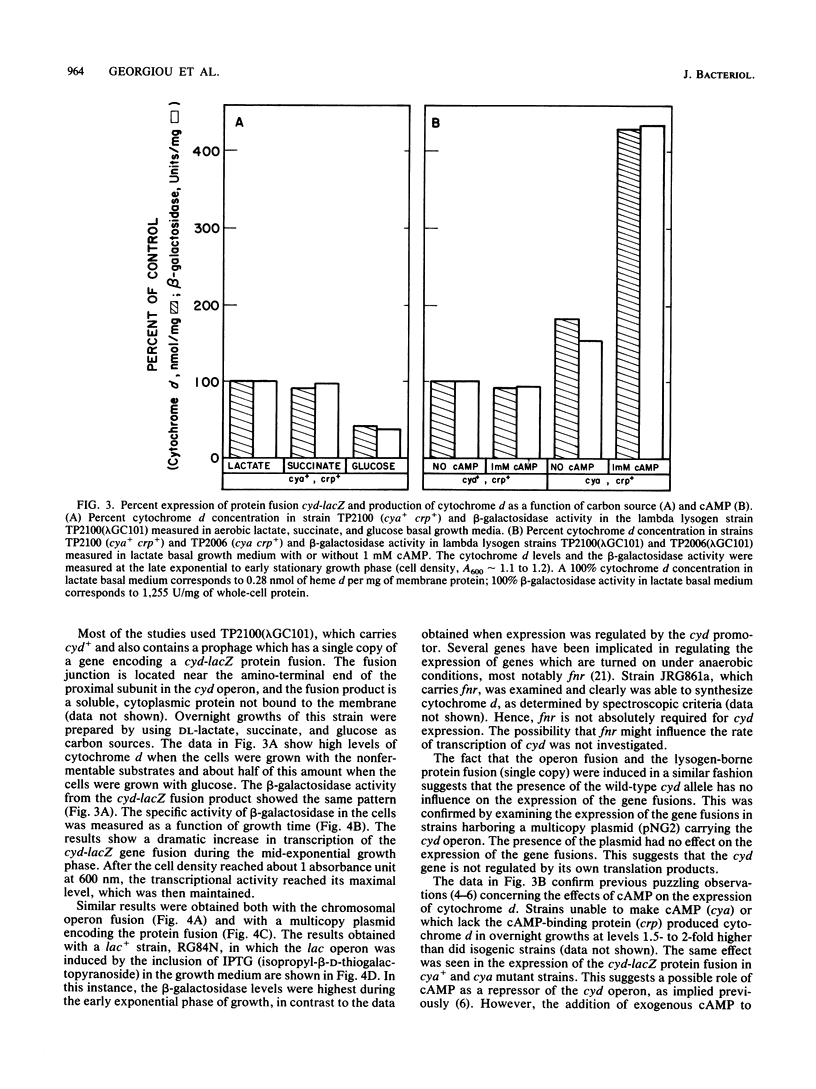
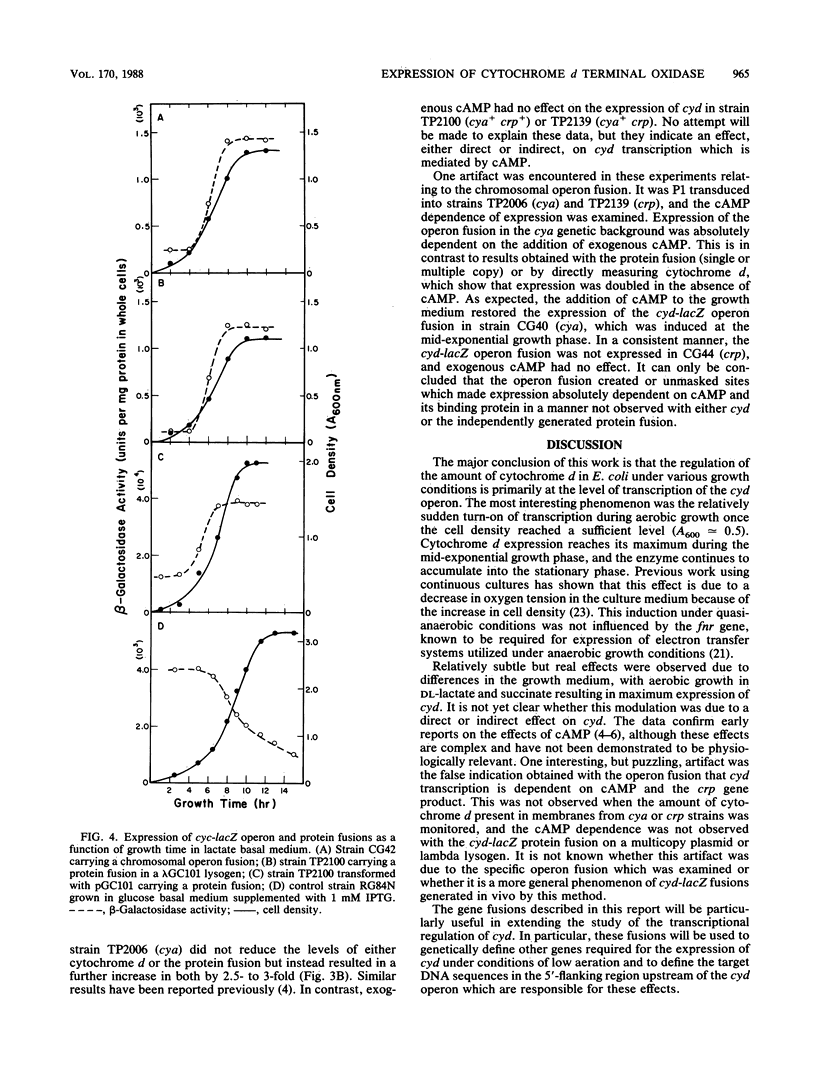
Abstract
The cytochrome d complex is one of the two terminal oxidases in the aerobic respiratory system of Escherichia coli. This enzyme is not present in cells grown with high levels of dissolved oxygen in the culture medium but accumulates after mid-exponential growth, reaching high levels in stationary-phase cells. In this study, the transcriptional activity of the cyd operon, encoding the two subunits of the enzyme, was examined under a variety of growth conditions. This was accomplished by the use of a chromosomal operon fusion, cyd-lacZ, generated in vivo by a lambda plac-Mu hopper bacteriophage and also by the use of a cyd-lacZ protein fusion created in vitro on a plasmid, transferred onto a lambda transducing phage, and examined as a single-copy lysogen. Transcription of the gene fusions was monitored by determination of beta-galactosidase activity. The data clearly show that cyd is transcriptionally regulated and that induction is observed when the culture reaches a sufficient cell density so as to substantially reduce the steady-state levels of dissolved oxygen. The transcriptional activity is also regulated by other growth conditions, including the carbon source. The turn-on of cyd under semianaerobic conditions does not require the fnr gene product, cyclic AMP, or the cyclic AMP-binding protein.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ashcroft J. R., Haddock B. A. Synthesis of alternative membrane-bound redox carriers during aerobic growth of Escherichia coli in the presence of potassium cyanide. Biochem J. 1975 May;148(2):349–352. doi: 10.1042/bj1480349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Au D. C., Lorence R. M., Gennis R. B. Isolation and characterization of an Escherichia coli mutant lacking the cytochrome o terminal oxidase. J Bacteriol. 1985 Jan;161(1):123–127. doi: 10.1128/jb.161.1.123-127.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bremer E., Silhavy T. J., Weinstock G. M. Transposable lambda placMu bacteriophages for creating lacZ operon fusions and kanamycin resistance insertions in Escherichia coli. J Bacteriol. 1985 Jun;162(3):1092–1099. doi: 10.1128/jb.162.3.1092-1099.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Broman R. L., Dobrogosz W. J. Stimulation of cytochrome synthesis in Escherichia coli by cyclic AMP. Arch Biochem Biophys. 1974 Jun;162(2):595–601. doi: 10.1016/0003-9861(74)90220-3. [DOI] [PubMed] [Google Scholar]
