Abstract
Two new x-type high-molecular-weight glutenin subunits with similar size to 1Dx5, designated 1Dx5*t and 1Dx5.1*t in Aegilops tauschii, were identified by SDS–PAGE, RP-HPLC, and MALDI-TOF-MS. The coding sequences were isolated by AS-PCR and the complete ORFs were obtained. Allele 1Dx5*t consists of 2481 bp encoding a mature protein of 827 residues with deduced Mr of 85,782 Da whereas 1Dx5.1*t comprises 2526 bp encoding 842 residues with Mr of 87,663 Da. The deduced Mr's of both genes were consistent with those determined by MALDI-TOF-MS. Molecular structure analysis showed that the repeat motifs of 1Dx5*t were correspondingly closer to the consensus compared to 1Dx5.1*t and 1Dx5 subunits. A total of 11 SNPs (3 in 1Dx5*t and 8 in 1Dx5.1*t) and two indels in 1Dx5*t were identified, among which 8 SNPs were due to C-T or A-G transitions (an average of 73%). Expression of the cloned ORFs and N-terminal sequencing confirmed the authenticities of the two genes. Interestingly, several hybrid clones of 1Dx5*t expressed a slightly smaller protein relative to the authentic subunit present in seed proteins; this was confirmed to result from a deletion of 180 bp through illegitimate recombination as well as an in-frame stop codon. Network analysis demonstrated that 1Dx5*t, 1Dx2t, 1Dx1.6t, and 1Dx2.2* represent a root within a network and correspond to the common ancestors of the other Glu-D-1-1 alleles in an associated star-like phylogeny, suggesting that there were at least four independent origins of hexaploid wheat. In addition to unequal homologous recombination, duplication and deletion of large fragments occurring in Glu-D-1-1 alleles were attributed to illegitimate recombination.
WHEAT is the most important grain crop in the world, with total annual yields of almost 600 million tonnes (Shewry et al. 2001). The seed storage proteins, mainly including gliadins and glutenins that initially deposit in ER-derived protein bodies, have the ability to form gluten polymers linked by disulfide bonds, and these are among the largest protein molecules in nature (Wrigley 1996). Gliadins are monomeric and confer dough tractility whereas glutenins are polymeric and consist of high-molecular-weight (HMW) and low-molecular-weight (LMW) subunits, which contribute to the visco-elasticity of dough (Shewry et al. 1992). Although the HMW glutenin subunits (HMW-GSs) compose only 8–10% of the total extractable flour protein, they play an important role in flour-processing quality due to network formation in dough by gluten polymerization, allowing wheat flour to be processed into bread, pasta, noodles, and a range of other food products (Shewry et al. 1992; Shewry and Halford 2002; Ma et al. 2005).
The HMW subunits are encoded by the Glu-1 loci located on the long arms of chromosomes 1A, 1B, and 1D, and each locus consists of two closely linked genes, designated x- and y-types, with higher and lower molecular weights at two tightly linked loci, Glu-1-1 and Glu-1-2, respectively (Payne 1987). In general, the number of cysteine residues is four in x-type and seven in y-type subunits; repetitive motifs with hexapeptides and nanopeptides are present in both, and tripeptides are present only in x-type subunits (Shewry et al. 1992). Consequently, three loci encoding up to six HMW-GSs are present in hexaploid bread or common wheat (Triticum aestivum, AABBDD). However, silencing of specific genes leads to variation in the number of subunits from three to five while allelic variation in the subunits encoded by active genes results in proteins with different electrophoretic mobilities (Payne 1987; Shewry et al. 2001).
It is generally accepted that Aegilops tauschii (2n = 2x = 14, DD) is the D-genome donor of hexaploid wheat, which is presumed to have arisen from interspecific hybridization between T. dicoccum (AABB) and Ae. tauschii, with subsequent chromosome doubling, in southwestern Asia 8000–12,000 years ago (McFadden and Sears 1946a,b; Dvorak et al. 1998; Giles and Brown 2006). Recent investigations suggest that this polyploidization event occurred at least twice (Lelley et al. 2000; Caldwell et al. 2004; Giles and Brown 2006), suggesting multiple origins of hexaploid wheats (Dvorak et al. 1998; Allaby et al. 1999; Huang et al. 2002; Yan et al. 2003a,b; Gu et al. 2004).
Although a considerable amount of information is already available for the evolutionary origins of common wheat, some aspects need to be verified independently. For example, Ae. tauschii possesses extensive allelic variation in seed storage proteins (Lagudah and Halloran 1988; Yan et al. 2003a) that should provide useful evidence for insights into the evolution of hexaploid wheat, but only a few genes at the Glu-D1-1 locus of Ae. tauschii have been characterized (Wan et al. 2005). Evidence from molecular analysis demonstrated that the HMW glutenin subunits from wheat and related species have highly conserved structures, consisting of a signal peptide (21 residues), an N-terminal domain (86–89 residues in x-type and 104 in y-type subunits), a C-terminal domain (42 residues), and a central repetitive domain (630–830 residues) that is mainly responsible for differences in molecular weight of the subunits (Anderson et al. 1989; Wan et al. 2002, 2005; Yan et al. 2004; Sun et al. 2006; Zhang et al. 2006). The HMW glutenin genes, therefore, could derive from a common ancestor. The main molecular mechanisms for the evolution of glutenin genes at the Glu-1 loci appear to be single nucleotide polymorphism (SNP) and insertion/deletion (indel) variations, duplications, and deletions of large repeats, probably resulting from events such as unequal crossover and slip-mismatching (Anderson and Greene 1989; D'Ovidio et al. 1996; Zhang et al. 2006).
