Abstract
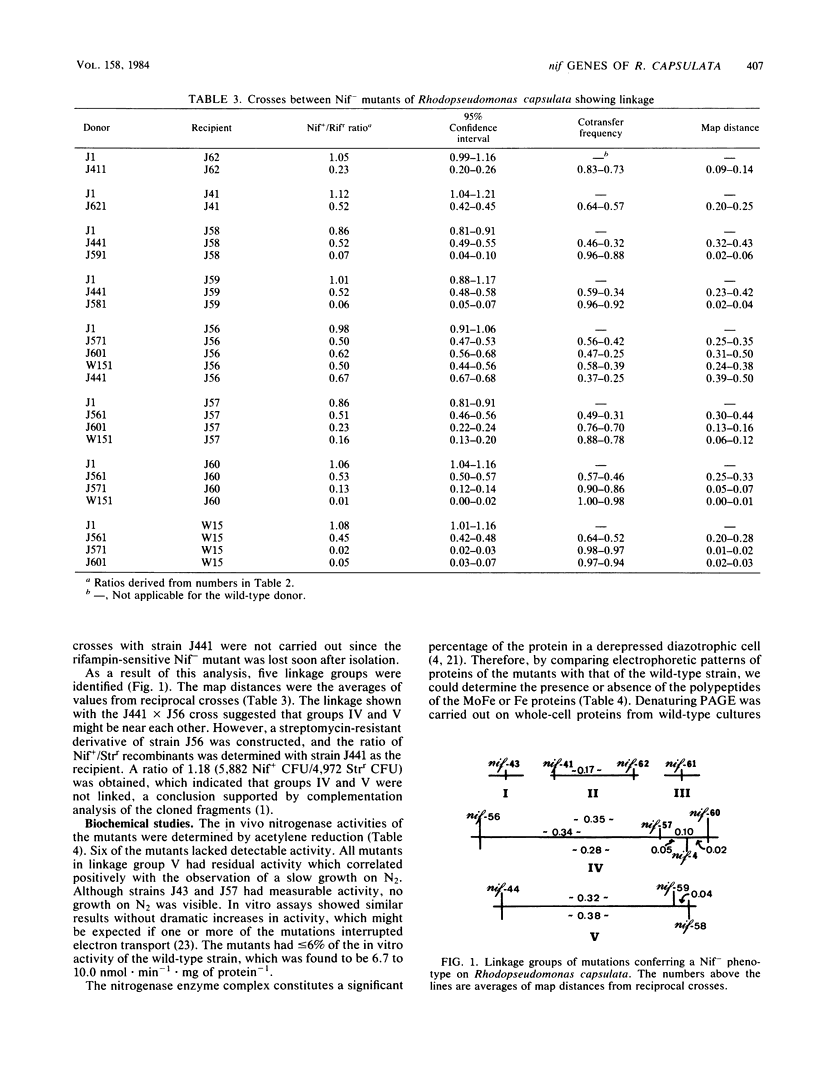
The endogenous gene transfer system of Rhodopseudomonas capsulata was used to analyze mutations which block the ability to use molecular nitrogen as the sole nitrogen source (nif). With this fine-structure mapping tool, linkage of nif mutations could be reliably established if separated by 2,700 base pairs or less. Eleven independent mutations were analyzed, and five linkage groups were found. The overall chromosomal arrangement of these groups awaits conjugational or physical analysis. A candidate for the inactive subunit of R. capsulata Fe protein was located in gels at a position of about 38,000 molecular weight, 5,000 more than that of the presumed active subunit.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Avtges P., Scolnik P. A., Haselkorn R. Genetic and physical map of the structural genes (nifH,D,K) coding for the nitrogenase complex of Rhodopseudomonas capsulata. J Bacteriol. 1983 Oct;156(1):251–256. doi: 10.1128/jb.156.1.251-256.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Emerich D. W., Burris R. H. Interactions of heterologous nitrogenase components that generate catalytically inactive complexes. Proc Natl Acad Sci U S A. 1976 Dec;73(12):4369–4373. doi: 10.1073/pnas.73.12.4369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hallenbeck P. C., Meyer C. M., Vignais P. M. Nitrogenase from the photosynthetic bacterium Rhodopseudomonas capsulata: purification and molecular properties. J Bacteriol. 1982 Feb;149(2):708–717. doi: 10.1128/jb.149.2.708-717.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hallenbeck P. C., Meyer C. M., Vignais P. M. Regulation of nitrogenase in the photosynthetic bacterium Rhodopseudomonas capsulata as studied by two-dimensional gel electrophoresis. J Bacteriol. 1982 Sep;151(3):1612–1616. doi: 10.1128/jb.151.3.1612-1616.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Ludden P. W., Burris R. H. Purification and properties of nitrogenase from Rhodospirillum rubrum, and evidence for phosphate, ribose and an adenine-like unit covalently bound to the iron protein. Biochem J. 1978 Oct 1;175(1):251–259. doi: 10.1042/bj1750251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ludden P. W., Burris R. H. Removal of an adenine-like molecule during activation of dinitrogenase reductase from Rhodospirillum rubrum. Proc Natl Acad Sci U S A. 1979 Dec;76(12):6201–6205. doi: 10.1073/pnas.76.12.6201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ludden P. W., Preston G. G., Dowling T. E. Comparison of active and inactive forms of iron protein from Rhodospirillum rubrum. Biochem J. 1982 Jun 1;203(3):663–668. doi: 10.1042/bj2030663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marrs B. Genetic recombination in Rhodopseudomonas capsulata. Proc Natl Acad Sci U S A. 1974 Mar;71(3):971–973. doi: 10.1073/pnas.71.3.971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mazur B. J., Rice D., Haselkorn R. Identification of blue-green algal nitrogen fixation genes by using heterologous DNA hybridization probes. Proc Natl Acad Sci U S A. 1980 Jan;77(1):186–190. doi: 10.1073/pnas.77.1.186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neilson A. H., Nordlund S. Regulation of nitrogenase synthesis in intact cells of Rhodospirillum rubrum: inactivation of nitrogen fixation by ammonia, L-glutamine and L-asparagine. J Gen Microbiol. 1975 Nov;91(1):53–62. doi: 10.1099/00221287-91-1-53. [DOI] [PubMed] [Google Scholar]
