Abstract
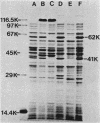
The glpTQ operon and the glpA and glpB genes are located adjacent to one another near min 49 of the linkage map of Escherichia coli K-12. The positions and directions of transcription of the glpA and glpB genes with respect to the glpTQ operon were determined in the present work. Strains harboring Mu d1(Ap lac) fusions in either glpA or glpB were converted to the respective lambda p1(209) lysogens. Induction of these lysogens with mitomycin C resulted in production of Lac+ phage progeny which carried adjacent chromosomal DNA. Genetic crosses with a collection of glpT mutant strains were performed with several such phage lines. A fine-structure deletion map of the glpT gene was thus constructed. All phages used for this mapping carried DNA starting with the promoter-proximal end of glpT. This indicated that the glpTQ operon and the glpA and glpB genes are transcribed divergently. Additional evidence supporting this conclusion was obtained by physical mapping of restriction endonuclease cleavage sites in plasmids carrying these genes and in plasmids carrying glpA-lacZ or glpB-lacZ fusions. A new designation (glpC) for the gene encoding the 41,000-Mr subunit of the anaerobic sn-glycerol-3-phosphate dehydrogenase was proposed to distinguish it from the glpA gene, which encodes the 62,000-Mr subunit of the dehydrogenase, and the glpB gene, which encodes a membrane anchor subunit of the dehydrogenase. These three genes were present in an operon transcribed in the order glpA glpC glpB in the clockwise direction on the linkage map of E. coli.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bachmann B. J. Linkage map of Escherichia coli K-12, edition 7. Microbiol Rev. 1983 Jun;47(2):180–230. doi: 10.1128/mr.47.2.180-230.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Casadaban M. J., Cohen S. N. Lactose genes fused to exogenous promoters in one step using a Mu-lac bacteriophage: in vivo probe for transcriptional control sequences. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4530–4533. doi: 10.1073/pnas.76.9.4530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casadaban M. J. Transposition and fusion of the lac genes to selected promoters in Escherichia coli using bacteriophage lambda and Mu. J Mol Biol. 1976 Jul 5;104(3):541–555. doi: 10.1016/0022-2836(76)90119-4. [DOI] [PubMed] [Google Scholar]
- Cecchini G., Ackrell B. A., Deshler J. O., Gunsalus R. P. Reconstitution of quinone reduction and characterization of Escherichia coli fumarate reductase activity. J Biol Chem. 1986 Feb 5;261(4):1808–1814. [PubMed] [Google Scholar]
- HAYASHI S., KOCH J. P., LIN E. C. ACTIVE TRANSPORT OF L-ALPHA-GLYCEROPHOSPHATE IN ESCHERICHIA COLI. J Biol Chem. 1964 Sep;239:3098–3105. [PubMed] [Google Scholar]
- Hengge R., Larson T. J., Boos W. sn-Glycerol-3-phosphate transport in Salmonella typhimurium. J Bacteriol. 1983 Jul;155(1):186–195. doi: 10.1128/jb.155.1.186-195.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iuchi S., Kuritzkes D. R., Lin E. C. Escherichia coli mutant with altered respiratory control of the frd operon. J Bacteriol. 1985 Mar;161(3):1023–1028. doi: 10.1128/jb.161.3.1023-1028.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kistler W. S., Lin E. C. Anaerobic L- -glycerophosphate dehydrogenase of Escherichia coli: its genetic locus and its physiological role. J Bacteriol. 1971 Dec;108(3):1224–1234. doi: 10.1128/jb.108.3.1224-1234.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Komeda Y. Fusions of flagellar operons to lactose genes on a mu lac bacteriophage. J Bacteriol. 1982 Apr;150(1):16–26. doi: 10.1128/jb.150.1.16-26.