In this work, we identified and isolated two new x-type HMW subunit genes in Ae. tauschii accessions, and their molecular characteristics provided new evidence for multiple origins of hexaploid wheat. In particular, a large fragment deletion in the repetitive domain occurring in the Escherichia coli expression system suggested illegitimate recombination as a possible mechanism for duplication and deletion of large fragments at the Glu-D1-1 locus.
MATERIALS AND METHODS
Plant materials:
Two accessions of Ae. tauschii (Coss.) Schmal., TD81 and TD130, were kindly provided by GenBank (Braunschweig, Germany). The Yugoslav common wheat cultivar Dunav and Chinese Spring (CS) were used as standards for HMW-GS identification.
Sodium dodecyl sulfate–polyacrylamide gel electrophoresis, reverse-phase high-performance liquid chromatography, and matrix-assisted laser desorption/ionization time of flight mass spectrometry:
HMW glutenin subunits were extracted and analyzed by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS–PAGE) based on the protocol of Yan et al. (2003a). Reverse-phase high performance liquid chromatography (RP-HPLC) was based on the method of Andrews et al. (1994) with slight modifications. The column was deaerated and equilibrated; the mixture (8 μl) was used and eluted with a linear 50-min solvent gradient of 21–48% acetonitrile containing trifluoroacetic acid (0.06%) at the flow rate of 1.00 ml/min on Agilent 1100. The column was maintained at 50° and was returned to the initial solvent composition and reequilibrated for 15 min before the next analysis. Eluted protein components were detected at 210 nm. Matrix-assisted laser desorption/ionization time of flight mass spectrometry (MALDI-TOF-MS) for determining the molecular mass of HMW-GS was as described by Zhang et al. (2006).
PCR amplification, cloning, and sequencing:
Genomic DNA was extracted as described by Sun et al. (2004). The complete coding region sequences of the genes encoding x-type subunit genes at the Glu-D1t locus were amplified by primers designed from the sequences of subunits 1Dx5 and 1Dx2 (Sugiyama et al. 1985; Anderson et al. 1989), namely P1:5′-ATGGCTAAGCGGTTAGTC-3′ and P2:5′-GCTGCAGA GAGTTCTATC-3′ (synthesized by Sangong, Shanghai, China). The expected amplified products covering the start and stop codons were ∼2500 bp according to previously characterized x-type HMW subunit genes (Anderson et al. 1989).
PCR was carried out using a Perkin-Elmer Cetus DNA thermal cycler (PE Applied Biosystems, Foster City, CA). A 50-μl reaction mix was used, including 100 ng of DNA, 25 μl of 2× GC buffer II (MgCl2 plus), 0.4 mm of each dNTP, 0.5 μm of each primer, 2.5 units LA Taq polymerase (TaKaRa). The PCR reaction was performed at 94° for 2 min, followed by 35 cycles at 94° for 45 sec, at 58° for 60 sec, and at 72° for 150 sec, and then concluded at 72° for 10 min. The amplified products of expected size were cloned into pGEM-T vector (Promega, Madison, WI) or pET30a (Novagen, expression step as follows). DNA sequencing of three clones was performed on an automatic DNA sequencer (TaKaRa Biotech, DaLian City, China).
Identification of SNPs and indels:
The identification of SNP and indel variations in the cloned ORFs was based on multiple alignments of DNA and amino acid sequences and performed by ClustalW (Thompson et al. 1994).
Heterologous expression in E. coli and N-terminal microsequencing:
The cloned HMW subunit genes were amplified to remove the signal peptides using the primers Pbd-1:5′-ACC CAT ATG GAA GGT GAG GCC TCT-3′and Pbd-2:5′-CTA GAA TTC CTA TCA CTG GCT GGC-3′ (the added NdeI and EcoRI restriction sites are underlined).
PCR products were cloned into the bacterial expression vector pET30a (Novagen), and the hybrid vector (pET30a-1Dx5*t/1Dx5.1*t) was transformed into E. coli strain BL21 (DE3) pLsS. BL21 (DE3) pLsS cells containing the hybrid vector were grown in 2× YT medium (containing 50 μg ml−1 kanamycin and 34 μg ml−1 chloramphenicol) in a shaking incubator at 37° until the OD600 reached 0.6. The expression of HMW subunit proteins was induced by adding 1–1.2 mm isopropyl β-Δ-thiogalactopyranoside for 4–6 hr. The expressed proteins were extracted from 1.2 ml of bacterial cells according to the method of Wan et al. (2002) for SDS–PAGE analysis.
N-terminal amino acid microsequencing of the expressed proteins was performed by PROCISE cLC 491 protein sequence system (Applied Biosystems) after transferring the proteins from the SDS gel to a polyvinylidene fluoride microporous membrane (Millipore, Bedford, MA) with a tank system (Bio-Rad mini trans-blot cell).
Network construction and phylogenetic analysis:
Networks were constructed as described by Allaby and Brown (2001) using the nucleotide sequences of the signal peptide plus the N-terminal domain, which were considered phylogenetically informative (Li et al. 2004). A neighbor-joining tree was constructed by MEGA3 on the basis of the alignment of complete coding sequences using ClustalW. Bootstrap values were calculated as a percentage of 1000 trials.
RESULTS
Identification of novel HMW-GS in Ae. tauschii:
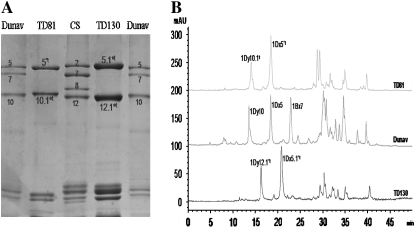
SDS–PAGE analyses (Figure 1A) showed that Ae. tauschii accessions TD81 and TD130 possessed a pair of novel HMW glutenin subunits, designated 1Dx5*t +1Dy10.1t and 1Dx5.1*t +1Dy12.1*t, respectively. The coding gene for the 1Dy10.1t subunit was isolated and characterized in an earlier investigation (Zhang et al. 2006). The subunits 1Dx5*t and 1Dy10.1t moved slightly faster and slower than the standard 1Dx5 and 1Dy10 subunits whereas 1Dx5.1*t and 1Dy12.1*t ran slightly slower and faster than the 1Dx5 and 1Dy12 subunits, respectively.
