Abstract
Mutant strains of Escherichia coli unable to synthesize cyclic adenosine 3',5'-monophosphate (cAMP) or the cyclic adenosine monophosphate receptor protein (CRP) were more resistant than wild-type cells to infection by bacteriophage T6. This resistance was found to be associated with the decreased production of specific T6 receptor protein (also the colicin K receptor) located in the outer membrane protein fraction of these cells. Transcription of this particular outer membrane protein was regulated by the cAMP-CRP complex. A novel affinity technique coupled with sodium dodecyl sulfate-polyacrylamide gel electrophoresis was used in these investigations.
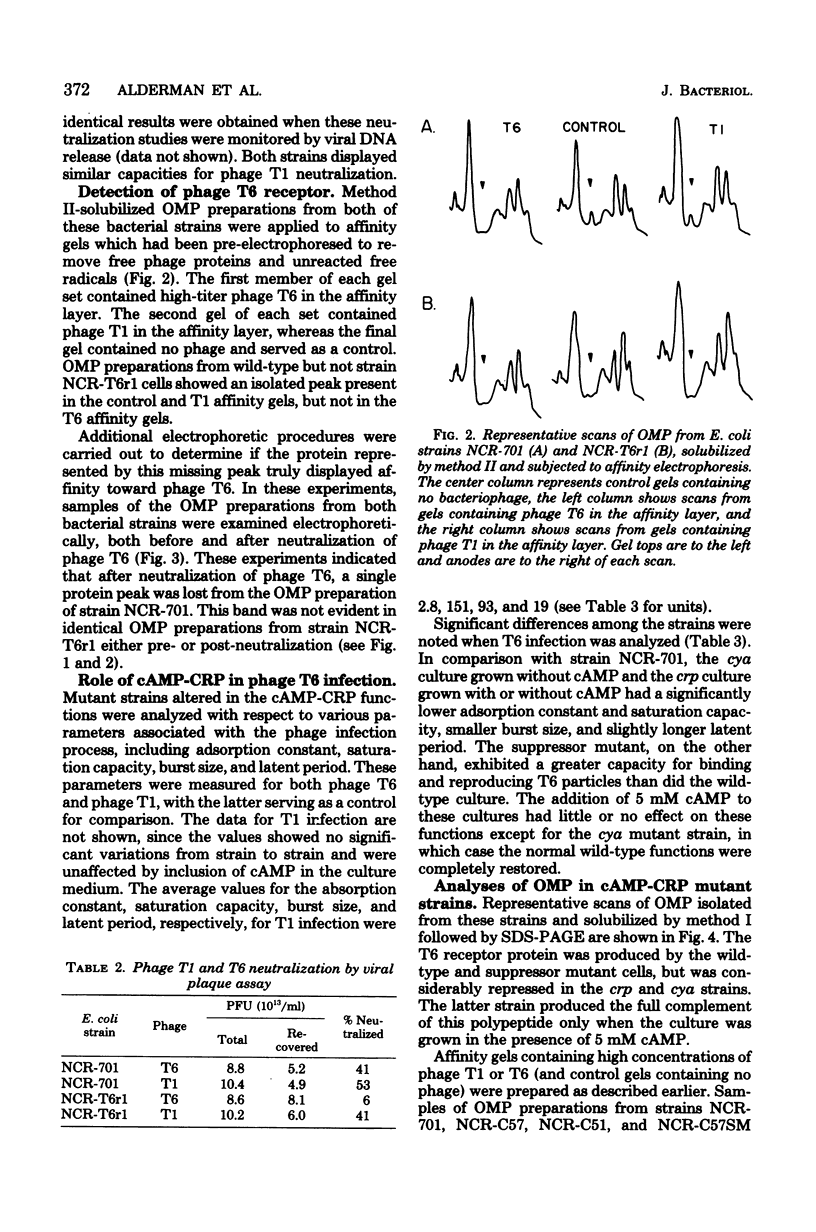
Full text
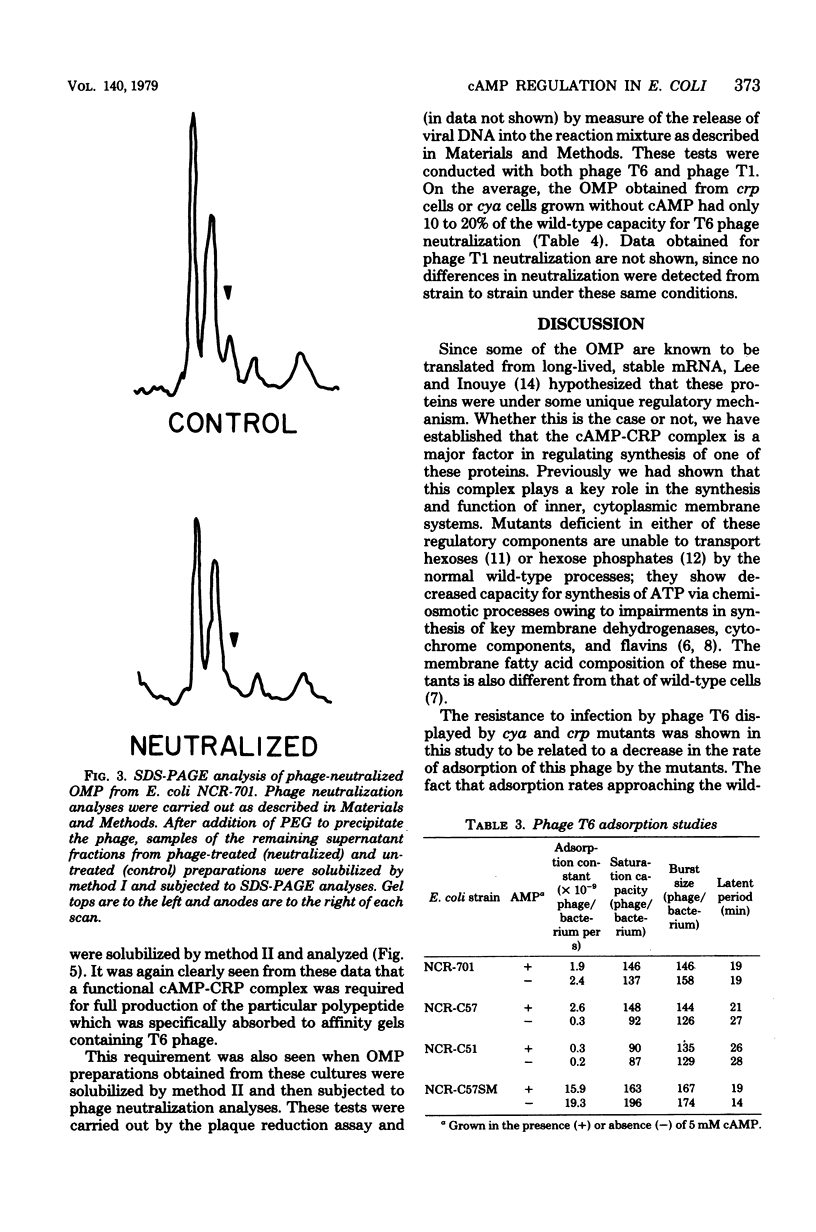
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bassford P. J., Jr, Diedrich D. L., Schnaitman C. L., Reeves P. Outer membrane proteins of Escherichia coli. VI. Protein alteration in bacteriophage-resistant mutants. J Bacteriol. 1977 Aug;131(2):608–622. doi: 10.1128/jb.131.2.608-622.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bertolini M. J., Tankersley D. L., Schroeder D. D. Staining and destaining polyacrylamide gels: a comparison of coomassie blue and fast green protein dyes. Anal Biochem. 1976 Mar;71(1):6–13. doi: 10.1016/0003-2697(76)90003-8. [DOI] [PubMed] [Google Scholar]
- Bragg P. D., Hou C. Organization of proteins in the native and reformed outer membrane of Escherichia coli. Biochim Biophys Acta. 1972 Aug 9;274(2):478–488. doi: 10.1016/0005-2736(72)90193-9. [DOI] [PubMed] [Google Scholar]
- Broman R. L., Dobrogosz W. J. Stimulation of cytochrome synthesis in Escherichia coli by cyclic AMP. Arch Biochem Biophys. 1974 Jun;162(2):595–601. doi: 10.1016/0003-9861(74)90220-3. [DOI] [PubMed] [Google Scholar]
- Dallas W. S., Tseng Y., Dobrogosz W. J. Regulation of membrane functions and fatty acid composition in Escherichia coli by cyclic AMP receptor protein. Arch Biochem Biophys. 1976 Jul;175(1):295–302. doi: 10.1016/0003-9861(76)90511-7. [DOI] [PubMed] [Google Scholar]