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuritzkes D. R., Zhang X. Y., Lin E. C. Use of phi(glp-lac) in studies of respiratory regulation of the Escherichia coli anaerobic sn-glycerol-3-phosphate dehydrogenase genes (glpAB). J Bacteriol. 1984 Feb;157(2):591–598. doi: 10.1128/jb.157.2.591-598.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larson T. J., Ludtke D. N., Bell R. M. sn-Glycerol-3-phosphate auxotrophy of plsB strains of Escherichia coli: evidence that a second mutation, plsX, is required. J Bacteriol. 1984 Nov;160(2):711–717. doi: 10.1128/jb.160.2.711-717.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larson T. J., Schumacher G., Boos W. Identification of the glpT-encoded sn-glycerol-3-phosphate permease of Escherichia coli, an oligomeric integral membrane protein. J Bacteriol. 1982 Dec;152(3):1008–1021. doi: 10.1128/jb.152.3.1008-1021.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lemire B. D., Robinson J. J., Weiner J. H. Identification of membrane anchor polypeptides of Escherichia coli fumarate reductase. J Bacteriol. 1982 Dec;152(3):1126–1131. doi: 10.1128/jb.152.3.1126-1131.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin E. C. Glycerol dissimilation and its regulation in bacteria. Annu Rev Microbiol. 1976;30:535–578. doi: 10.1146/annurev.mi.30.100176.002535. [DOI] [PubMed] [Google Scholar]
- Ludtke D., Larson T. J., Beck C., Boos W. Only one gene is required for the glpT-dependent transport of sn-glycerol-3-phosphate in Escherichia coli. Mol Gen Genet. 1982;186(4):540–547. doi: 10.1007/BF00337962. [DOI] [PubMed] [Google Scholar]
- Mazodier P., Cossart P., Giraud E., Gasser F. Completion of the nucleotide sequence of the central region of Tn5 confirms the presence of three resistance genes. Nucleic Acids Res. 1985 Jan 11;13(1):195–205. doi: 10.1093/nar/13.1.195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miki K., Silhavy T. J., Andrews K. J. Resolution of glpA and glpT loci into separate operons in Escherichia coli K-12 strains. J Bacteriol. 1979 Apr;138(1):268–269. doi: 10.1128/jb.138.1.268-269.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schryvers A., Weiner J. H. The anaerobic sn-glycerol-3-phosphate dehydrogenase of Escherichia coli. Purification and characterization. J Biol Chem. 1981 Oct 10;256(19):9959–9965. [PubMed] [Google Scholar]
- Schryvers A., Weiner J. H. The anaerobic sn-glycerol-3-phosphate dehydrogenase: cloning and expression of the glpA gene of Escherichia coli and identification of the glpA products. Can J Biochem. 1982 Mar;60(3):224–231. doi: 10.1139/o82-027. [DOI] [PubMed] [Google Scholar]
- Schweizer H., Boos W. Cloning of the ugp region containing the structural genes for the pho regulon-dependent sn-glycerol-3-phosphate transport system of Escherichia coli. Mol Gen Genet. 1983;192(1-2):177–186. doi: 10.1007/BF00327664. [DOI] [PubMed] [Google Scholar]
- Silhavy T. J., Brickman E., Bassford P. J., Jr, Casadaban M. J., Shuman H. A., Schwartz V., Guarente L., Schwartz M., Beckwith J. R. Structure of the malB region in Escherichia coli K12. II. Genetic map of the malE,F,G operon. Mol Gen Genet. 1979 Jul 24;174(3):249–259. doi: 10.1007/BF00267797. [DOI] [PubMed] [Google Scholar]
- Yamada M., Takeda Y., Okamoto K., Hirota Y. Physical map of the nrdA-nrdB-ftsB-glpT region of the chromosomal DNA of Escherichia coli. Gene. 1982 Jun;18(3):309–318. doi: 10.1016/0378-1119(82)90169-x. [DOI] [PubMed] [Google Scholar]
- Yanisch-Perron C., Vieira J., Messing J. Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mp18 and pUC19 vectors. Gene. 1985;33(1):103–119. doi: 10.1016/0378-1119(85)90120-9. [DOI] [PubMed] [Google Scholar]