Figure 1.—
Identification of HMW subunits from Ae. tauschii accessions TD81 and TD130. (A) SDS–PAGE. (B) RP-HPLC. Dunav and/or CS were used as references.
Figure 1B shows the RP-HPLC patterns of HMW glutenin subunits TD81 and TD130 as well as a control, Dunav (N, 7, 5+10); two subunits in each accession were separated. According to Gianibelli et al. (2002), the surface hydrophobicities of HMW-GS subunits were different and the order was 1Dy < 1By < 1Dx < 1Bx <1Ax. Therefore, the x- and y-type subunits were readily identified as indicated in Figure 1B. The hydrophobicities of the seven HMW-GSs were 1Bx7 > 1Dx5.1*t > 1Dx5*t and 1Dx5 > 1Dy12.1*t > 1Dy10.1t > 1Dy10.
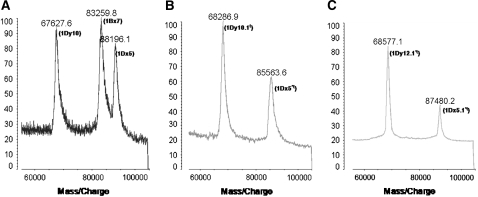
The accurate molecular weights of the HMW glutenin subunits from TD81 and TD130 as well as the control, Dunav (N, 7, 5+10), were obtained by MALDI-TOF-MS (Figure 2). Both accessions possessed an x-type HMW-GS with molecular masses of 85,563.6 Da in TD81 and 87,480.2 Da in TD130, which correspond closely to those of the 1Dx5*t and 1Dx5.1*t subunits, respectively. Both subunits were smaller than the mature 1Dx5 subunit (88,196.1 Da) from cultivar Dunav, with differences of 2632.5 and 715.9 Da, respectively. The molecular mass of the 1Dx5.1*t subunit was different from that indicated for SDS–PAGE in Figure 1A. The anomalous electrophoretic behavior might result from fundamental conformational and structural differences between 1Dx5 and 1Dx5.1*t, similar to those reported for 1Dy10 and 1Dy12 (Goldsbrough et al. 1989), as well as gliadins (Tatham and Shewry 1985).
Figure 2.—
The accurate molecular mass of HMW glutenin subunits from Dunav (A), TD81 (B), and TD130 (C) as determined by MALDI-TOF-MS. The subunits 1Dx5*t and 1Dx5.1*t with 85,563.6 and 87,480.2 Da, respectively, are shown.
Molecular characterization of 1Dx5*t and 1Dx5.1*t subunit genes:
The designed degenerate oligonucleotide primers P1 + P2 were used to amplify the coding regions of the x-type HMW-GS genes in TD81 and TD130. Both accessions gave amplified products of 2500 bp, consistent with the size of the 1Dx5 subunit gene from common wheat (Anderson et al. 1989). After purifying, cloning, and sequencing of the expected segments, two complete coding sequences with typical characters of HMW-GS genes were obtained. Both genes ended at a double stop codon (TGA, TAG) and introns were not present as for most other HMW-GS genes characterized so far. The 1Dx5*t gene consisted of 2487 bp encoding a mature protein of 827 residues whereas 1Dx5.1*t had 2532 bp encoding 842 amino acid residues.
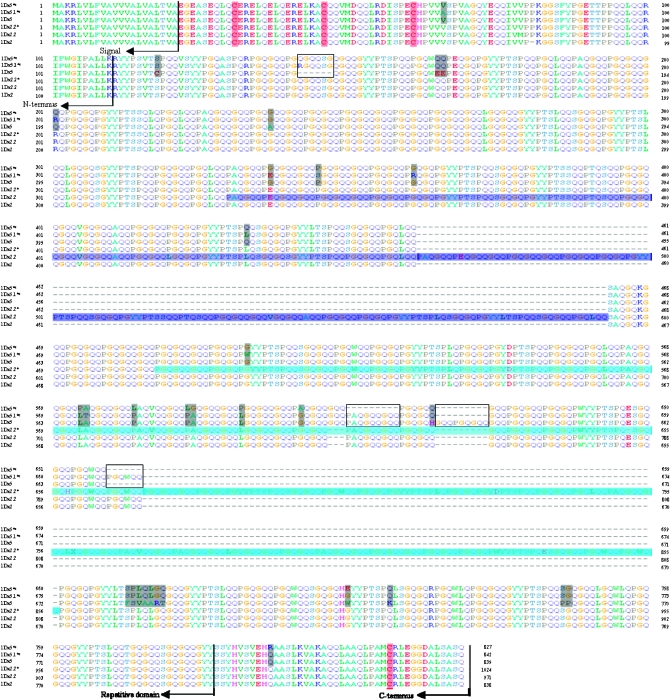
Comparison of the deduced amino acid sequences (Figure 3) indicated that the 1Dx5*t and 1Dx5.1*t subunits shared a primary structure identical to those of 1Dx5 and other subunits from common wheat. Three structural domains were present in all subunits: a nonrepetitive N-terminal domain of 89 amino acid residues, repetitive domains of 109 and 112 residues, respectively, and a nonrepetitive C-terminal domain of 42 residues. Both subunits contained the expected four cysteine residues at conserved positions (Table 1): three in the N-terminal domain (at positions 31, 46, and 61) and one in the C-terminal domain (at positions 815 and 830). Like their orthologous subunits, the repetitive domains consisted of tandem and interspersed repeats based on tripeptide (consensus GQQ), hexapeptide (PGQGQQ), and nonapeptide (GYYPTSLQQ) motifs, indicating that they were typical x-type subunits. The deduced molecular masses were 85,782 Da (1Dx5*t) and 87,663 Da (1Dx5.1*t), which were consistent with those determined by MALDI-TOF-MS, and the differences were within the limits of experimental error in the mass range of HMW glutenin (Hickman et al. 1995). This suggested that both subunits lacked extensive post-translational modifications, such as glycosylation and phosphorylation, as did other HMW-GSs analyzed by different proteome approaches (Cozzolino et al. 2001; Cunsolo et al. 2004; Alberghina et al. 2005). The two novel subunit genes were deposited in the GenBank under accession nos. DQ681076 (1Dx5*t) and DQ681077 (1Dx5.1*t).
