Abstract
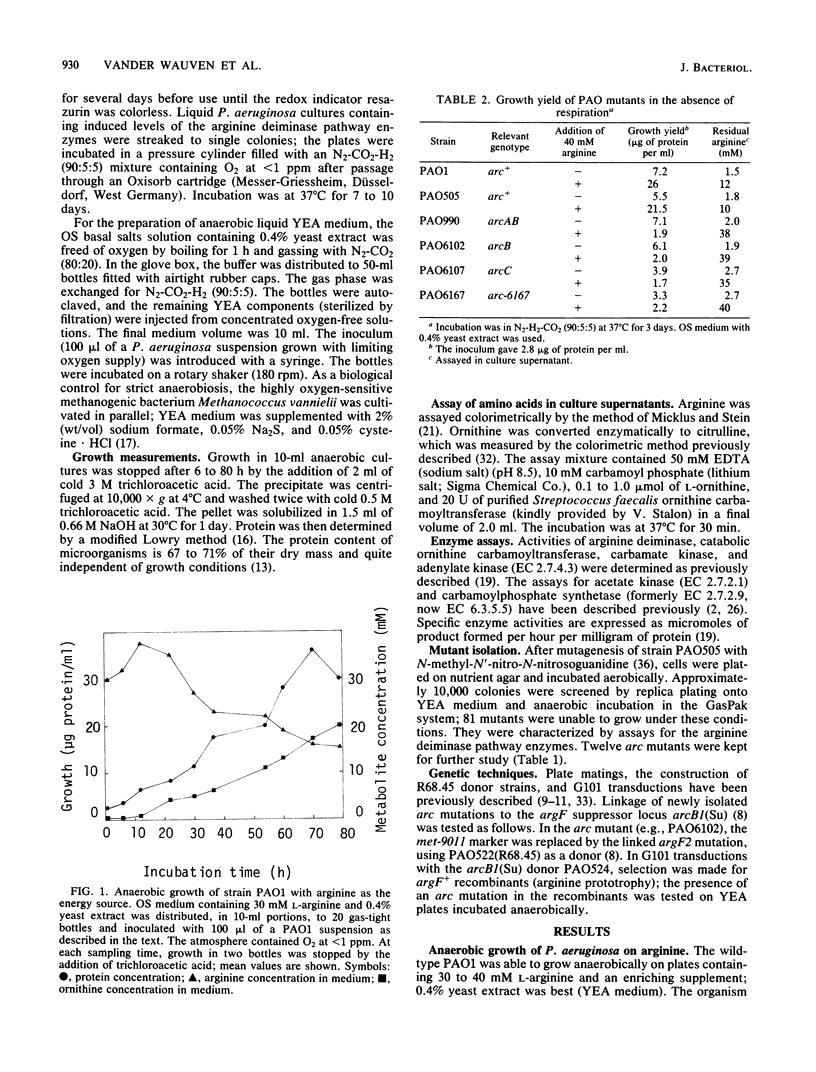
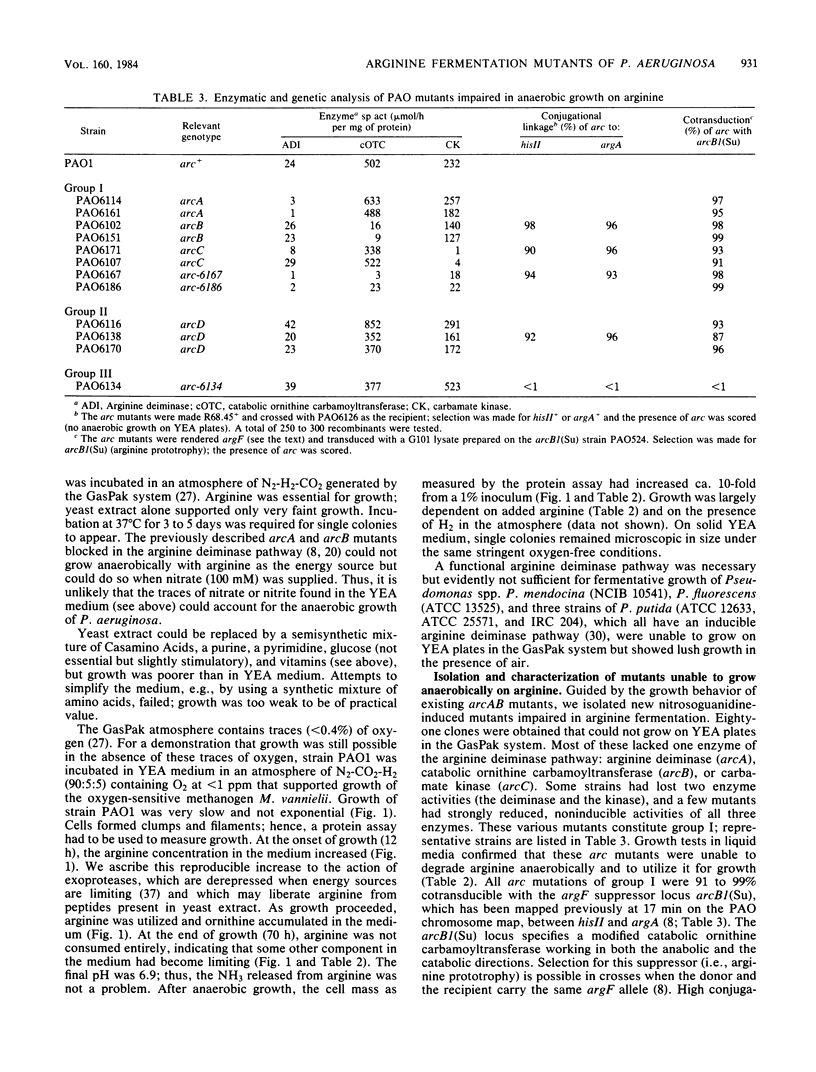
Pseudomonas aeruginosa PAO was able to grow in the absence of exogenous terminal electron acceptors, provided that the medium contained 30 to 40 mM L-arginine and 0.4% yeast extract. Under strictly anaerobic conditions (O2 at less than 1 ppm), growth could be measured as an increase in protein and proceeded in a non-exponential way; arginine was largely converted to ornithine but not entirely consumed at the end of growth. In the GasPak anaerobic jar (Becton Dickinson and Co.), the wild-type strain PAO1 grew on arginine-yeast extract medium in 3 to 5 days; mutants could be isolated that were unable to grow under these conditions. All mutants (except one) were defective in at least one of the three enzymes of the arginine deiminase pathway (arcA, arcB, and arcC mutants) or in a novel function that might be involved in anaerobic arginine uptake (arcD mutants). The mutations arcA (arginine deiminase), arcB (catabolic ornithine carbamoyltransferase), arcC (carbamate kinase), and arcD were highly cotransducible and mapped in the 17-min chromosome region. Some mutations in the arc cluster led to low, noninducible levels of all three arginine deiminase pathway enzymes and thus may affect control elements required for induction of the postulated arc operon. Two fluorescent pseudomonads (P. putida and P. fluorescens) and P. mendocina, as well as one PAO mutant, possessed an inducible arginine deiminase pathway and yet were unable to grow fermentatively on arginine. The ability to use arginine-derived ATP for growth may provide P. aeruginosa with a selective advantage when oxygen and nitrate are scarce.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abdelal A. T., Bibb W. F., Nainan O. Carbamate kinase from Pseudomonas aeruginosa: purification, characterization, physiological role, and regulation. J Bacteriol. 1982 Sep;151(3):1411–1419. doi: 10.1128/jb.151.3.1411-1419.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abdelal A. T., Bussey L., Vickers L. Carbamoylphosphate synthetase from Pseudomonas aeruginosa. Subunit composition, kinetic analysis and regulation. Eur J Biochem. 1983 Jan 1;129(3):697–702. [PubMed] [Google Scholar]
