Abstract
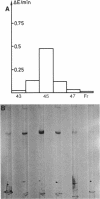
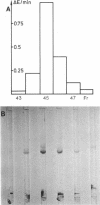
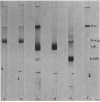
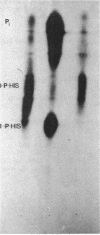
Recently we reported the phosphoenolpyruvate (PEP)-dependent phosphorylation of a 55-kilodalton protein of Streptococcus faecalis catalyzed by enzyme I and histidine-containing protein (HPr) of the phosphotransferase system (J. Deutscher, FEMS Microbiol. Lett. 29:237-243, 1985). The purified 55-kilodalton protein was found to exhibit dihydroxyacetone kinase activity. Glycerol was six times more slowly phosphorylated than dihydroxyacetone. The Kms were found to be 0.7 mM for ATP, 0.45 mM for dihydroxyacetone, and 0.9 mM for glycerol. PEP-dependent phosphorylation of dihydroxyacetone kinase stimulated phosphorylation of both substrates about 10-fold. Fructose 1,6-diphosphate at concentrations higher than 2 mM inhibited the activity of phosphorylated and unphosphorylated dihydroxyacetone kinase in a noncompetitive manner. The rate of PEP-dependent phosphorylation of dihydroxyacetone kinase was about 200-fold slower than the phosphorylation rate of III proteins (also called enzyme III or factor III), which so far have been considered the only phosphoryl acceptors of histidyl-phosphorylated HPr. P-Dihydroxyacetone kinase was found to be able to transfer its phosphoryl group in a backward reaction to HPr. Following [32P]PEP-dependent phosphorylation and tryptic digestion of dihydroxyacetone kinase, we isolated a labeled peptide composed of 37 amino acids, as determined by amino acid analysis. The single histidyl residue of this peptide most likely carries the phosphoryl group in phosphorylated dihydroxyacetone kinase.
Full text
PDF


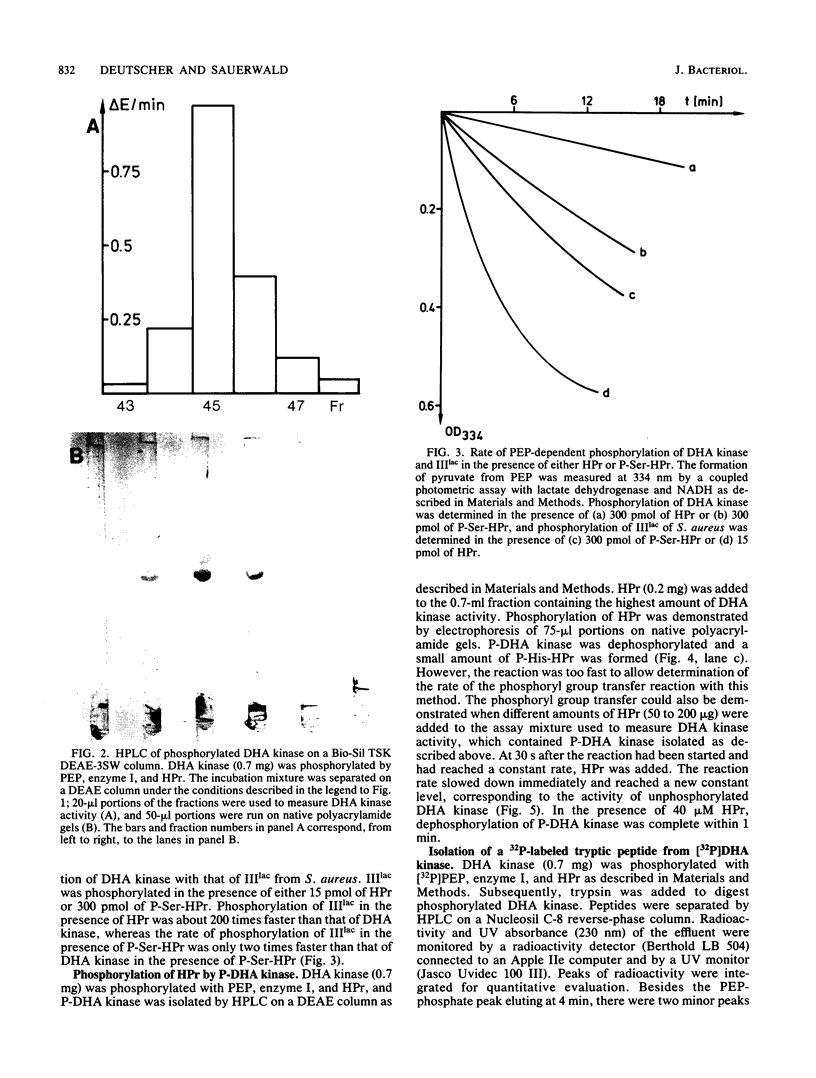
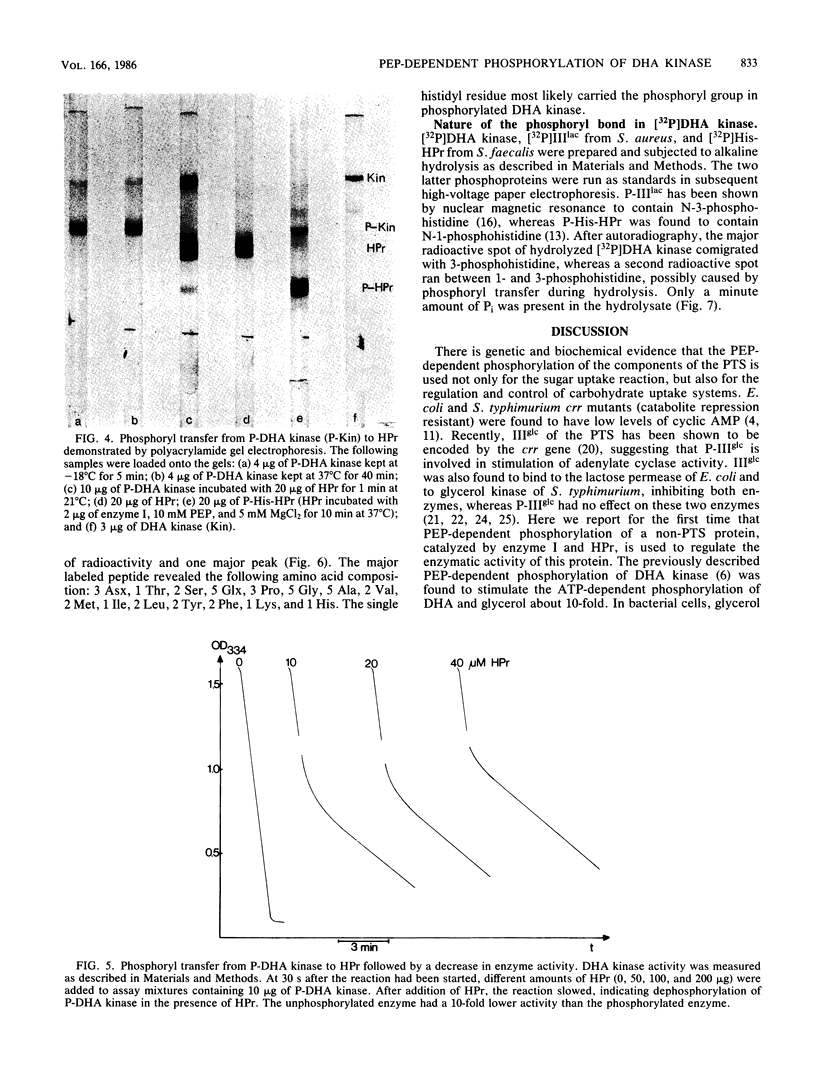



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alpert C. A., Frank R., Stüber K., Deutscher J., Hengstenberg W. Phosphoenolpyruvate-dependent protein kinase enzyme I of Streptococcus faecalis: purification and properties of the enzyme and characterization of its active center. Biochemistry. 1985 Feb 12;24(4):959–964. doi: 10.1021/bi00325a023. [DOI] [PubMed] [Google Scholar]
- Beyreuther K., Raufuss H., Schrecker O., Hengstenberg W. The phosphoenolpyruvate-dependent phosphotransferase system of Staphylococcus aureus. 1. Amino-acid sequence of the phosphocarrier protein HPr. Eur J Biochem. 1977 May 2;75(1):275–286. doi: 10.1111/j.1432-1033.1977.tb11527.x. [DOI] [PubMed] [Google Scholar]