Figure 3.—
Multiple sequence alignment of the deduced amino acid sequences of the 1Dx5*t, 1Dx5.1*t, 1Dx5 1Dx2.2*, 1Dx2.2, and 1Dx2 subunits. Alignments were assembled by eye to demonstrate the repeat structure of the central domains. Cysteine residues are highlighted by red shading. Deletions are indicated by dashes, The gray shading and box indicate differences and indels among 1Dx5*t, 1Dx5.1*t, and 1Dx5, respectively. The regions repeated directly in 1Dx2.2* and 1Dx2.2 are dark blue and light blue, respectively.
TABLE 1.
Characteristics of the deduced mature proteins of the 1Dx5*t and 1Dx51*t subunits compared to previously reported HMW-GSs from Ae. tauschii and common wheat
| No. of amino acid residues and repeat unit variations
|
||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| RD
| ||||||||||||||||||||||||||||||
| Tripeptide variants
|
Hexapeptide variants
|
Nanopeptide variants
|
||||||||||||||||||||||||||||
| 1 | 2 | 3 | 1 | 2 | 3 | 4 | 5 | 6 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | No. of Cys residues
|
MW (Da) | |||||||||||
| Subunits | ND | G | Q | Q | V/Ta | P | G | Q | G | Q | Q | V/Ta | G | Y | Y | P | T | S | L | Q | Q | V/Ta | V/T%b | CD | M | ND | RD | CD | Total | |
| Dx5*t | 89 | 1 | 1 | 1 | 2/20 | 20 | 10 | 0 | 13 | 0 | 16 | 46/70 | 2 | 0 | 1 | 2 | 0 | 0 | 12 | 2 | 3 | 14/19 | 67.41 | 42 | 0.91 | 3 | 0 | 1 | 4 | 85,782 |
| Dx5.1*t | 89 | 1 | 2 | 1 | 3/21 | 23 | 13 | 1 | 14 | 2 | 16 | 52/72 | 1 | 0 | 1 | 2 | 0 | 0 | 15 | 3 | 3 | 16/19 | 75.82 | 42 | 1.03 | 3 | 0 | 1 | 4 | 87,663 |
| Dx5 | 89 | 1 | 1 | 1 | 2/22 | 20 | 13 | 1 | 14 | 3 | 18 | 50/71 | 2 | 0 | 1 | 2 | 0 | 1 | 15 | 3 | 4 | 15/19 | 72.22 | 42 | 1.08 | 3 | 1 | 1 | 5 | 87,189 |
| Dx2 | 88 | 1 | 2 | 1 | 3/20 | 21 | 11 | 0 | 14 | 2 | 15 | 49/72 | 1 | 0 | 1 | 2 | 0 | 0 | 15 | 3 | 3 | 15/19 | 70.32 | 42 | 0.97 | 3 | 0 | 1 | 4 | 87,023 |
| Dx2.1t | 89 | 2 | 1 | 1 | 3/21 | 20 | 11 | 0 | 13 | 2 | 16 | 47/71 | 2 | 0 | 1 | 2 | 0 | 0 | 15 | 2 | 3 | 15/19 | 68.89 | 42 | 0.97 | 3 | 0 | 1 | 4 | 88,914 |
| Dx2.2t | 89 | 1 | 2 | 1 | 3/23 | 20 | 15 | 0 | 15 | 1 | 20 | 54/81 | 1 | 0 | 1 | 2 | 0 | 0 | 18 | 4 | 3 | 18/21 | 70.59 | 42 | 0.98 | 3 | 0 | 1 | 4 | 103,048 |
| Dx2t | 89 | 1 | 1 | 1 | 2/21 | 20 | 9 | 0 | 12 | 2 | 16 | 45/71 | 1 | 0 | 1 | 2 | 0 | 0 | 15 | 2 | 3 | 15/19 | 66.67 | 42 | 0.92 | 3 | 0 | 1 | 4 | 88,700 |
| Dx3t | 89 | 1 | 1 | 1 | 2/21 | 20 | 9 | 1 | 13 | 2 | 16 | 45/71 | 1 | 0 | 1 | 2 | 1 | 0 | 15 | 2 | 3 | 15/19 | 66.67 | 42 | 0.96 | 3 | 0 | 1 | 4 | 88,829 |
| Dx5.2t | 89 | 1 | 1 | 1 | 2/21 | 21 | 11 | 0 | 13 | 3 | 11 | 45/71 | 1 | 1 | 1 | 2 | 0 | 0 | 14 | 2 | 4 | 15/19 | 66.67 | 42 | 0.93 | 3 | 1 | 1 | 5 | 86,779 |
| Dy12 | 104 | — | — | — | — | 18 | 6 | 3 | 13 | 6 | 5 | 30/50 | 1 | 10 | 2 | 1 | 8 | 0 | 10 | 0 | 2 | 15/19 | 65.22 | 42 | 1.21 | 5 | 1 | 1 | 7 | 68,711 |
| Dy10 | 104 | — | — | — | — | 15 | 6 | 3 | 12 | 4 | 5 | 25/48 | 1 | 10 | 2 | 1 | 7 | 0 | 10 | 0 | 2 | 15/19 | 59.70 | 42 | 1.15 | 5 | 1 | 1 | 7 | 67,495 |
M, average number of variants per unit, including hexapeptides and nonapeptides. ND, N-terminal domain. RD, repetitive domain. CD, C-terminal domain. MW, molecular weights calculated from the derived amino acid sequences.
