Abstract
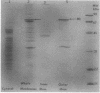
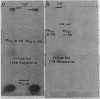
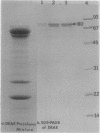
Under low-iron conditions, Paracoccus denitrificans excretes a catecholamine siderophore, L-parabactin, to sequester and utilize iron. In this report, we demonstrate the presence of stereospecific high-affinity ferric L-parabactin-binding activity associated with P. denitrificans membranes grown in low-iron medium. Isolated outer membrane components were shown to be three to four times higher in specific activity for ferric L-parabactin. The same amount of binding activity existed whether or not the radiolabel was present in the metal (55Fe) or the ligand (3H) portion of ferric parabactin chelate, suggesting that binding was to the intact complex. Ion-exchange chromatography of a Triton X-100-solubilized outer membrane mixture on DEAE-cellulose resulted in a 10-fold increase in binding activity relative to that present in whole membranes. Polypeptide profiles by sodium dodecyl sulfate-polyacrylamide gel electrophoresis of the products of each stage of the purification showed that binding activity copurified with one or more of the low-iron-induced outer membrane proteins in the 80-kilodalton (kDa) region. Membrane proteins and [55Fe]ferric L-parabactin electrophoresed in nondenaturing gels demonstrated the presence of membrane component(s) which stereo-specifically bound ferric L-parabactin, thus providing independent confirmation of the binding assay results. Moreover, when the band labeled by [55Fe]ferric L-parabactin was excised and profiled by sodium dodecyl sulfate-polyacrylamide gel electrophoresis, 80-kDa polypeptides were the major components present. These results demonstrate the presence of a high-affinity ferric L-parabactin receptor in P. denitrificans membranes and suggest that one or more of the 80-kDa low-iron-induced polypeptides are components of the ferric L-parabactin receptor.
Full text
PDF


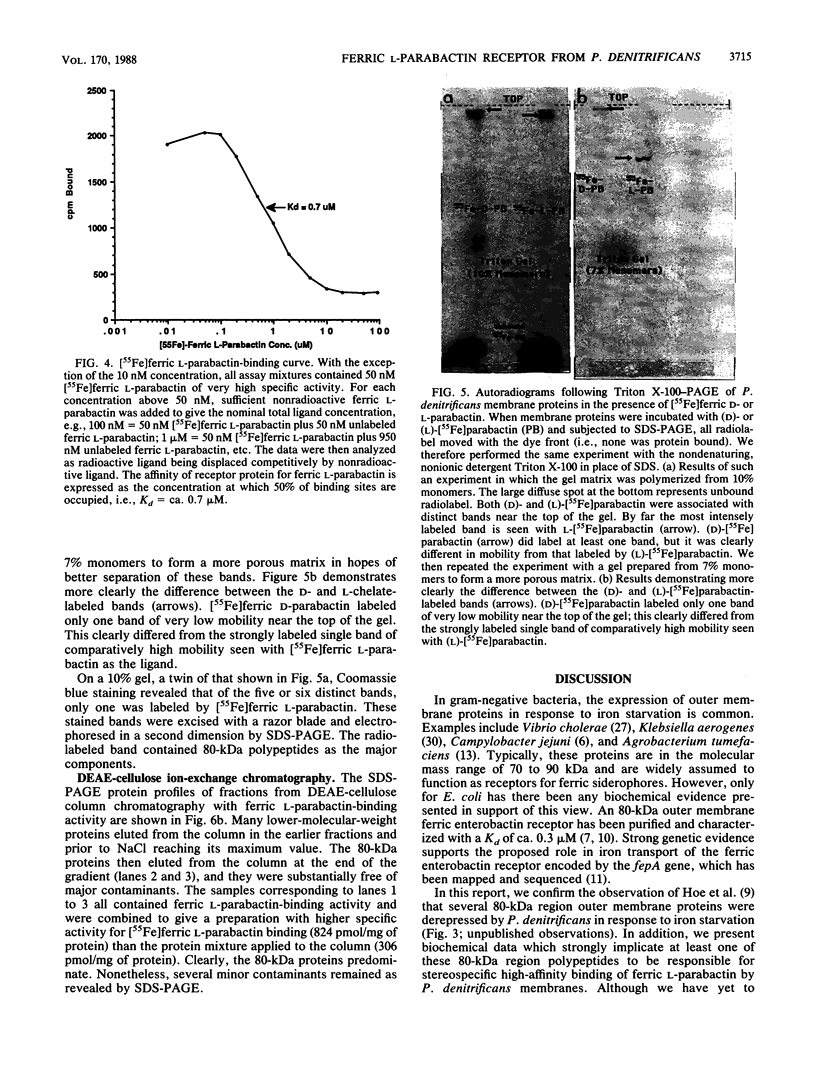


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Akera T., Cheng V. K. A simple method for the determination of affinity and binding site concentration in receptor binding studies. Biochim Biophys Acta. 1977 Nov 1;470(3):412–423. doi: 10.1016/0005-2736(77)90132-8. [DOI] [PubMed] [Google Scholar]
- Bergeron R. J., Dionis J. B., Elliott G. T., Kline S. J. Mechanism and stereospecificity of the parabactin-mediated iron-transport system in Paracoccus denitrificans. J Biol Chem. 1985 Jul 5;260(13):7936–7944. [PubMed] [Google Scholar]
- Field L. H., Headley V. L., Payne S. M., Berry L. J. Influence of iron on growth, morphology, outer membrane protein composition, and synthesis of siderophores in Campylobacter jejuni. Infect Immun. 1986 Oct;54(1):126–132. doi: 10.1128/iai.54.1.126-132.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fiss E. H., Stanley-Samuelson P., Neilands J. B. Properties and proteolysis of ferric enterobactin outer membrane receptor in Escherichia coli K12. Biochemistry. 1982 Aug 31;21(18):4517–4522. doi: 10.1021/bi00261a050. [DOI] [PubMed] [Google Scholar]