- Castro L., Feucht B. U., Morse M. L., Saier M. H., Jr Regulation of carbohydrate permeases and adenylate cyclase in Escherichia coli. Studies with mutant strains in which enzyme I of the phosphoenolpyruvate:sugar phosphotransferase system is thermolabile. J Biol Chem. 1976 Sep 25;251(18):5522–5527. [PubMed] [Google Scholar]
- Cozzarelli N. R., Freedberg W. B., Lin E. C. Genetic control of L-alpha-glycerophosphate system in Escherichia coli. J Mol Biol. 1968 Feb 14;31(3):371–387. doi: 10.1016/0022-2836(68)90415-4. [DOI] [PubMed] [Google Scholar]
- Deutscher J., Beyreuther K., Sobek H. M., Stüber K., Hengstenberg W. Phosphoenolpyruvate-dependent phosphotransferase system of Staphylococcus aureus: factor IIIlac, a trimeric phospho-carrier protein that also acts as a phase transfer catalyst. Biochemistry. 1982 Sep 28;21(20):4867–4873. doi: 10.1021/bi00263a006. [DOI] [PubMed] [Google Scholar]
- Deutscher J., Kessler U., Hengstenberg W. Streptococcal phosphoenolpyruvate: sugar phosphotransferase system: purification and characterization of a phosphoprotein phosphatase which hydrolyzes the phosphoryl bond in seryl-phosphorylated histidine-containing protein. J Bacteriol. 1985 Sep;163(3):1203–1209. doi: 10.1128/jb.163.3.1203-1209.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dörschug M., Frank R., Kalbitzer H. R., Hengstenberg W., Deutscher J. Phosphoenolpyruvate-dependent phosphorylation site in enzyme IIIglc of the Escherichia coli phosphotransferase system. Eur J Biochem. 1984 Oct 1;144(1):113–119. doi: 10.1111/j.1432-1033.1984.tb08438.x. [DOI] [PubMed] [Google Scholar]
- Feucht B. U., Saier M. H., Jr Fine control of adenylate cyclase by the phosphoenolpyruvate:sugar phosphotransferase systems in Escherichia coli and Salmonella typhimurium. J Bacteriol. 1980 Feb;141(2):603–610. doi: 10.1128/jb.141.2.603-610.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forage R. G., Lin E. C. DHA system mediating aerobic and anaerobic dissimilation of glycerol in Klebsiella pneumoniae NCIB 418. J Bacteriol. 1982 Aug;151(2):591–599. doi: 10.1128/jb.151.2.591-599.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gassner M., Stehlik D., Schrecker O., Hengstenberg W., Maurer W., Rüterjans H. The phosphoenolpyruvate-dependent phosphotransferase system of Staphylococcus aureus. 2. 1H and 31P-nuclear-magnetic-resonance studies on the phosphocarrier protein HPr, phosphohistidines and phosphorylated HPr. Eur J Biochem. 1977 May 2;75(1):287–296. doi: 10.1111/j.1432-1033.1977.tb11528.x. [DOI] [PubMed] [Google Scholar]