Variant motif number/total motif number.
Percentage of variant motifs relative to total motifs.
1Dx5*t and 1Dx5.1*t had 12 residues fewer and three residues more, respectively, than 1Dx5. There were 19 amino acid substitutions, a hexapeptide insertion, and a hexapeptide and two nonapeptide deletions in 1Dx5*t relative to 1Dx5, and 24 amino acid substitutions, two hexapeptide insertions, and a nonapeptide deletion in 1Dx5.1*t relative to 1Dx5 (Figure 3). The characteristics of the deduced mature proteins of 1Dx5*t and 1Dx5.1*t and previously reported HMW-GSs from Ae. tauschii and common wheat are summarized in Table 1. In particular, the frequencies of amino acid variations from the tripeptide, hexapeptide, and nonapeptide consensus at each of the 18 positions are indicated. Obviously, more variations occurred at positions 1 and 6, and at positions 4 and 2, among hexapeptides of x-type subunits, whereas the variant positions in y-type subunits were 1 and 4. There were more variations at positions 7, 8, and 9 of the nonapeptides in x-type subunits whereas the variant positions in y-type subunits were mostly 2, 7, and 5. The average numbers of variants per repeat unit and the mean frequency of variant units of subunit 1Dx5*t were relatively lower, indicating that its repeat units were closer to the consensus.
SNP and indel analyses:
The coding sequences of the 1Dx5*t and 1Dx5.1*t genes were aligned with the other 11 Glu-1Dx subunit genes and the SNPs and indels were identified (Table 2). A total of 11 SNPs were detected at different positions and the numbers of SNPs in 1Dx5*t and 1Dx5.1*t were 3 (1/827 bp) and 8 (1/316 bp), respectively. Of the 11 SNPs, 8 were due to A-G or C-T transitions (average 73%), approximating the results for Glu-1Dy10.1t (Zhang et al. 2006). In nine cases, SNPs at positions 45 and 711 in 1Dx5.1*t were found to produce amino acid residue substitutions (nonsynonymous SNP), namely 1718 T → C at position 573 (leucine → proline), 1745 C → T at postion 582 (proline → leucine), 2384 A → G at position 795 (glutamine → arginine) in 1Dx5*t, and 197 T → C at position 66 (valine → alanine), 425 A → G at position 142 (glutamine → arginine), 1033 C → T at position 345 (proline → serine), 1081 G → A at position 361 (glycine → arginine), 1501 G → T at position 501 (glycine → tryptophan), and 1720 G → A at position 574 (alanine → threonine) in 1Dx5.1*t. In addition, 15 and 3 residue deletions, located at positions 1824–1838 and 1848–1850, respectively, were also found in 1Dx5*t.
TABLE 2.
Positions of SNPs and indels identified in the 1Dx5*t and 1Dx51*t ORFs
| HMW-GS gene | 45 | 197 | 425 | 711 | 1033 | 1081 | 1501 | 1718 | 1720 | 1745 | 1824–1838 | 1848–1850 | 2384 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1Dx5*t | C | T | A | G | C | G | G | C | G | T | – – – – – – – – – – – – – – – | – – – | G |
| 1Dx5.1*t | A | C | G | A | T | A | T | T | A | C | G C C A G G A C A A G G G C A | G C A | A |
| 11 other Glu-1Dx genes | C | T | A | G | C | G | G | T | G | C | G C C A G G A C A A G G G C A | G C A | A |
SNPs are indicated by italics and indels by a horizontal dash.
E. coli expression analysis:
The coding regions of the two new genes were expressed in E. coli using the pET-30α expression vector after removal of the sequences encoding signal peptide (Figure 4). As shown in Figure 5, A-N5*t and B-N5.1*t showed the expressed proteins of 1Dx5*t and 1Dx5.1*t ORFs in E. coli comigrating with the protein subunits present in TD81 and TD130 seeds, respectively. This confirmed that the cloned sequences were an accurate representation of each ORF and ruled out the possibility of glycosylation and other post-translational modifications of both subunits, consistent with the results of mass spectrometry.
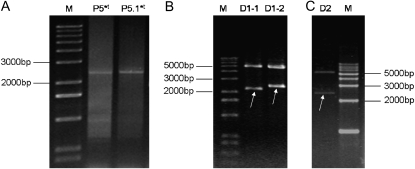
Figure 4.—
Construction and identification of expression vectors pET30a-1Dx5*t and pET30a-1Dx5.1*t. (A) Amplified products using the primer Pbd-1+Pbd-2 from the hybrid pGEM-T vector containing the identified genes. (B) Digestion of the hybrid pET30a-1Dx5*t with NdeI and EcoRI restriction enzymes in tracks D1-1 and D1-2. (C) Digestion of the hybrid pET30a-1Dx5.1*t with NdeI and EcoRI restriction enzymes in track D3. M, 1-kb ladder markers. Inserted fragments are indicated by arrows.