- Randall D. D., Williams M., Rapp B. J. Phosphorylation-dephosphorylation of pyruvate dehydrogenase complex from pea leaf mitochondria. Arch Biochem Biophys. 1981 Apr 1;207(2):437–444. doi: 10.1016/0003-9861(81)90051-5. [DOI] [PubMed] [Google Scholar]
- Roberts G. P., Brill W. J. Genetics and regulation of nitrogen fixation. Annu Rev Microbiol. 1981;35:207–235. doi: 10.1146/annurev.mi.35.100181.001231. [DOI] [PubMed] [Google Scholar]
- Roberts G. P., MacNeil T., MacNeil D., Brill W. J. Regulation and characterization of protein products coded by the nif (nitrogen fixation) genes of Klebsiella pneumoniae. J Bacteriol. 1978 Oct;136(1):267–279. doi: 10.1128/jb.136.1.267-279.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruvkun G. B., Ausubel F. M. Interspecies homology of nitrogenase genes. Proc Natl Acad Sci U S A. 1980 Jan;77(1):191–195. doi: 10.1073/pnas.77.1.191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- St John R. T., Johnston H. M., Seidman C., Garfinkel D., Gordon J. K., Shah V. K., Brill W. J. Biochemistry and genetics of Klebsiella pneumoniae mutant strains unable to fix N2. J Bacteriol. 1975 Mar;121(3):759–765. doi: 10.1128/jb.121.3.759-765.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sweet W. J., Burris R. H. Inhibition of nitrogenase activity by NH+4 in Rhodospirillum rubrum. J Bacteriol. 1981 Feb;145(2):824–831. doi: 10.1128/jb.145.2.824-831.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor D. P., Cohen S. N., Clark W. G., Marrs B. L. Alignment of genetic and restriction maps of the photosynthesis region of the Rhodopseudomonas capsulata chromosome by a conjugation-mediated marker rescue technique. J Bacteriol. 1983 May;154(2):580–590. doi: 10.1128/jb.154.2.580-590.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wall J. D., Gest H. Derepression of nitrogenase activity in glutamine auxotrophs of Rhodopseudomonas capsulata. J Bacteriol. 1979 Mar;137(3):1459–1463. doi: 10.1128/jb.137.3.1459-1463.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wall J. D., Johansson B. C., Gest H. A pleiotropic mutant of Rhodopseudomonas capsulata defective in nitrogen metabolism. Arch Microbiol. 1977 Dec 15;115(3):259–263. doi: 10.1007/BF00446450. [DOI] [PubMed] [Google Scholar]
- Wall J. D., Weaver P. F., Gest H. Genetic transfer of nitrogenase-hydrogenase activity in Rhodopseudomonas capsulata. Nature. 1975 Dec 18;258(5536):630–631. doi: 10.1038/258630a0. [DOI] [PubMed] [Google Scholar]
- Yen H. C., Hu N. T., Marrs B. L. Characterization of the gene transfer agent made by an overproducer mutant of Rhodopseudomonas capsulata. J Mol Biol. 1979 Jun 25;131(2):157–168. doi: 10.1016/0022-2836(79)90071-8. [DOI] [PubMed] [Google Scholar]
- Yen H. C., Marrs B. Map of genes for carotenoid and bacteriochlorophyll biosynthesis in Rhodopseudomonas capsulata. J Bacteriol. 1976 May;126(2):619–629. doi: 10.1128/jb.126.2.619-629.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]