosporium. Gene. 1988 Dec 15;73(1):77–85. doi: 10.1016/0378-1119(88)90314-9. [DOI] [PubMed] [Google Scholar]
- Bumpus J. A. Biodegradation of polycyclic hydrocarbons by Phanerochaete chrysosporium. Appl Environ Microbiol. 1989 Jan;55(1):154–158. doi: 10.1128/aem.55.1.154-158.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faison B. D., Kirk T. K., Farrell R. L. Role of Veratryl Alcohol in Regulating Ligninase Activity in Phanerochaete chrysosporium. Appl Environ Microbiol. 1986 Aug;52(2):251–254. doi: 10.1128/aem.52.2.251-254.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forney L. J., Reddy C. A., Tien M., Aust S. D. The involvement of hydroxyl radical derived from hydrogen peroxide in lignin degradation by the white rot fungus Phanerochaete chrysosporium. J Biol Chem. 1982 Oct 10;257(19):11455–11462. [PubMed] [Google Scholar]
- Glenn J. K., Gold M. H. Decolorization of Several Polymeric Dyes by the Lignin-Degrading Basidiomycete Phanerochaete chrysosporium. Appl Environ Microbiol. 1983 Jun;45(6):1741–1747. doi: 10.1128/aem.45.6.1741-1747.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glenn J. K., Gold M. H. Purification and characterization of an extracellular Mn(II)-dependent peroxidase from the lignin-degrading basidiomycete, Phanerochaete chrysosporium. Arch Biochem Biophys. 1985 Nov 1;242(2):329–341. doi: 10.1016/0003-9861(85)90217-6. [DOI] [PubMed] [Google Scholar]
- Gold M. H., Kuwahara M., Chiu A. A., Glenn J. K. Purification and characterization of an extracellular H2O2-requiring diarylpropane oxygenase from the white rot basidiomycete, Phanerochaete chrysosporium. Arch Biochem Biophys. 1984 Nov 1;234(2):353–362. doi: 10.1016/0003-9861(84)90280-7. [DOI] [PubMed] [Google Scholar]
- Holzbaur E. L., Andrawis A., Tien M. Structure and regulation of a lignin peroxidase gene from Phanerochaete chrysosporium. Biochem Biophys Res Commun. 1988 Sep 15;155(2):626–633. doi: 10.1016/s0006-291x(88)80541-2. [DOI] [PubMed] [Google Scholar]
- Jäger A., Croan S., Kirk T. K. Production of Ligninases and Degradation of Lignin in Agitated Submerged Cultures of Phanerochaete chrysosporium. Appl Environ Microbiol. 1985 Nov;50(5):1274–1278. doi: 10.1128/aem.50.5.1274-1278.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelley R. L., Reddy C. A. Purification and characterization of glucose oxidase from ligninolytic cultures of Phanerochaete chrysosporium. J Bacteriol. 1986 Apr;166(1):269–274. doi: 10.1128/jb.166.1.269-274.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kersten P. J., Kirk T. K. Involvement of a new enzyme, glyoxal oxidase, in extracellular H2O2 production by Phanerochaete chrysosporium. J Bacteriol. 1987 May;169(5):2195–2201. doi: 10.1128/jb.169.5.2195-2201.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirk T. K., Farrell R. L. Enzymatic "combustion": the microbial degradation of lignin. Annu Rev Microbiol. 1987;41:465–505. doi: 10.1146/annurev.mi.41.100187.002341. [DOI] [PubMed] [Google Scholar]
- Leisola M. S., Kozulic B., Meussdoerffer F., Fiechter A. Homology among multiple extracellular peroxidases from Phanerochaete chrysosporium. J Biol Chem. 1987 Jan 5;262(1):419–424. [PubMed] [Google Scholar]
- Liwicki R., Paterson A., MacDonald M. J., Broda P. Phenotypic classes of phenoloxidase-negative mutants of the lignin-degrading fungus Phanerochaete chrysosporium. J Bacteriol. 1985 May;162(2):641–644. doi: 10.1128/jb.162.2.641-644.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paszczyński A., Huynh V. B., Crawford R. Comparison of ligninase-I and peroxidase-M2 from the white-rot fungus Phanerochaete chrysosporium. Arch Biochem Biophys. 1986 Feb 1;244(2):750–765. doi: 10.1016/0003-9861(86)90644-2. [DOI] [PubMed] [Google Scholar]