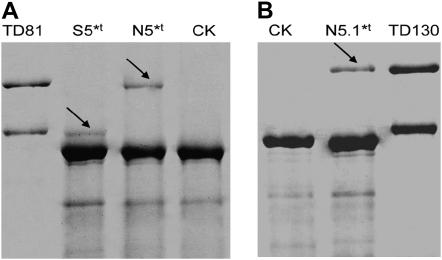
Figure 5.—
Expression of the cloned 1Dx5*t and 1Dx5.1*t gene ORFs in E. coli. (A) Expressed proteins of different pET30a-1Dx5*t plasmid clones. The expressed normal and smaller proteins are indicated in tracks N5*t and S5*t, respectively, by arrows. (B) Expressed protein of pET30a-1Dx5.1*t plasmid clones (indicated by the arrow).
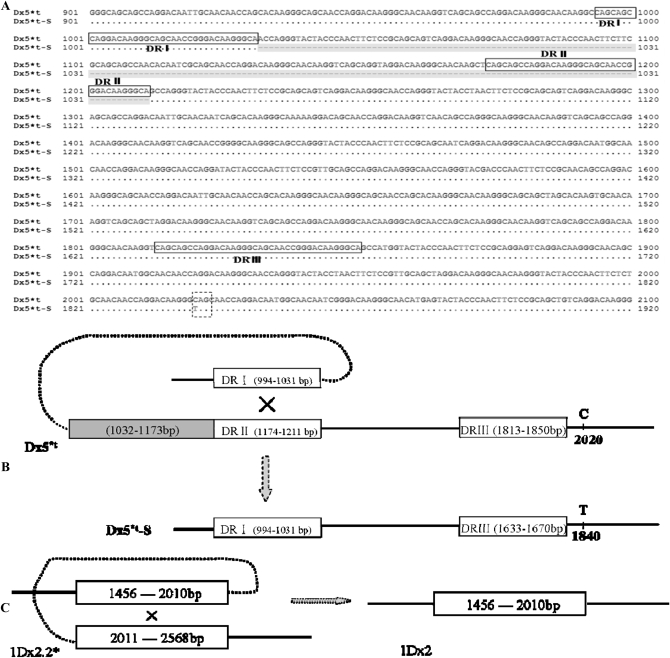
Figure 5 shows that several other hybrid clones expressed a smaller protein (Figure 5A, S5*t) relative to the authentic subunits present in seeds. The results of N-terminal sequencing, however, confirmed that the proteins of N5*t, N5.1*t, and S5*t from E. coli expression and 1Dx5*t from seed extracts had the same amino acid residues. As shown in Figure 4A, the size of PCR products amplified by the Pbd-1 and Pbd-2 primers was exactly as expected, but after cloning with the expression vector pET-30a, some target fragments became shorter (Figure 4B, D1-1), including expression of a smaller peptide (Figure 5A, S5*t). After sequencing the hybrid expressing plasmid pET30a-1Dx5*t, it became clear that a long deletion of 180 bp (from 1211 to 1032) and a key mutation (C → T) at position 1840 forming a stop codon TAG) were present (Figure 6A). The derived molecular weight of the small protein was 65,220.33 Da, again consistent with the result of SDS–PAGE. The sequence analysis showed that there were three direct repeats (DR) in the repetitive domain as indicated by the boxes designated DRI, DRII, and DRIII in Figure 6. We believe that this was due to illegitimate recombination between repeats DRI and DRII. A proposed mechanism for the 180-bp deletion resulting from illegitimate recombination in 1Dx5*t is shown in Figure 6B. A similar phenomenon was reported in Arabidopsis (Devos et al. 2002). We also considered the possibility of illegitimate recombination between the other two pairs of repeats (DRI and DRIII or DRII and DRIII), but the frequencies would be much lower because of the much longer distances separating them. It is also possible that illegitimate recombination occurred in the 1Dx2.2* gene as shown in Figure 6C, resulting in a 558-bp deletion (from 2568 to 2011) and the generation of 1Dx2 in common wheat.
Figure 6.—
Comparison of the coding sequences of 1Dx5*t and 1Dx5*t-S from the deletion hybrid pET30a-1Dx5*t (A), the possible mechanisms generating 1Dx5*t-S (B) and 1Dx2 from 1Dx2.2* through illegitimate recombination (C). (A) The same sequences and deletions are indicated by a dot and by light gray shaded dashes, respectively. The direct repeats are boxed and designated as DRI, DRII, and DRIII, respectively. The key mutation resulting in a stop codon is boxed by dashed line. (B) Illegitimate recombination between DRI and DRII (see A) resulting in the deletion of the domain (the shaded box) and repeat II. (C) The illegitimate recombination that probably occurred in 1Dx2.2* that resulted in the 1Dx2 gene. The dotted lines in B and C indicate the illegitimate recombination event.
Similar phenomena were observed for LMW-GS gene recombination clones when using different E. coli strains (Altenbach 1998; Masci et al. 1998). There, single deletions of 50–200 bp occurred within the repetitive domains of different recombinant clones. Differences among HMW glutenin subunits are mainly due to size and variation in the central repeat regions. Our investigations present direct evidence for recombinant clones and provide useful information for further studies on glutenin gene variation and its evolutionary direction.
Network and neighbor-joining analysis:
To investigate the phylogenetic relationships of x-type HMW-GSs encoded by the Glu-D1-1 locus, we constructed a network with the signal peptides and N-terminal coding sequences and neighbor-joining trees with complete coding sequences (Figure 7). Thirteen x-type genes were analyzed, namely: 1Dx5*t and 1Dx5.1*t (this study, DQ681076 and DQ681077), 1Dx5 (Anderson et al. 1989, X12928), 1Dx2 (Sugiyama et al. 1985, X03346), 1Dx2.1t (AF480486), 1Dx2t (AF480485), 1Dx1.5t (AY594355), 1Dx1.6t (DQ857243), 1Dx2.1 (AY517724), 1Dx2.2 (Wan et al. 2005, AY159367), 1Dx2.2* (D'Ovidio et al. 1996; Wan et al. 2005, AJ893508), 1Dx3t (DQ307383), and 1Dx5.2t (DQ307384).
