Abstract
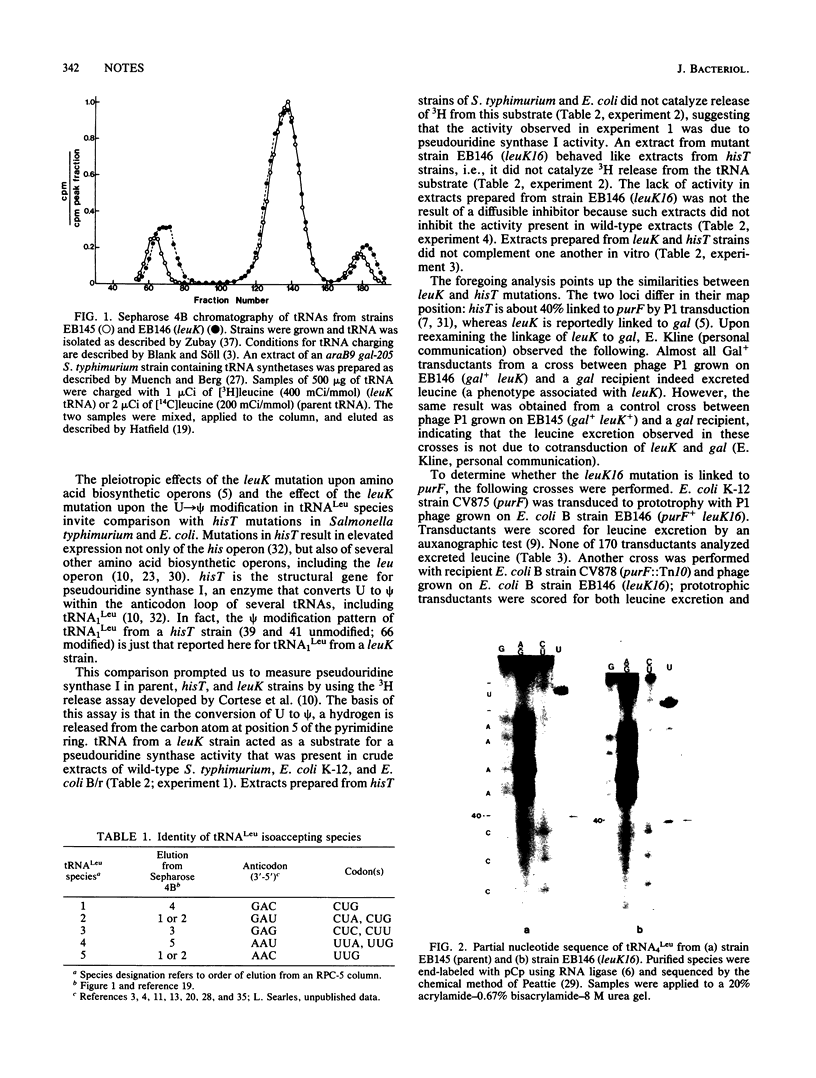
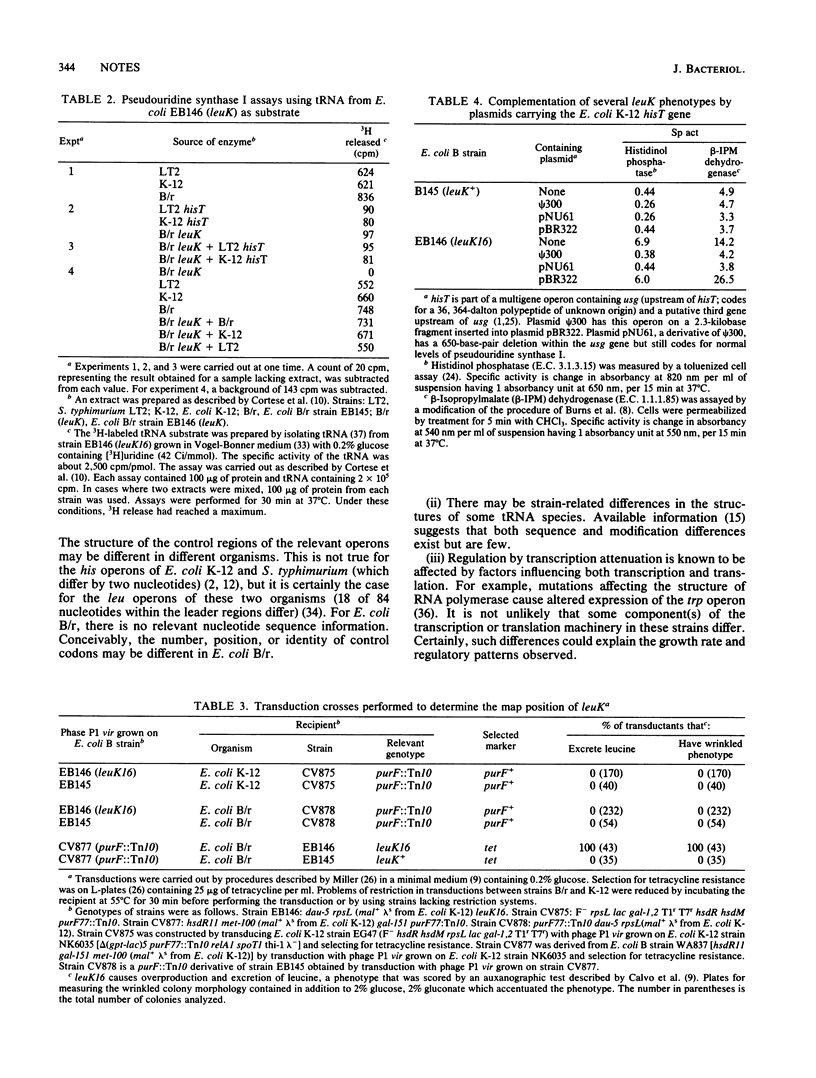
Escherichia coli B/r strain EB146 containing mutation leuK16 has elevated levels of enzymes involved in the synthesis of leucine, valine, isoleucine, histidine, and tryptophan (Brown et al., J. Bacteriol. 135:542-550, 1978). We show here that strain EB146 (leuK16) has properties that are similar to those of E. coli and Salmonella typhimurium hisT strains. In tRNA1Leu from both hisT and leuK strains, positions 39 and 41 are uridine residues rather than pseudouridine residues. Furthermore, in tRNA3Leu and tRNA4Leu from a leuK strain, uridine residues at positions 39 and 40, respectively, are unmodified. Pseudouridine synthase I activity is missing in extracts of strain EB146 (leuK16), and extracts of strain EB146 (leuK16) and of a hisT strain do not complement one another in vitro. Four phenotypes of strain EB146 (leuK16), leucine excretion, wrinkled colony morphology, and elevated levels of leu and his enzymes, are complemented by a plasmid having a 1.65-kilobase DNA fragment containing the E. coli K-12 hisT locus. These results indicate that either leuK codes for pseudouridine synthase I (and is thus a hisT locus in reality) or, less likely, it codes for a product that affects the synthesis or activity of pseudouridine synthase I.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arps P. J., Marvel C. C., Rubin B. C., Tolan D. A., Penhoet E. E., Winkler M. E. Structural features of the hisT operon of Escherichia coli K-12. Nucleic Acids Res. 1985 Jul 25;13(14):5297–5315. doi: 10.1093/nar/13.14.5297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnes W. M. DNA sequence from the histidine operon control region: seven histidine codons in a row. Proc Natl Acad Sci U S A. 1978 Sep;75(9):4281–4285. doi: 10.1073/pnas.75.9.4281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blank H. U., Söll D. Purification of five leucine transfer ribonucleic acid species from Escherichia coli and their acylation by heterologous leucyl-transfer ribonucleic acid synthetase. J Biol Chem. 1971 Aug 25;246(16):4947–4950. [PubMed] [Google Scholar]
- Blank H. U., Söll D. The nucleotide sequence of two leucine tRNA species from Escherichia coli K12. Biochem Biophys Res Commun. 1971 Jun 4;43(5):1192–1197. doi: 10.1016/0006-291x(71)90589-4. [DOI] [PubMed] [Google Scholar]