- Hollifield W. C., Jr, Neilands J. B. Ferric enterobactin transport system in Escherichia coli K-12. Extraction, assay, and specificity of the outer membrane receptor. Biochemistry. 1978 May 16;17(10):1922–1928. doi: 10.1021/bi00603a019. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Leong S. A., Neilands J. B. Relationship of siderophore-mediated iron assimilation to virulence in crown gall disease. J Bacteriol. 1981 Aug;147(2):482–491. doi: 10.1128/jb.147.2.482-491.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markwell M. A., Haas S. M., Bieber L. L., Tolbert N. E. A modification of the Lowry procedure to simplify protein determination in membrane and lipoprotein samples. Anal Biochem. 1978 Jun 15;87(1):206–210. doi: 10.1016/0003-2697(78)90586-9. [DOI] [PubMed] [Google Scholar]
- Neilands J. B. Iron absorption and transport in microorganisms. Annu Rev Nutr. 1981;1:27–46. doi: 10.1146/annurev.nu.01.070181.000331. [DOI] [PubMed] [Google Scholar]
- Ong S. A., Peterson T., Neilands J. B. Agrobactin, a siderophore from Agrobacterium tumefaciens. J Biol Chem. 1979 Mar 25;254(6):1860–1865. [PubMed] [Google Scholar]
- Penefsky H. S. Reversible binding of Pi by beef heart mitochondrial adenosine triphosphatase. J Biol Chem. 1977 May 10;252(9):2891–2899. [PubMed] [Google Scholar]
- Reenstra W. W., Patel L., Rottenberg H., Kaback H. R. Electrochemical proton gradient in inverted membrane vesicles from Escherichia coli. Biochemistry. 1980 Jan 8;19(1):1–9. doi: 10.1021/bi00542a001. [DOI] [PubMed] [Google Scholar]
- Rogers H. J. Iron-Binding Catechols and Virulence in Escherichia coli. Infect Immun. 1973 Mar;7(3):445–456. doi: 10.1128/iai.7.3.445-456.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schnaitman C. A. Solubilization of the cytoplasmic membrane of Escherichia coli by Triton X-100. J Bacteriol. 1971 Oct;108(1):545–552. doi: 10.1128/jb.108.1.545-552.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sigel S. P., Payne S. M. Effect of iron limitation on growth, siderophore production, and expression of outer membrane proteins of Vibrio cholerae. J Bacteriol. 1982 Apr;150(1):148–155. doi: 10.1128/jb.150.1.148-155.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tait G. H. The identification and biosynthesis of siderochromes formed by Micrococcus denitrificans. Biochem J. 1975 Jan;146(1):191–204. doi: 10.1042/bj1460191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weinberg E. D. Iron withholding: a defense against infection and neoplasia. Physiol Rev. 1984 Jan;64(1):65–102. doi: 10.1152/physrev.1984.64.1.65. [DOI] [PubMed] [Google Scholar]
- Williams P., Brown M. R., Lambert P. A. Effect of iron deprivation on the production of siderophores and outer membrane proteins in Klebsiella aerogenes. J Gen Microbiol. 1984 Sep;130(9):2357–2365. doi: 10.1099/00221287-130-9-2357. [DOI] [PubMed] [Google Scholar]
- Zalman L. S., Nikaido H. Dimeric porin from Paracoccus denitrificans. J Bacteriol. 1985 Apr;162(1):430–433. doi: 10.1128/jb.162.1.430-433.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]