- Armitage J. P., Evans M. C. The motile and tactic behaviour of Pseudomonas aeruginosa in anaerobic environments. FEBS Lett. 1983 May 30;156(1):113–118. doi: 10.1016/0014-5793(83)80259-2. [DOI] [PubMed] [Google Scholar]
- Bachmann B. J. Linkage map of Escherichia coli K-12, edition 7. Microbiol Rev. 1983 Jun;47(2):180–230. doi: 10.1128/mr.47.2.180-230.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barker H. A. Amino acid degradation by anaerobic bacteria. Annu Rev Biochem. 1981;50:23–40. doi: 10.1146/annurev.bi.50.070181.000323. [DOI] [PubMed] [Google Scholar]
- Broman K., Lauwers N., Stalon V., Wiame J. M. Oxygen and nitrate in utilization by Bacillus licheniformis of the arginase and arginine deiminase routes of arginine catabolism and other factors affecting their syntheses. J Bacteriol. 1978 Sep;135(3):920–927. doi: 10.1128/jb.135.3.920-927.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fenske J. D., Kenny G. E. Role of arginine deiminase in growth of Mycoplasma hominis. J Bacteriol. 1976 Apr;126(1):501–510. doi: 10.1128/jb.126.1.501-510.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haas D., Holloway B. W. Chromosome mobilization by the R plasmid R68.45: a tool in Pseudomonas genetics. Mol Gen Genet. 1978 Jan 17;158(3):229–237. doi: 10.1007/BF00267194. [DOI] [PubMed] [Google Scholar]
- Haas D., Holloway B. W. R factor variants with enhanced sex factor activity in Pseudomonas aeruginosa. Mol Gen Genet. 1976 Mar 30;144(3):243–251. doi: 10.1007/BF00341722. [DOI] [PubMed] [Google Scholar]
- Haas D., Holloway B. W., Schamböck A., Leisinger T. The genetic organization of arginine biosynthesis in Pseudomonas aeruginosa. Mol Gen Genet. 1977 Jul 7;154(1):7–22. doi: 10.1007/BF00265571. [DOI] [PubMed] [Google Scholar]
- Haas D., Matsumoto H., Moretti P., Stalon V., Mercenier A. Arginine degradation in Pseudomonas aeruginosa mutants blocked in two arginine catabolic pathways. Mol Gen Genet. 1984;193(3):437–444. doi: 10.1007/BF00382081. [DOI] [PubMed] [Google Scholar]
- Harder W., Dijkhuizen L. Strategies of mixed substrate utilization in microorganisms. Philos Trans R Soc Lond B Biol Sci. 1982 Jun 11;297(1088):459–480. doi: 10.1098/rstb.1982.0055. [DOI] [PubMed] [Google Scholar]