- Pribnow D., Mayfield M. B., Nipper V. J., Brown J. A., Gold M. H. Characterization of a cDNA encoding a manganese peroxidase, from the lignin-degrading basidiomycete Phanerochaete chrysosporium. J Biol Chem. 1989 Mar 25;264(9):5036–5040. [PubMed] [Google Scholar]
- Pruzansky J. J., Patterson R. Binding constants of IgE receptors on human blood basophils for IgE. Immunology. 1986 Jun;58(2):257–262. [PMC free article] [PubMed] [Google Scholar]
- Ramasamy K., Kelley R. L., Reddy C. A. Lack of lignin degradation by glucose oxidase-negative mutants of Phanerochaete chrysosporium. Biochem Biophys Res Commun. 1985 Aug 30;131(1):436–441. doi: 10.1016/0006-291x(85)91821-2. [DOI] [PubMed] [Google Scholar]
- Renganathan V., Miki K., Gold M. H. Multiple molecular forms of diarylpropane oxygenase, an H2O2-requiring, lignin-degrading enzyme from Phanerochaete chrysosporium. Arch Biochem Biophys. 1985 Aug 15;241(1):304–314. doi: 10.1016/0003-9861(85)90387-x. [DOI] [PubMed] [Google Scholar]
- Schalch H., Gaskell J., Smith T. L., Cullen D. Molecular cloning and sequences of lignin peroxidase genes of Phanerochaete chrysosporium. Mol Cell Biol. 1989 Jun;9(6):2743–2747. doi: 10.1128/mcb.9.6.2743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith T. L., Schalch H., Gaskell J., Covert S., Cullen D. Nucleotide sequence of a ligninase gene from Phanerochaete chrysosporium. Nucleic Acids Res. 1988 Feb 11;16(3):1219–1219. doi: 10.1093/nar/16.3.1219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tien M., Kirk T. K. Lignin-degrading enzyme from Phanerochaete chrysosporium: Purification, characterization, and catalytic properties of a unique H(2)O(2)-requiring oxygenase. Proc Natl Acad Sci U S A. 1984 Apr;81(8):2280–2284. doi: 10.1073/pnas.81.8.2280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tien M. Properties of ligninase from Phanerochaete chrysosporium and their possible applications. Crit Rev Microbiol. 1987;15(2):141–168. doi: 10.3109/10408418709104456. [DOI] [PubMed] [Google Scholar]
- Tien M., Tu C. P. Cloning and sequencing of a cDNA for a ligninase from Phanerochaete chrysosporium. Nature. 1987 Apr 2;326(6112):520–523. doi: 10.1038/326520a0. [DOI] [PubMed] [Google Scholar]
- Walther I., Kälin M., Reiser J., Suter F., Fritsche B., Saloheimo M., Leisola M., Teeri T., Knowles J. K., Fiechter A. Molecular analysis of a Phanerochaete chrysosporium lignin peroxidase gene. Gene. 1988 Oct 15;70(1):127–137. doi: 10.1016/0378-1119(88)90111-4. [DOI] [PubMed] [Google Scholar]
- Wariishi H., Akileswaran L., Gold M. H. Manganese peroxidase from the basidiomycete Phanerochaete chrysosporium: spectral characterization of the oxidized states and the catalytic cycle. Biochemistry. 1988 Jul 12;27(14):5365–5370. doi: 10.1021/bi00414a061. [DOI] [PubMed] [Google Scholar]
- Zhang Y. Z., Zylstra G. J., Olsen R. H., Reddy C. A. Identification of cDNA clones for ligninase from Phanerochaete chrysosporium using synthetic oligonucleotide probes. Biochem Biophys Res Commun. 1986 Jun 13;137(2):649–656. doi: 10.1016/0006-291x(86)91127-7. [DOI] [PubMed] [Google Scholar]
- de Boer H. A., Zhang Y. Z., Collins C., Reddy C. A. Analysis of nucleotide sequences of two ligninase cDNAs from a white-rot filamentous fungus, Phanerochaete chrysosporium. Gene. 1987;60(1):93–102. doi: 10.1016/0378-1119(87)90217-4. [DOI] [PubMed] [Google Scholar]


