Abstract
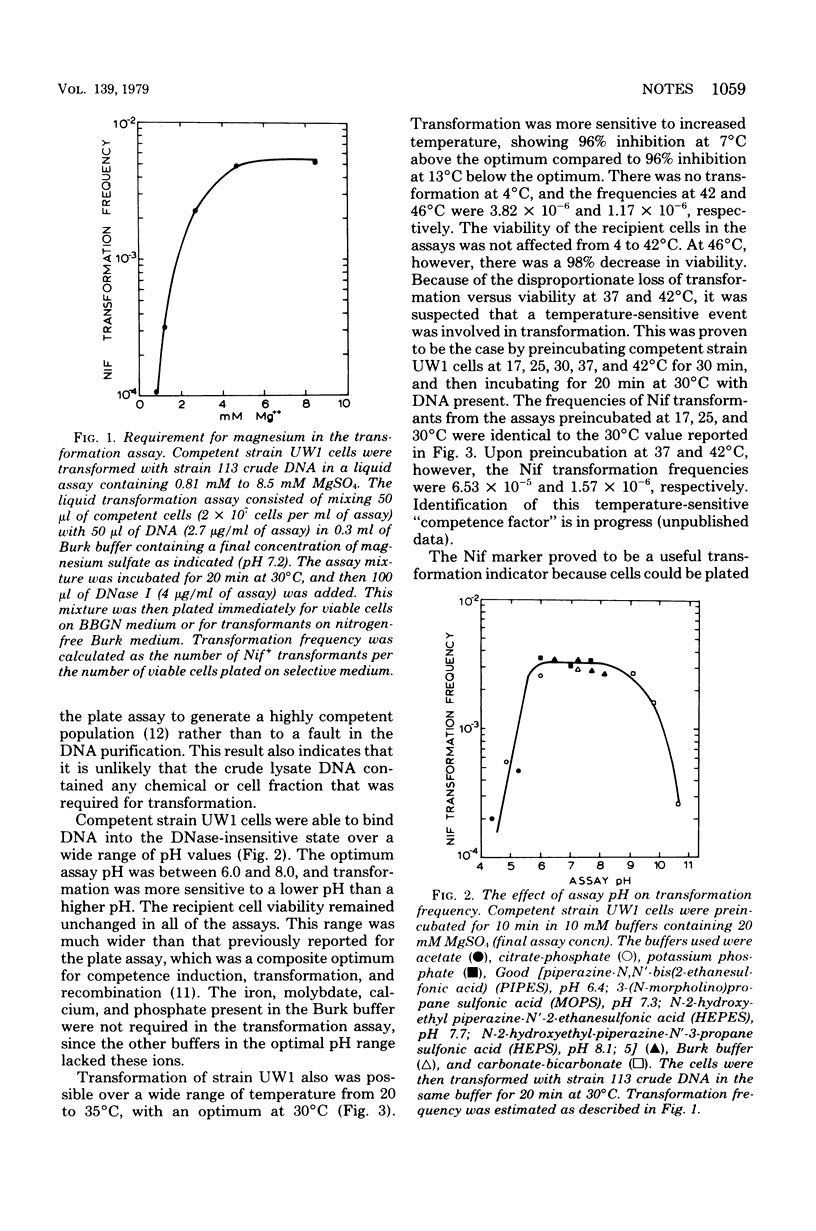
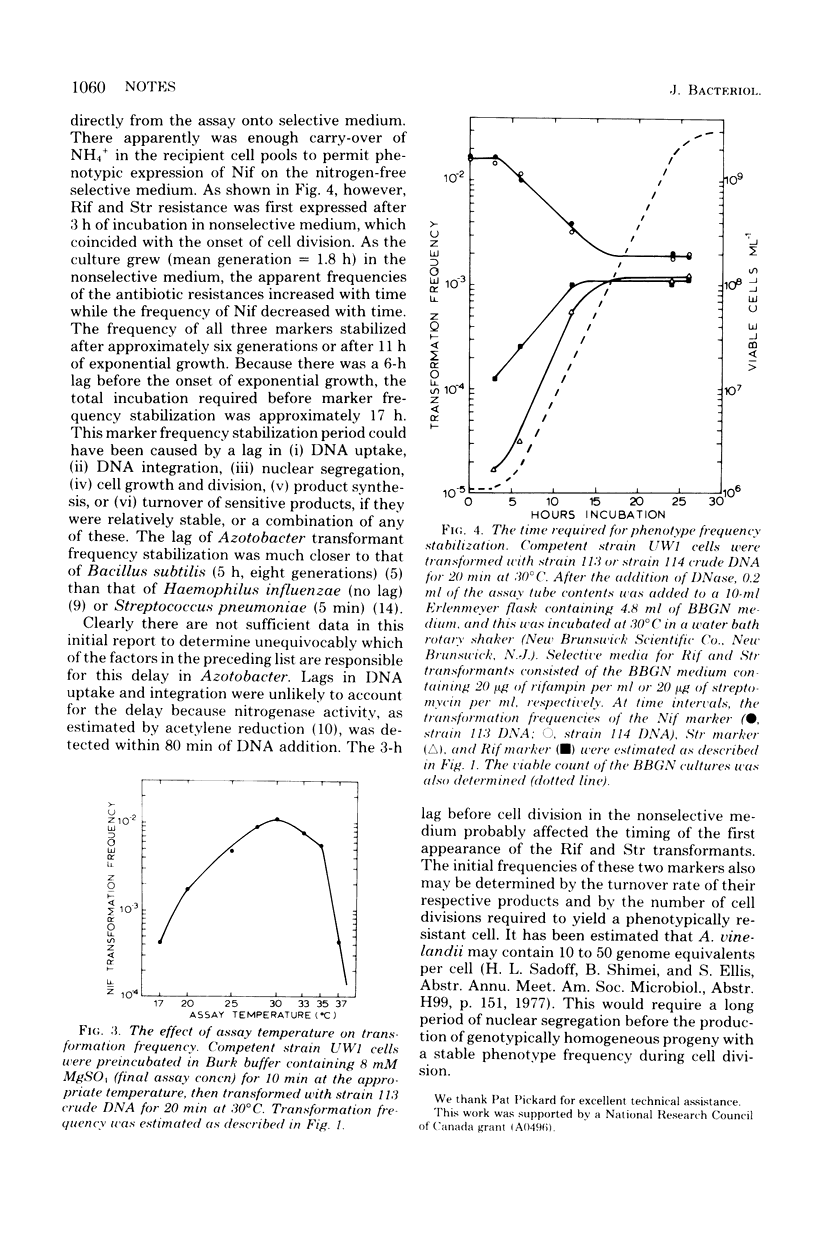
Optimal transformation of Azotobacter vinelandii OP required a 20-min incubation of the competent cells with deoxyribonucleic acid at 30 degrees C in buffer (pH 6.0 to 8.0) containing 8 mM magnesium sulfate. Nitrogen-fixing transformants of nitrogen fixation-deficient recipients could be plated immediately on selective medium, but transformants acquiring rifampin and streptomycin resistance required preincubation in nonselective medium. The three phenotypes achieved an approximately equal and stable frequency after 17 h (six generations) of growth in nonselective medium.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BURTON K. A study of the conditions and mechanism of the diphenylamine reaction for the colorimetric estimation of deoxyribonucleic acid. Biochem J. 1956 Feb;62(2):315–323. doi: 10.1042/bj0620315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bishop P. E., Brill W. J. Genetic analysis of Azotobacter vinelandii mutant strains unable to fix nitrogen. J Bacteriol. 1977 May;130(2):954–956. doi: 10.1128/jb.130.2.954-956.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bishop P. E., Dazzo F. B., Appelbaum E. R., Maier R. J., Brill W. J. Intergeneric transfer of genes involved in the Rhizobium-legume symbiosis. Science. 1977 Dec 2;198(4320):938–940. doi: 10.1126/science.929179. [DOI] [PubMed] [Google Scholar]
- Cosloy S. D., Oishi M. The nature of the transformation process in Escherichia coli K12. Mol Gen Genet. 1973 Jul 31;124(1):1–10. doi: 10.1007/BF00267159. [DOI] [PubMed] [Google Scholar]
- FOX M. S., HOTCHKISS R. D. Fate of transforming deoxyribonucleate following fixation by transformable bacteria. Nature. 1960 Sep 17;187:1002–1006. doi: 10.1038/1871002a0. [DOI] [PubMed] [Google Scholar]
- Good N. E., Winget G. D., Winter W., Connolly T. N., Izawa S., Singh R. M. Hydrogen ion buffers for biological research. Biochemistry. 1966 Feb;5(2):467–477. doi: 10.1021/bi00866a011. [DOI] [PubMed] [Google Scholar]