- Brown C. S., West R., Hilderman R. H., Bayliss F. T., Klines E. L. A new locus (leuK) affecting the regulation of branched-chain amino acid, histidine, and tryptophan biosynthetic enzymes. J Bacteriol. 1978 Aug;135(2):542–550. doi: 10.1128/jb.135.2.542-550.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bruce A. G., Uhlenbeck O. C. Reactions at the termini of tRNA with T4 RNA ligase. Nucleic Acids Res. 1978 Oct;5(10):3665–3677. doi: 10.1093/nar/5.10.3665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bruni C. B., Colantuoni V., Sbordone L., Cortese R., Blasi F. Biochemical and regulatory properties of Escherichia coli K-12 hisT mutants. J Bacteriol. 1977 Apr;130(1):4–10. doi: 10.1128/jb.130.1.4-10.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burns R. O., Calvo J., Margolin P., Umbarger H. E. Expression of the leucine operon. J Bacteriol. 1966 Apr;91(4):1570–1576. doi: 10.1128/jb.91.4.1570-1576.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calvo J. M., Freundlich M., Umbarger H. E. Regulation of branched-chain amino acid biosynthesis in Salmonella typhimurium: isolation of regulatory mutants. J Bacteriol. 1969 Mar;97(3):1272–1282. doi: 10.1128/jb.97.3.1272-1282.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cortese R., Landsberg R., Haar R. A., Umbarger H. E., Ames B. N. Pleiotropy of hisT mutants blocked in pseudouridine synthesis in tRNA: leucine and isoleucine-valine operons. Proc Natl Acad Sci U S A. 1974 May;71(5):1857–1861. doi: 10.1073/pnas.71.5.1857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di Nocera P. P., Blasi F., Di Lauro R., Frunzio R., Bruni C. B. Nucleotide sequence of the attenuator region of the histidine operon of Escherichia coli K-12. Proc Natl Acad Sci U S A. 1978 Sep;75(9):4276–4280. doi: 10.1073/pnas.75.9.4276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dube S. K., Marcker K. A., Yudelevich A. The nucleotide sequence of a leucine transfer RNA from E. coli. FEBS Lett. 1970 Sep 6;9(3):168–170. doi: 10.1016/0014-5793(70)80345-3. [DOI] [PubMed] [Google Scholar]
- Garel J. P., Garber R. L., Siddiqui M. A. Transfer RNA in posterior silk gland of Bombyx mori: polyacrylamide gel mapping of mature transfer RNA, identification and partial structural characterization of major isoacceptor species. Biochemistry. 1977 Aug 9;16(16):3618–3624. doi: 10.1021/bi00635a018. [DOI] [PubMed] [Google Scholar]