- Dills S. E., Dobrogosz W. J. Cyclic adenosine 3',5'-monophosphate regulation of membrane energetics in Escherichia coli. J Bacteriol. 1977 Sep;131(3):854–865. doi: 10.1128/jb.131.3.854-865.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ezzell J. W., Dobrogosz W. J. Altered hexose transport and salt sensitivity in cyclic adenosine 3',5'-monophosphate-deficient Escherichia coli. J Bacteriol. 1975 Nov;124(2):815–824. doi: 10.1128/jb.124.2.815-824.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ezzell J. W., Dobrogosz W. J. Cyclic AMP regulation of the hexose phosphate transport system in Escherichia coli. J Bacteriol. 1978 Feb;133(2):1047–1049. doi: 10.1128/jb.133.2.1047-1049.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar S. Properties of adenyl cyclase and cyclic adenosine 3',5'-monophosphate receptor protein-deficient mutants of Escherichia coli. J Bacteriol. 1976 Feb;125(2):545–555. doi: 10.1128/jb.125.2.545-555.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lee N., Inouye M. Outer membrane proteins of Escherichia coli: biosynthesis and assembly. FEBS Lett. 1974 Feb 15;39(2):167–170. doi: 10.1016/0014-5793(74)80043-8. [DOI] [PubMed] [Google Scholar]
- MAGASANIK B. Catabolite repression. Cold Spring Harb Symp Quant Biol. 1961;26:249–256. doi: 10.1101/sqb.1961.026.01.031. [DOI] [PubMed] [Google Scholar]
- Manning P. A., Reeves P. Outer membrane of Escherichia coli K-12: TSX mutants (resistant to bacteriophage T6 and colicin K) lack an outer membrane protein. Biochem Biophys Res Commun. 1976 Jul 26;71(2):466–471. doi: 10.1016/0006-291x(76)90810-x. [DOI] [PubMed] [Google Scholar]
- Pastan I., Adhya S. Cyclic adenosine 5'-monophosphate in Escherichia coli. Bacteriol Rev. 1976 Sep;40(3):527–551. doi: 10.1128/br.40.3.527-551.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schnaitman C. A. Effect of ethylenediaminetetraacetic acid, Triton X-100, and lysozyme on the morphology and chemical composition of isolate cell walls of Escherichia coli. J Bacteriol. 1971 Oct;108(1):553–563. doi: 10.1128/jb.108.1.553-563.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schnaitman C. A. Outer membrane proteins of Escherichia coli. 3. Evidence that the major protein of Escherichia coli O111 outer membrane consists of four distinct polypeptide species. J Bacteriol. 