Abstract
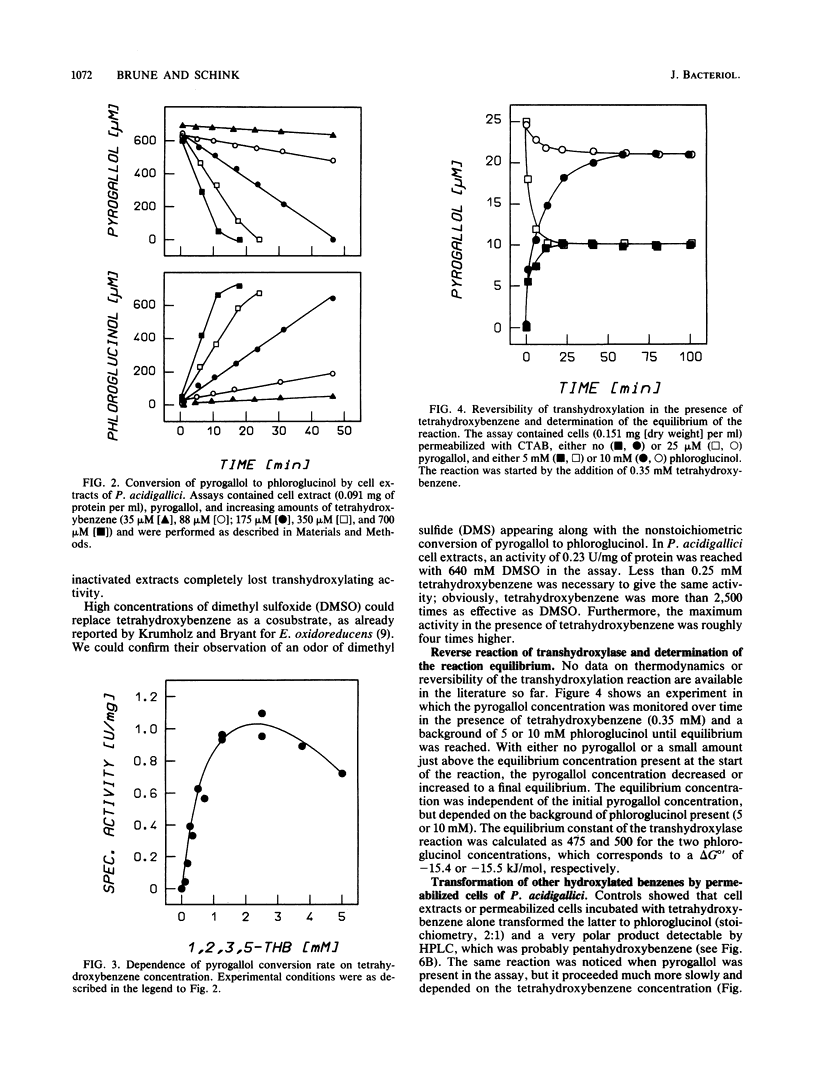
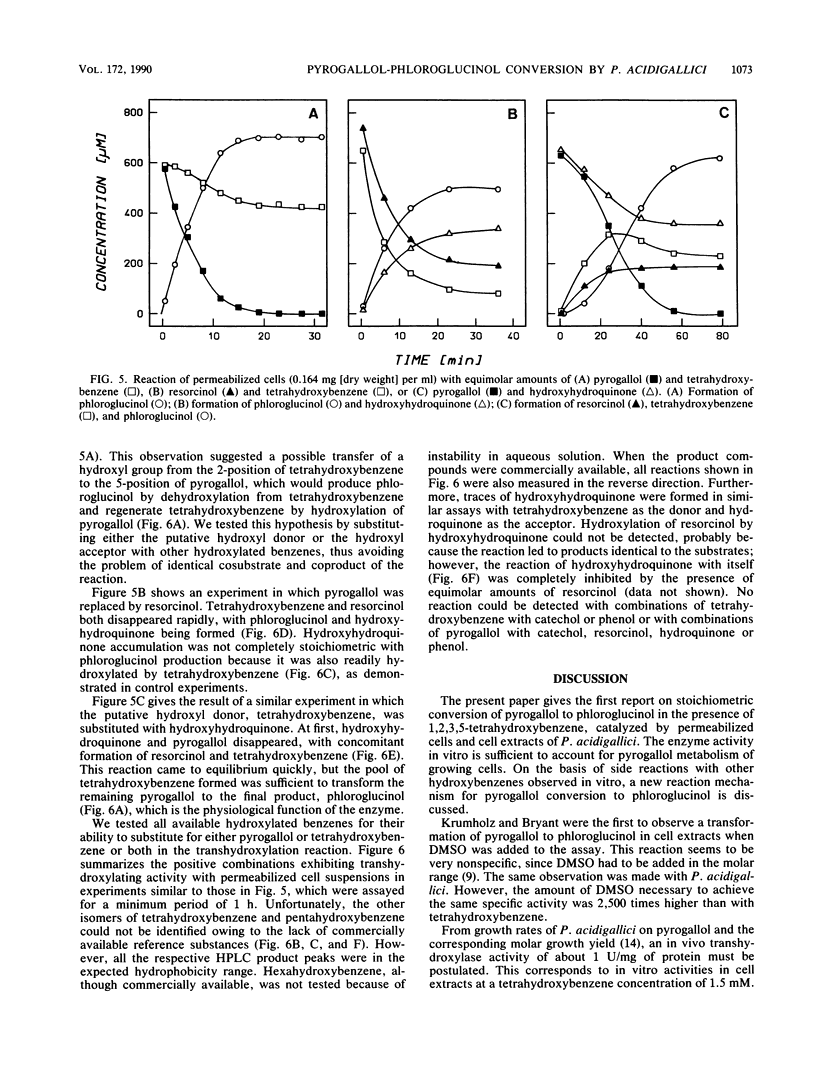
Permeabilized cells and cell extracts of Pelobacter acidigallici catalyzed the conversion of pyrogallol (1,2,3-trihydroxybenzene) to phloroglucinol (1,3,5-trihydroxybenzene) in the presence of 1,2,3,5-tetrahydroxybenzene. Pyrogallol consumption by resting cells stopped after lysis by French press or mild detergent (cetyltrimethylammonium bromide [CTAB]) treatment. Addition of 1,2,3,5-tetrahydroxybenzene to the assay mixture restored pyrogallol consumption and led to stoichiometric phloroglucinol accumulation. The stoichiometry of pyrogallol conversion to phloroglucinol was independent of the amount of tetrahydroxybenzene added. The tetrahydroxybenzene concentration limited the velocity of the transhydroxylation reaction, which reached a maximum at 1.5 mM tetrahydroxybenzene (1 U/mg of protein). Transhydroxylation was shown to be reversible. The equilibrium constant of the reaction was determined, and the free-energy change (delta G degree') of phloroglucinol formation from pyrogallol was calculated to be -15.5 kJ/mol. Permeabilized cells and cell extracts also catalyzed the transfer of hydroxyl moieties between other hydroxylated benzenes. Tetrahydroxybenzene and hydroxyhydroquinone participated as hydroxyl donors and as hydroxyl acceptors in the reaction, whereas pyrogallol, resorcinol, and phloroglucinol were hydroxylated by both donors. A novel mechanism deduced from these data involves intermolecular transfer of the hydroxyl moiety from the cosubstrate (1,2,3,5-tetrahydroxybenzene) to the substrate (pyrogallol), thus forming the product (phloroglucinol) and regenerating the cosubstrate.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Evans W. C., Fuchs G. Anaerobic degradation of aromatic compounds. Annu Rev Microbiol. 1988;42:289–317. doi: 10.1146/annurev.mi.42.100188.001445. [DOI] [PubMed] [Google Scholar]
- Haddock J. D., Ferry J. G. Purification and properties of phloroglucinol reductase from Eubacterium oxidoreducens G-41. J Biol Chem. 1989 Mar 15;264(8):4423–4427. [PubMed] [Google Scholar]
- Jamieson W. D., Taylor A., Mathur D. K., Blackwood A. C. Identification of an intermediate of phloroglucinol degradation by mass spectroscopic analysis. Can J Biochem. 1970 Feb;48(2):215–216. doi: 10.1139/o70-036. [DOI] [PubMed] [Google Scholar]