- Gauss D. H., Sprinzl M. Compilation of tRNA sequences. Nucleic Acids Res. 1983 Jan 11;11(1):r1–53. [PMC free article] [PubMed] [Google Scholar]
- Gillam I., Blew D., Warrington R. C., von Tigerstrom M., Tener G. M. A general procedure for the isolation of specific transfer ribonucleic acids. Biochemistry. 1968 Oct;7(10):3459–3468. doi: 10.1021/bi00850a022. [DOI] [PubMed] [Google Scholar]
- Gupta R. C., Randerath K. Rapid print-readout technique for sequencing of RNA's containing modified nucleotides. Nucleic Acids Res. 1979 Aug 10;6(11):3443–3458. doi: 10.1093/nar/6.11.3443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gupta R. C., Roe B. A., Randerath K. Sequence of human glycine transfer ribonucleic acid (anticodon CCC). Determination by a newly developed thin-layer readout sequencing technique and comparison with other glycine transfer ribonucleic acids. Biochemistry. 1980 Apr 15;19(8):1699–1705. doi: 10.1021/bi00549a028. [DOI] [PubMed] [Google Scholar]
- Hatfield G. W. Sepharose chromatography of transfer ribonucleic acids using reverse salt gradients. Methods Enzymol. 1979;59:215–218. doi: 10.1016/0076-6879(79)59084-3. [DOI] [PubMed] [Google Scholar]
- Kan J., Nirenberg M. W., Sueooka N. Coding specificity of Escherichia coli leucine transfer ribonucleic acids and effect of bacteriophage T2 infection. J Mol Biol. 1970 Sep 14;52(2):179–193. doi: 10.1016/0022-2836(70)90024-0. [DOI] [PubMed] [Google Scholar]
- Kolter R., Yanofsky C. Attenuation in amino acid biosynthetic operons. Annu Rev Genet. 1982;16:113–134. doi: 10.1146/annurev.ge.16.120182.000553. [DOI] [PubMed] [Google Scholar]
- Lawther R. P., Hatfield W. Biochemical characterization of an Escherichia coli hisT strain. J Bacteriol. 1977 Apr;130(1):552–557. doi: 10.1128/jb.130.1.552-557.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lewis J. A., Ames B. N. Histidine regulation in Salmonella typhimurium. XI. The percentage of transfer RNA His charged in vivo and its relation to the repression of the histidine operon. J Mol Biol. 1972 Apr 28;66(1):131–142. doi: 10.1016/s0022-2836(72)80011-1. [DOI] [PubMed] [Google Scholar]