1974 May;118(2):442–453. doi: 10.1128/jb.118.2.442-453.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schnaitman C. A. Outer membrane proteins of Escherichia coli. I. Effect of preparative conditions on the migration of protein in polyacrylamide gels. Arch Biochem Biophys. 1973 Aug;157(2):541–552. doi: 10.1016/0003-9861(73)90673-5. [DOI] [PubMed] [Google Scholar]
- Schnaitman C. A. Outer membrane proteins of Escherichia coli. II. Heterogeneity of major outer membrane polypeptides. Arch Biochem Biophys. 1973 Aug;157(2):553–560. doi: 10.1016/0003-9861(73)90674-7. [DOI] [PubMed] [Google Scholar]
- Schnaitman C. A. Outer membrane proteins of Escherichia coli. IV. Differences in outer membrane proteins due to strain and cultural differences. J Bacteriol. 1974 May;118(2):454–464. doi: 10.1128/jb.118.2.454-464.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schnaitman C. A. Solubilization of the cytoplasmic membrane of Escherichia coli by Triton X-100. J Bacteriol. 1971 Oct;108(1):545–552. doi: 10.1128/jb.108.1.545-552.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shapiro A. L., Viñuela E., Maizel J. V., Jr Molecular weight estimation of polypeptide chains by electrophoresis in SDS-polyacrylamide gels. Biochem Biophys Res Commun. 1967 Sep 7;28(5):815–820. doi: 10.1016/0006-291x(67)90391-9. [DOI] [PubMed] [Google Scholar]
- Weiss M. J., Luria S. E. Reduction of membrane potential, an immediate effect of colicin K. Proc Natl Acad Sci U S A. 1978 May;75(5):2483–2487. doi: 10.1073/pnas.75.5.2483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto K. R., Alberts B. M., Benzinger R., Lawhorne L., Treiber G. Rapid bacteriophage sedimentation in the presence of polyethylene glycol and its application to large-scale virus purification. Virology. 1970 Mar;40(3):734–744. doi: 10.1016/0042-6822(70)90218-7. [DOI] [PubMed] [Google Scholar]
- Yu J., Fischman D. A., Steck T. L. Selective solubilization of proteins and phospholipids from red blood cell membranes by nonionic detergents. J Supramol Struct. 1973;1(3):233–248. doi: 10.1002/jss.400010308. [DOI] [PubMed] [Google Scholar]