- Hartmann R., Sickinger H. D., Oesterhelt D. Anaerobic growth of halobacteria. Proc Natl Acad Sci U S A. 1980 Jul;77(7):3821–3825. doi: 10.1073/pnas.77.7.3821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hass D., Evans R., Mercenier A., Simon J. P., Stalon V. Genetic and physiological characterization of Pseudomonas aeruginosa mutants affected in the catabolic ornithine carbamoyltransferase. J Bacteriol. 1979 Sep;139(3):713–720. doi: 10.1128/jb.139.3.713-720.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holloway B. W. Genetics of Pseudomonas. Bacteriol Rev. 1969 Sep;33(3):419–443. doi: 10.1128/br.33.3.419-443.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kennedy S. I., Fewson C. A. Enzymes of the mandelate pathway in Bacterium N.C.I.B. 8250. Biochem J. 1968 Apr;107(4):497–506. doi: 10.1042/bj1070497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mercenier A., Simon J. P., Haas D., Stalon V. Catabolism of L-arginine by Pseudomonas aeruginosa. J Gen Microbiol. 1980 Feb;116(2):381–389. doi: 10.1099/00221287-116-2-381. [DOI] [PubMed] [Google Scholar]
- Mercenier A., Simon J. P., Vander Wauven C., Haas D., Stalon V. Regulation of enzyme synthesis in the arginine deiminase pathway of Pseudomonas aeruginosa. J Bacteriol. 1980 Oct;144(1):159–163. doi: 10.1128/jb.144.1.159-163.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mercenier A., Stalon V., Simon J. P., Haas D. Mapping of the arginine deiminase gene in Pseudomonas aeruginosa. J Bacteriol. 1982 Feb;149(2):787–788. doi: 10.1128/jb.149.2.787-788.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Micklus M. J., Stein I. M. The colorimetric determination of mono- and disubstituted guanidines. Anal Biochem. 1973 Aug;54(2):545–553. doi: 10.1016/0003-2697(73)90386-2. [DOI] [PubMed] [Google Scholar]
- Ornston L. N., Stanier R. Y. The conversion of catechol and protocatechuate to beta-ketoadipate by Pseudomonas putida. J Biol Chem. 1966 Aug 25;241(16):3776–3786. [PubMed] [Google Scholar]
- ROSE I. A., GRUNBERG-MANAGO M., KOREY S. R., OCHOA S. Enzymatic phosphorylation of acetate. J Biol Chem. 1954 Dec;211(2):737–756. [PubMed] [Google Scholar]
- Rahman M., Laverack P. D., Clarke P. H. The catabolism of arginine by Pseudomonas aeruginosa. J Gen Microbiol. 1980 Feb;116(2):371–380. doi: 10.1099/00221287-116-2-371. [DOI] [PubMed] [Google Scholar]