- Hengstenberg W. Enzymology of carbohydrate transport in bacteria. Curr Top Microbiol Immunol. 1977;77:97–126. doi: 10.1007/978-3-642-66740-4_4. [DOI] [PubMed] [Google Scholar]
- Jin R. Z., Lin E. C. An inducible phosphoenolpyruvate: dihydroxyacetone phosphotransferase system in Escherichia coli. J Gen Microbiol. 1984 Jan;130(1):83–88. doi: 10.1099/00221287-130-1-83. [DOI] [PubMed] [Google Scholar]
- Kalbitzer H. R., Deutscher J., Hengstenberg W., Rösch P. Phosphoenolpyruvate-dependent phosphotransferase system of Staphylococcus aureus: 1H nuclear magnetic resonance studies on phosphorylated and unphosphorylated factor IIIlac and its interaction with the phosphocarrier protein HPr. Biochemistry. 1981 Oct 13;20(21):6178–6185. doi: 10.1021/bi00524a041. [DOI] [PubMed] [Google Scholar]
- Kalbitzer H. R., Hengstenberg W., Rösch P., Muss P., Bernsmann P., Engelmann R., Dörschug M., Deutscher J. HPr proteins of different microorganisms studied by hydrogen-1 high-resolution nuclear magnetic resonance: similarities of structures and mechanisms. Biochemistry. 1982 Jun 8;21(12):2879–2885. doi: 10.1021/bi00541a012. [DOI] [PubMed] [Google Scholar]
- Lin E. C. Glycerol dissimilation and its regulation in bacteria. Annu Rev Microbiol. 1976;30:535–578. doi: 10.1146/annurev.mi.30.100176.002535. [DOI] [PubMed] [Google Scholar]
- Nelson S. O., Schuitema A. R., Benne R., van der Ploeg L. H., Plijter J. S., Aan F., Postma P. W. Molecular cloning, sequencing, and expression of the crr gene: the structural gene for IIIGlc of the bacterial PEP:glucose phosphotransferase system. EMBO J. 1984 Jul;3(7):1587–1593. doi: 10.1002/j.1460-2075.1984.tb02015.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson S. O., Wright J. K., Postma P. W. The mechanism of inducer exclusion. Direct interaction between purified III of the phosphoenolpyruvate:sugar phosphotransferase system and the lactose carrier of Escherichia coli. EMBO J. 1983;2(5):715–720. doi: 10.1002/j.1460-2075.1983.tb01490.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Novotny M. J., Frederickson W. L., Waygood E. B., Saier M. H., Jr Allosteric regulation of glycerol kinase by enzyme IIIglc of the phosphotransferase system in Escherichia coli and Salmonella typhimurium. J Bacteriol. 1985 May;162(2):810–816. doi: 10.1128/jb.162.2.810-816.