- Krumholz L. R., Bryant M. P. Characterization of the pyrogallol-phloroglucinol isomerase of Eubacterium oxidoreducens. J Bacteriol. 1988 Jun;170(6):2472–2479. doi: 10.1128/jb.170.6.2472-2479.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krumholz L. R., Crawford R. L., Hemling M. E., Bryant M. P. Metabolism of gallate and phloroglucinol in Eubacterium oxidoreducens via 3-hydroxy-5-oxohexanoate. J Bacteriol. 1987 May;169(5):1886–1890. doi: 10.1128/jb.169.5.1886-1890.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McEwan A. G., Ferguson S. J., Jackson J. B. Electron flow to dimethylsulphoxide or trimethylamine-N-oxide generates a membrane potential in Rhodopseudomonas capsulata. Arch Microbiol. 1983 Dec;136(4):300–305. doi: 10.1007/BF00425221. [DOI] [PubMed] [Google Scholar]
- Patel T. R., Jure K. G., Jones G. A. Catabolism of phloroglucinol by the rumen anaerobe coprococcus. Appl Environ Microbiol. 1981 Dec;42(6):1010–1017. doi: 10.1128/aem.42.6.1010-1017.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thauer R. K., Jungermann K., Decker K. Energy conservation in chemotrophic anaerobic bacteria. Bacteriol Rev. 1977 Mar;41(1):100–180. doi: 10.1128/br.41.1.100-180.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsai C. G., Jones G. A. Isolation and identification of rumen bacteria capable of anaerobic phloroglucinol degradation. Can J Microbiol. 1975 Jun;21(6):794–801. doi: 10.1139/m75-117. [DOI] [PubMed] [Google Scholar]
- Whittle P. J., Lunt D. O., Evans W. C. Anaerobic photometabolism of aromatic compounds by Rhodopseudomonas sp. Biochem Soc Trans. 1976;4(3):490–491. doi: 10.1042/bst0040490. [DOI] [PubMed] [Google Scholar]
- Widdel F., Pfennig N. Studies on dissimilatory sulfate-reducing bacteria that decompose fatty acids. I. Isolation of new sulfate-reducing bacteria enriched with acetate from saline environments. Description of Desulfobacter postgatei gen. nov., sp. nov. Arch Microbiol. 1981 Jul;129(5):395–400. doi: 10.1007/BF00406470. [DOI] [PubMed] [Google Scholar]
- Wood P. M. The redox potential for dimethyl sulphoxide reduction to dimethyl sulphide: evaluation and biochemical implications. FEBS Lett. 1981 Feb 9;124(1):11–14. doi: 10.1016/0014-5793(81)80042-7. [DOI] [PubMed] [Google Scholar]
- Zinder S. H., Brock T. D. Dimethyl sulfoxide as an electron acceptor for anaerobic growth. Arch Microbiol. 1978 Jan 23;116(1):35–40. doi: 10.1007/BF00408731. [DOI] [PubMed] [Google Scholar]