Figure 7.—
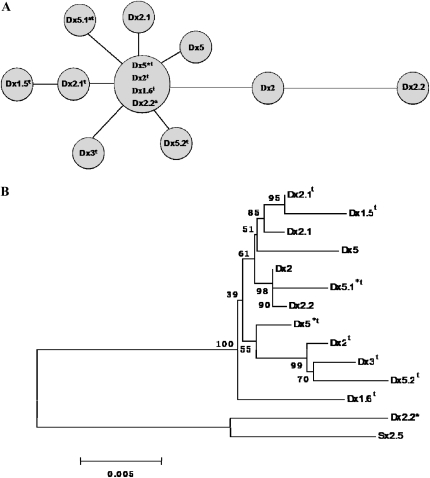
Phylogenetic network based on nucleic acid sequence variations of signal and N-terminal domains (A) and the neighbor-joining tree based on multi-alignment of complete coding sequences of 13 Glu-D1-1 alleles from Ae. tauschii and common wheat and of 1Sx2.5 from the Sb genome of Ae. bicornis (B).
As shown in Figure 7A, the network placed the alleles 1Dx5*t, 1Dx2t, 1Dx1.6t, and 1Dx2.2* at a principal node. Among them, 1Dx2.2* is the largest subunit detected so far in hexaploid wheat and related species. The other alleles radiated in a star-like phylogeny from the principal node. Allele 1Dx5.1*t, as well as 1Dx5, was directly linked to the principal node and had no direct link with any other allele. Moreover, 1Dx5 has an extra cysteine in the repetitive region, which is believed to have a positive effect on quality properties. This implied that, although deriving from the ilk principal node, one relative independent mutation case might have occurred in the 1Dx5 during the evolutionary process. Apparently, a point mutation at position 290 (C → G) in 1Dx5 produced an amino acid residue substitution at position 97 (serine → cysteine) and therefore resulted in generating the extra cysteine. This extra cysteine remains in 1Dx5.1*t and 1Dx5.1*t, indicating that these two proteins can be used as new source for wheat quality improvement.
The 1Dx2.1t and 1Dx2 genes to the principal node still linked directly to 1Dx1.5t and 1Dx2.2, respectively, implying that 1Dx1.5t and 1Dx2.2 might be more recent genes deriving from 1Dx2.1t and 1Dx2, respectively. The network analysis demonstrated that 1Dx5*t, 1Dx2t, 1Dx1.6t, and 1Dx2.2* might represent ancestral sequences and that recombination with other star-like phylogenic Glu-1Dx alleles had not occurred. This suggested that there were at least four Ae. tauschii sources that contributed germplasm to the D genome of hexaploid wheats.
The neighbor-joining tree (Figure 7B) revealed that Glu-1Dx subunit genes were apparently divided into two clades; the greater group consisted of 12 genes at the top and the other group had only 2 genes at the bottom. Within the upper clade, a bootstrapping value of 100% gave high support for the spilt between 1Dx1.6t and the other alleles. The 1Dx5*t and 1Dx5.1*t were clustered into a separate subgroup. In the other clade, it was interesting that 1Dx2.2* was clustered with 1Sx2.5 from the Sb genome of Ae. bicornis, which was used as the outlier. This is consistent with the network analysis indicating that 1Dx2.2* represents an ancestral gene although it was detected only in hexaploid wheats.
DISCUSSION
Network analysis for revealing relationships between glutenin alleles:
Network analysis has been applied to study relationships between 5S rDNA sequences (Allaby and Brown 2001) and Glu-Dy (Giles and Brown 2006) alleles. Such studies provide insights into the origin and evolution of hexaploid wheats and related species. Since network analysis makes few assumptions about the direction of evolution, the extent of sexual isolation, and the pattern of ancestry and descent, it describes relationships between sequences in a more realistic fashion than conventional tree building (Allaby and Brown 2001). Previous research showed that it was valid to study glutenin phylogenetics by comparing partial glutenin sequences. Furthermore, the majority of partial alignments gave the same topology as the complete sequence alignments except for several of the partial repetitive regions (Allaby et al. 1999). Because the software for construction of networks has not been developed and the difficulties in dealing with the complications involved in complete ORFs of 2.5–3.1 kb in length, it appears to be feasible and reasonable to perform network analysis with partial glutenin sequences. Giles and Brown (2006) made a network analysis by examining a 284-bp sequence from the promoter region of the Glu-Dy locus and revealed aspects of the evolution and geographical origins of hexaploid wheat. In this work, we used the signal peptides and N-terminal domains of Glu-D1-1 alleles to construct a network because the signal peptides and N-terminal sequences are relatively conserved among different HMW glutenin subunit genes. It is probably owing to their important roles, such as targeting the newly synthesized subunits into protein bodies and maintaining the high-order structure of the subunits. This suggests that these regions are subject to progressive changes during the evolution of HMW glutenin subunit genes and therefore are phylogenetically informative (Li et al. 2004).
Implications for the evolution of the Glu-D1-1 locus and the origin of cultivated wheats:
As shown in this work, the 1Dx5*t, 1Dx2t, and 1Dx1.6t genes from diploid Ae. tauchii and 1Dx2.2* from common wheat were located at the shared principal node in the phylogenetic network, representing a root within a network and corresponding to the common ancestor of the genes in the associated star-like phylogeny (Allaby and Brown 2001). These nodal Glu-D1-1 genes appear to be of considerable antiquity, suggesting that there were at least four independent origins of hexaploid wheat. Two very large HMW subunits, 1Dx2.2* and1Dx2.2, were detected only in hexaploid wheat, and similar large Dx subunits have not been found in Ae. tauschii accessions (Lagudah and Halloran 1988; Yan et al. 2003a). Molecular characterization showed that the large size of the 1Dx2.2* gene is due to a single duplication of 561 bp within the repetitive domain, possibly arising from unequal crossing over during meiosis (D'Ovidio et al. 1996). According to our network analysis, it is most likely that the 1Dx2.2* gene could have been present in Ae. tauschii and then transferred by rare hybridization into common wheat as suggested by D'Ovidio et al. (1996). The absence of large 1Dx2.2*-like Dx genes in Ae. tauschii is probably due to the limited number of accessions surveyed so far or could have been lost from the population.