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O Y. K., Freese E. B., Freese E. Abnormal septation and inhibition of sporulation by accumulation of L- -glycerophosphate in Bacillus subtilis mutants. J Bacteriol. 1973 Feb;113(2):1034–1045. doi: 10.1128/jb.113.2.1034-1045.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Osumi T., Saier M. H., Jr Regulation of lactose permease activity by the phosphoenolpyruvate:sugar phosphotransferase system: evidence for direct binding of the glucose-specific enzyme III to the lactose permease. Proc Natl Acad Sci U S A. 1982 Mar;79(5):1457–1461. doi: 10.1073/pnas.79.5.1457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Postma P. W., Epstein W., Schuitema A. R., Nelson S. O. Interaction between IIIGlc of the phosphoenolpyruvate:sugar phosphotransferase system and glycerol kinase of Salmonella typhimurium. J Bacteriol. 1984 Apr;158(1):351–353. doi: 10.1128/jb.158.1.351-353.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reizer J., Novotny M. J., Hengstenberg W., Saier M. H., Jr Properties of ATP-dependent protein kinase from Streptococcus pyogenes that phosphorylates a seryl residue in HPr, a phosphocarrier protein of the phosphotransferase system. J Bacteriol. 1984 Oct;160(1):333–340. doi: 10.1128/jb.160.1.333-340.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reizer J., Novotny M. J., Stuiver I., Saier M. H., Jr Regulation of glycerol uptake by the phosphoenolpyruvate-sugar phosphotransferase system in Bacillus subtilis. J Bacteriol. 1984 Jul;159(1):243–250. doi: 10.1128/jb.159.1.243-250.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robillard G. T. The enzymology of the bacterial phosphoenolpyruvate-dependent sugar transport systems. Mol Cell Biochem. 1982 Jul 7;46(1):3–24. doi: 10.1007/BF00215577. [DOI] [PubMed] [Google Scholar]
- Ruch F. E., Lengeler J., Lin E. C. Regulation of glycerol catabolism in Klebsiella aerogenes. J Bacteriol. 1974 Jul;119(1):50–56. doi: 10.1128/jb.119.1.50-56.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson J., Torchia D. A. Use of 31P nuclear magnetic resonance spectroscopy and 14C fluorography in studies of glycolysis and regulation of pyruvate kinase in Streptococcus lactis. J Bacteriol. 1984 Jun;158(3):791–800. doi: 10.1128/jb.158.3.791-800.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waygood E. B., Mattoo R. L. A novel phosphoprotein dependent on the bacterial phosphoenolpyruvate-sugar phosphotransferase system. Can J Biochem Cell Biol. 1983 Feb-Mar;61(2-3):150–153. doi: 10.1139/o83-022. [DOI] [PubMed] [Google Scholar]