Abstract
Type 4 fimbriae (pili) are found in a wide variety of gram-negative bacteria and are composed of small structural subunits which share significant sequence homology among different species, especially at their amino-terminal ends. Previous studies demonstrating morphogenetic expression of Bacteroides nodosus fimbriae from cloned subunit genes in Pseudomonas aeruginosa suggested that there is a common mechanism for type 4 fimbriae assembly and that the structural subunits are interchangeable (J. S. Mattick et al., J. Bacteriol. 169:33-41, 1987). Here we have examined the expression of Moraxella bovis fimbrial subunits in P. aeruginosa. M. bovis subunits were assembled into extracellular fimbriae in this host, in some cases as a homopolymer but in others as a mosaic with the indigenous subunit, indicating structural equivalence. This result contrasts with other studies in which recombinant P. aeruginosa expressing different subunits produced fimbriae composed almost exclusively of one subunit or the other (T. C. Elleman and J. E. Peterson, Mol. Microbiol. 1:377-380, 1987). Both observations can be explained by reversibility of subunit-subunit interactions at the site of assembly, with the forward equilibrium favoring chain extension between compatible subunits.
Full text
PDF
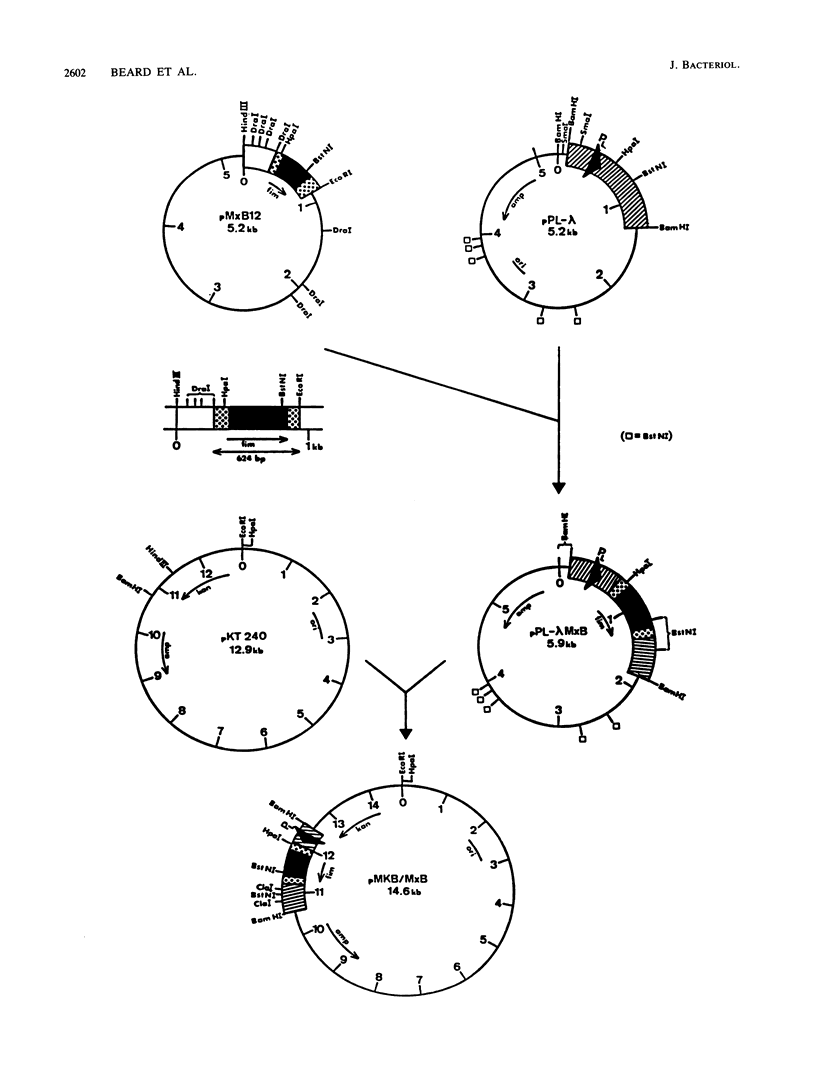





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anderson B. J., Bills M. M., Egerton J. R., Mattick J. S. Cloning and expression in Escherichia coli of the gene encoding the structural subunit of Bacteroides nodosus fimbriae. J Bacteriol. 1984 Nov;160(2):748–754. doi: 10.1128/jb.160.2.748-754.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson B. J., Kristo C. L., Egerton J. R., Mattick J. S. Variation in the structural subunit and basal protein antigens of Bacteroides nodosus fimbriae. J Bacteriol. 1986 May;166(2):453–460. doi: 10.1128/jb.166.2.453-460.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bagdasarian M. M., Amann E., Lurz R., Rückert B., Bagdasarian M. Activity of the hybrid trp-lac (tac) promoter of Escherichia coli in Pseudomonas putida. Construction of broad-host-range, controlled-expression vectors. Gene. 1983 Dec;26(2-3):273–282. doi: 10.1016/0378-1119(83)90197-x. [DOI] [PubMed] [Google Scholar]
- Bradley D. E. A function of Pseudomonas aeruginosa PAO polar pili: twitching motility. Can J Microbiol. 1980 Feb;26(2):146–154. doi: 10.1139/m80-022. [DOI] [PubMed] [Google Scholar]
- Bradley D. E. The adsorption of Pseudomonas aeruginosa pilus-dependent bacteriophages to a host mutant with nonretractile pili. Virology. 1974 Mar;58(1):149–163. doi: 10.1016/0042-6822(74)90150-0. [DOI] [PubMed] [Google Scholar]
- Dalrymple B., Mattick J. S. An analysis of the organization and evolution of type 4 fimbrial (MePhe) subunit proteins. J Mol Evol. 1987;25(3):261–269. doi: 10.1007/BF02100020. [DOI] [PubMed] [Google Scholar]
- Egerton J. R., Cox P. T., Anderson B. J., Kristo C., Norman M., Mattick J. S. Protection of sheep against footrot with a recombinant DNA-based fimbrial vaccine. Vet Microbiol. 1987 Sep;14(4):393–409. doi: 10.1016/0378-1135(87)90030-7. [DOI] [PubMed] [Google Scholar]