- Marvel C. C., Arps P. J., Rubin B. C., Kammen H. O., Penhoet E. E., Winkler M. E. hisT is part of a multigene operon in Escherichia coli K-12. J Bacteriol. 1985 Jan;161(1):60–71. doi: 10.1128/jb.161.1.60-71.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakajima N., Ozeki H., Shimura Y. Organization and structure of an E. coli tRNA operon containing seven tRNA genes. Cell. 1981 Jan;23(1):239–249. doi: 10.1016/0092-8674(81)90288-9. [DOI] [PubMed] [Google Scholar]
- Peattie D. A. Direct chemical method for sequencing RNA. Proc Natl Acad Sci U S A. 1979 Apr;76(4):1760–1764. doi: 10.1073/pnas.76.4.1760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rizzino A. A., Bresalier R. S., Freundlich M. Derepressed levels of the isoleucine-valine and leucine enzymes in his T 1504, a strain of Salmonella typhimurium with altered leucine transfer ribonucleic acid. J Bacteriol. 1974 Feb;117(2):449–455. doi: 10.1128/jb.117.2.449-455.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roth J. R., Antón D. N., Hartman P. E. Histidine regulatory mutants in Salmonella typhimurium. I. Isolation and general properties. J Mol Biol. 1966 Dec 28;22(2):305–323. doi: 10.1016/0022-2836(66)90134-3. [DOI] [PubMed] [Google Scholar]
- Singer C. E., Smith G. R., Cortese R., Ames B. N. [Mutant tRNA His ineffective in repression and lacking two pseudouridine modifications]. Nat New Biol. 1972 Jul 19;238(81):72–74. doi: 10.1038/newbio238072a0. [DOI] [PubMed] [Google Scholar]
- VOGEL H. J., BONNER D. M. Acetylornithinase of Escherichia coli: partial purification and some properties. J Biol Chem. 1956 Jan;218(1):97–106. [PubMed] [Google Scholar]
- Wessler S. R., Calvo J. M. Control of leu operon expression in Escherichia coli by a transcription attenuation mechanism. J Mol Biol. 1981 Jul 15;149(4):579–597. doi: 10.1016/0022-2836(81)90348-x. [DOI] [PubMed] [Google Scholar]
- Yamaizumi Z., Kuchino Y., Harada F., Nishimura S., McCloskey J. A. Primary structure of Escherichia coli tRNA UUR Leu. Presence of an unknown adenosine derivative in the first position of the anticodon which recognizes the UU codon series. J Biol Chem. 1980 Mar 10;255(5):2220–2225. [PubMed] [Google Scholar]
- Yanofsky C., Horn V. Rifampin resistance mutations that alter the efficiency of transcription termination at the tryptophan operon attenuator. J Bacteriol. 1981 Mar;145(3):1334–1341. doi: 10.1128/jb.145.3.1334-1341.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]