- Dallas W. S., Tseng Y., Dobrogosz W. J. Regulation of membrane functions and fatty acid composition in Escherichia coli by cyclic AMP receptor protein. Arch Biochem Biophys. 1976 Jul;175(1):295–302. doi: 10.1016/0003-9861(76)90511-7. [DOI] [PubMed] [Google Scholar]
- Daoud M. S., Haddock B. A. Electron transport in mutants of Escherichia coli deficient in their ability to synthesize adenosine 3':5'-cyclic monophosphate and the catabolite-gene activator protein. Biochem Soc Trans. 1976;4(4):711–714. doi: 10.1042/bst0040711. [DOI] [PubMed] [Google Scholar]
- Georgiou C. D., Fang H., Gennis R. B. Identification of the cydC locus required for expression of the functional form of the cytochrome d terminal oxidase complex in Escherichia coli. J Bacteriol. 1987 May;169(5):2107–2112. doi: 10.1128/jb.169.5.2107-2112.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green G. N., Gennis R. B. Isolation and characterization of an Escherichia coli mutant lacking cytochrome d terminal oxidase. J Bacteriol. 1983 Jun;154(3):1269–1275. doi: 10.1128/jb.154.3.1269-1275.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green G. N., Kranz J. E., Gennis R. B. Cloning the cyd gene locus coding for the cytochrome d complex of Escherichia coli. Gene. 1984 Dec;32(1-2):99–106. doi: 10.1016/0378-1119(84)90037-4. [DOI] [PubMed] [Google Scholar]
- Hanahan D. Studies on transformation of Escherichia coli with plasmids. J Mol Biol. 1983 Jun 5;166(4):557–580. doi: 10.1016/s0022-2836(83)80284-8. [DOI] [PubMed] [Google Scholar]
- Hattori M., Sakaki Y. Dideoxy sequencing method using denatured plasmid templates. Anal Biochem. 1986 Feb 1;152(2):232–238. doi: 10.1016/0003-2697(86)90403-3. [DOI] [PubMed] [Google Scholar]
- Ingledew W. J., Poole R. K. The respiratory chains of Escherichia coli. Microbiol Rev. 1984 Sep;48(3):222–271. doi: 10.1128/mr.48.3.222-271.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jamieson D. J., Higgins C. F. Two genetically distinct pathways for transcriptional regulation of anaerobic gene expression in Salmonella typhimurium. J Bacteriol. 1986 Oct;168(1):389–397. doi: 10.1128/jb.168.1.389-397.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koland J. G., Miller M. J., Gennis R. B. Reconstitution of the membrane-bound, ubiquinone-dependent pyruvate oxidase respiratory chain of Escherichia coli with the cytochrome d terminal oxidase. Biochemistry. 1984 Jan 31;23(3):445–453. doi: 10.1021/bi00298a008. [DOI] [PubMed] [Google Scholar]