- Elleman T. C., Hoyne P. A., McKern N. M., Stewart D. J. Nucleotide sequence of the gene encoding the two-subunit pilin of Bacteroides nodosus 265. J Bacteriol. 1986 Jul;167(1):243–250. doi: 10.1128/jb.167.1.243-250.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elleman T. C., Peterson J. E. Expression of multiple types of N-methyl Phe pili in Pseudomonas aeruginosa. Mol Microbiol. 1987 Nov;1(3):377–380. doi: 10.1111/j.1365-2958.1987.tb01945.x. [DOI] [PubMed] [Google Scholar]
- HOLLOWAY B. W. VARIATIONS IN RESTRICTION AND MODIFICATION OF BACTERIOPHAGE FOLLOWING INCREASE OF GROWTH TEMPERATURE OF PSEUDOMONAS AERUGINOSA. Virology. 1965 Apr;25:634–642. doi: 10.1016/0042-6822(65)90091-7. [DOI] [PubMed] [Google Scholar]
- Johnson K., Lory S. Characterization of Pseudomonas aeruginosa mutants with altered piliation. J Bacteriol. 1987 Dec;169(12):5663–5667. doi: 10.1128/jb.169.12.5663-5667.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marrs C. F., Schoolnik G., Koomey J. M., Hardy J., Rothbard J., Falkow S. Cloning and sequencing of a Moraxella bovis pilin gene. J Bacteriol. 1985 Jul;163(1):132–139. doi: 10.1128/jb.163.1.132-139.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mattick J. S., Anderson B. J., Mott M. R., Egerton J. R. Isolation and characterization of Bacteroides nodosus fimbriae: structural subunit and basal protein antigens. J Bacteriol. 1984 Nov;160(2):740–747. doi: 10.1128/jb.160.2.740-747.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mattick J. S., Bills M. M., Anderson B. J., Dalrymple B., Mott M. R., Egerton J. R. Morphogenetic expression of Bacteroides nodosus fimbriae in Pseudomonas aeruginosa. J Bacteriol. 1987 Jan;169(1):33–41. doi: 10.1128/jb.169.1.33-41.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ottow J. C. Ecology, physiology, and genetics of fimbriae and pili. Annu Rev Microbiol. 1975;29:79–108. doi: 10.1146/annurev.mi.29.100175.000455. [DOI] [PubMed] [Google Scholar]
- Pasloske B. L., Scraba D. G., Paranchych W. Assembly of mutant pilins in Pseudomonas aeruginosa: formation of pili composed of heterologous subunits. J Bacteriol. 1989 Apr;171(4):2142–2147. doi: 10.1128/jb.171.4.2142-2147.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sastry P. A., Finlay B. B., Pasloske B. L., Paranchych W., Pearlstone J. R., Smillie L. B. Comparative studies of the amino acid and nucleotide sequences of pilin derived from Pseudomonas aeruginosa PAK and PAO. J Bacteriol. 1985 Nov;164(2):571–577. doi: 10.1128/jb.164.2.571-577.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schoolnik G. K., Fernandez R., Tai J. Y., Rothbard J., Gotschlich E. C. Gonococcal pili. Primary structure and receptor binding domain. J Exp Med. 1984 May 1;159(5):1351–1370. doi: 10.1084/jem.159.5.1351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor R. K., Miller V. L., Furlong D. B., Mekalanos J. J. Use of phoA gene fusions to identify a pilus colonization factor coordinately regulated with cholera toxin. Proc Natl Acad Sci U S A. 1987 May;84(9):2833–2837. doi: 10.1073/pnas.84.9.2833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watts T. H., Worobec E. A., Paranchych W. Identification of pilin pools in the membranes of Pseudomonas aeruginosa. J Bacteriol. 1982 Nov;152(2):687–691. doi: 10.1128/jb.152.2.687-691.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]