- SHOESMITH J. H., SHERRIS J. C. Studies on the mechanism of arginine-activated motility in a Pseudomonas strain. J Gen Microbiol. 1960 Feb;22:10–24. doi: 10.1099/00221287-22-1-10. [DOI] [PubMed] [Google Scholar]
- Seip W. F., Evans G. L. Atmospheric analysis and redox potentials of culture media in the GasPak System. J Clin Microbiol. 1980 Mar;11(3):226–233. doi: 10.1128/jcm.11.3.226-233.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simon J. P., Wargnies B., Stalon V. Control of enzyme synthesis in the arginine deiminase pathway of Streptococcus faecalis. J Bacteriol. 1982 Jun;150(3):1085–1090. doi: 10.1128/jb.150.3.1085-1090.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stalon V., Mercenier A. L-arginine utilization by Pseudomonas species. J Gen Microbiol. 1984 Jan;130(1):69–76. doi: 10.1099/00221287-130-1-69. [DOI] [PubMed] [Google Scholar]
- Stalon V., Ramos F., Piérard A., Wiame J. M. Regulation of the catabolic ornithine carbamoyltransferase of Pseudomonas fluorescens. A comparison with the anabolic transferase and with a mutationally modified catabolic transferase. Eur J Biochem. 1972 Aug 18;29(1):25–35. doi: 10.1111/j.1432-1033.1972.tb01953.x. [DOI] [PubMed] [Google Scholar]
- Stalon V., Ramos F., Piérard A., Wiame J. M. The occurrence of a catabolic and an anabolic ornithine carbamoyltransferase in Pseudomonas. Biochim Biophys Acta. 1967 May 16;139(1):91–97. doi: 10.1016/0005-2744(67)90115-5. [DOI] [PubMed] [Google Scholar]
- Stanisich V. A., Holloway B. W. A mutant sex factor of Pseudomonas aeruginosa. Genet Res. 1972 Feb;19(1):91–108. doi: 10.1017/s0016672300014294. [DOI] [PubMed] [Google Scholar]
- Thauer R. K., Jungermann K., Decker K. Energy conservation in chemotrophic anaerobic bacteria. Bacteriol Rev. 1977 Mar;41(1):100–180. doi: 10.1128/br.41.1.100-180.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voellym R., Leisinger T. Role of 4-aminobutyrate aminotransferase in the arginine metabolism of Pseudomonas aeruginosa. J Bacteriol. 1976 Dec;128(3):722–729. doi: 10.1128/jb.128.3.722-729.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whooley M. A., O'Callaghan J. A., McLoughlin A. J. Effect of substrate on the regulation of exoprotease production by Pseudomonas aeruginosa ATCC 10145. J Gen Microbiol. 1983 Apr;129(4):981–988. doi: 10.1099/00221287-129-4-981. [DOI] [PubMed] [Google Scholar]