- Kranz R. G., Barassi C. A., Gennis R. B. Immunological analysis of the heme proteins present in aerobically grown Escherichia coli. J Bacteriol. 1984 Jun;158(3):1191–1194. doi: 10.1128/jb.158.3.1191-1194.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lowry C. V., Lieber R. H. Negative regulation of the Saccharomyces cerevisiae ANB1 gene by heme, as mediated by the ROX1 gene product. Mol Cell Biol. 1986 Dec;6(12):4145–4148. doi: 10.1128/mcb.6.12.4145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manoil C., Beckwith J. TnphoA: a transposon probe for protein export signals. Proc Natl Acad Sci U S A. 1985 Dec;82(23):8129–8133. doi: 10.1073/pnas.82.23.8129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ostrow K. S., Silhavy T. J., Garrett S. cis-acting sites required for osmoregulation of ompF expression in Escherichia coli K-12. J Bacteriol. 1986 Dec;168(3):1165–1171. doi: 10.1128/jb.168.3.1165-1171.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poole R. K., Haddock B. A. Effects of sulphate-limited growth in continuous culture on the electron-transport chain and energy conservation in Escherichia coli K12. Biochem J. 1975 Dec;152(3):537–546. doi: 10.1042/bj1520537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rice C. W., Hempfling W. P. Oxygen-limited continuous culture and respiratory energy conservation in Escherichia coli. J Bacteriol. 1978 Apr;134(1):115–124. doi: 10.1128/jb.134.1.115-124.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roy A., Haziza C., Danchin A. Regulation of adenylate cyclase synthesis in Escherichia coli: nucleotide sequence of the control region. EMBO J. 1983;2(5):791–797. doi: 10.1002/j.1460-2075.1983.tb01502.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winkelman J. W., Clark D. P. Anaerobically induced genes of Escherichia coli. J Bacteriol. 1986 Jul;167(1):362–367. doi: 10.1128/jb.167.1.362-367.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zagorec M., Labbe-Bois R. Negative control of yeast coproporphyrinogen oxidase synthesis by heme and oxygen. J Biol Chem. 1986 Feb 25;261(6):2506–2509. [PubMed] [Google Scholar]