- Khan N. C., Sen S. P. Further observations on genetic transformation in pseudomonas. J Gen Microbiol. 1974 Aug;83(2):251–259. doi: 10.1099/00221287-83-2-251. [DOI] [PubMed] [Google Scholar]
- Kloos W. E. Factors affecting transformation of Micrococcus lysodeikticus. J Bacteriol. 1969 Jun;98(3):1397–1399. doi: 10.1128/jb.98.3.1397-1399.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- NESTER E. W., STOCKER B. A. BIOSYNTHETIC LATENCY IN EARLY STAGES OF DEOXYRIBONUCLEIC ACIDTRANSFORMATION IN BACILLUS SUBTILIS. J Bacteriol. 1963 Oct;86:785–796. doi: 10.1128/jb.86.4.785-796.1963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Page W. J., Sadoff H. L. Physiological factors affecting transformation of Azotobacter vinelandii. J Bacteriol. 1976 Mar;125(3):1080–1087. doi: 10.1128/jb.125.3.1080-1087.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Page W. J. Transformation of Azotobacter vinelandii strains unable to fix nitrogen with Rhizobium spp. DNA. Can J Microbiol. 1978 Mar;24(3):209–214. doi: 10.1139/m78-038. [DOI] [PubMed] [Google Scholar]
- Page W. J., von Tigerstrom M. Induction of transformation competence in Azotobacter vinelandii iron-limited cultures. Can J Microbiol. 1978 Dec;24(12):1590–1594. doi: 10.1139/m78-254. [DOI] [PubMed] [Google Scholar]
- Rudin L., Sjöström J. E., Lindberg M., Philipson L. Factors affecting competence for transformation in Staphylococcus aureus. J Bacteriol. 1974 Apr;118(1):155–164. doi: 10.1128/jb.118.1.155-164.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voll M. J., Goodgal S. H. Loss of activity of transforming deoxyribonucleic acid after uptake by Haemophilus influenzae. J Bacteriol. 1965 Oct;90(4):873–883. doi: 10.1128/jb.90.4.873-883.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- YOUNG F. E., SPIZIZEN J. INCORPORATION OF DEOXYRIBONUCLEIC ACID IN THE BACILLUS SUBTILIS TRANSFORMATION SYSTEM. J Bacteriol. 1963 Sep;86:392–400. doi: 10.1128/jb.86.3.392-400.1963. [DOI] [PMC free article] [PubMed] [Google Scholar]