In terms of evolutionary analysis, the Glu-Dx subunit alleles arose mainly from four principal ancestor genes. We propose that 1Dx2.2*, deriving from a more primitive gene, was present in Ae. tauschii and was transferred by rare outcrossing into common wheats. A mutational event, probably by illegitimate recombination, resulted in loss of a large DNA fragment and generation of the 1Dx2 gene as indicated in Figure 6C (the corresponding amino acid sequences of the duplicated DNA sequences are light blue in Figure 3). In the recent evolutionary timescale of common wheats, one large fragment duplication in the repetitive domain of 1Dx2 (shaded dark blue in Figure 3) presumably resulted from unequal crossing over as well as illegitimate recombination to generate the 1Dx2.2 allele.
The hypotheses of multiple origins of the D genome in hexaploid wheat are supported by the studies of Lagudah and Halloran (1988) and Yan et al. (2003b) as well as by evidence from the A and B genomes (Allaby et al. 1999; Gu et al. 2004). More recently, Giles and Brown (2006) investigated the evolution and geographical origins of hexaploid wheat by examining Glu-Dy alleles. The existence of two shared alleles suggested that there were at least two independent origins of hexaploid wheat. Other investigations demonstrated that the A and B genomes might have introgressed into hexaploid wheat at different times and rates (Blatter et al. 2004; Zhang et al. 2006) and that, on the basis of the fact that there are two distinct types of alleles at the Glu-A1 locus, hexaploid wheat might have more than one tetraploid ancestor (Gu et al. 2004). Furthermore, surveys of sequence tagged sites (Talbert et al. 1998), RFLP markers (Dvorak et al. 1998), and microsatellites (Lelley et al. 2000) and analyses of the Xwye838 and Gss loci (Caldwell et al. 2004) also indicated multiple origins for hexaploid wheat.
A possible mechanism for duplication and deletion in Glu-1 genes through illegitimate recombination:
Modern hexaploid wheat is an allohexaploid species with genomes A, B, and D and an extremely large and complex genome (16,000 Mb). Although hexaploid wheat is the product of the hybridization between tetraploid wheat (AABB) and diploid goat grass (DD) that happened ∼10,000 years ago, its genomes have been evolving dynamically and rapidly (Huang et al. 2002; Anderson et al. 2003; Wicker et al. 2003; Gu et al. 2004). The evolution of these genomes was characterized as the balance between expansion and reduction through transposon and retrotransposon insertions, duplications, and deletions of large fragments. These rapid changes happened in overall gene organization not only in intergenic regions but also in coding regions as shown in this work on glutenin genes. In the Glu-1 loci, the extensive allelic variations are mainly the result of SNPs and indels, probably resulting from unequal crossing over, slip-mismatching, and point mutations. As shown for 1Dx5*t and1Dx5.1*t genes, many SNPs resulted from the C-T transitions because C is readily methylated and deaminated (Razin and Riggs 1980).
Illegitimate recombination was considered a major factor in the evolution of the wheat (Wicker et al. 2003) and Arabidopsis (Devos et al. 2002) genomes. This process generates duplications and deletions and therefore, like unequal homologous recombination, results in genome expansion and contraction. Because illegitimate recombination requires only a few base pairs of sequence identity (Wicker et al. 2003), it is likely to occur within Glu-D1-1 alleles because of a few long direct repeats generally present in the repetitive domain of x-type HMW genes as shown in 1Dx5*t and 1Dx2.2* (Figures 3 and 6). Although fragment deletions within the repetitive domains of glutenin genes in recombinant clones were found (Altenbach 1998; Masci et al. 1998), their molecular origins are still unknown. Our results provide direct evidence for illegitimate recombination in 1Dx5*t within the E. coli heterologous expression system and have led to a large fragment deletion and generation of a small protein. This suggests that duplications and deletions of large fragments within glutenin genes could be produced through illegitimate recombination and indicates a possible molecular mechanism for generating novel allelic variation at Glu-1 loci.
Acknowledgments
We are grateful to Robert McIntosh for constructive suggestions and for reviewing the manuscript. This research was financially supported by grants from the National Natural Science Foundation of China (30571154, 30771334) and the Chinese Ministry of Science and Technology (2002CB111300, 2006AA10Z186).
References
- Alberghina, G., R. Cozzolino, S. Fisichella, D. Garozzo and A. Savarino, 2005. Proteomics of gluten: mapping of the 1Bx7 glutenin subunit in Chinese Spring cultivar by matrix-assisted laser desorption/ionization. Rapid Commun. Mass Spectrom. 19: 2069–2074. [DOI] [PubMed] [Google Scholar]
- Allaby, R. G., and T. A. Brown, 2001. Network analysis provides insights into evolution of 5S rDNA arrays in Triticum and Aegilops. Genetics 157: 1331–1341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allaby, R. G., M. Banerjee and T. A. Brown, 1999. Evolution of the high molecular weight glutenin loci of the A, B, D and G genomes of wheat. Genome 42: 296–307. [PubMed] [Google Scholar]
- Altenbach, S. B., 1998. Quantification of individual low-molecular-weight glutenin subunit transcripts in developing wheat grains by competitive RT-PCR. Theor. Appl. Genet. 97: 413–421. [Google Scholar]
- Anderson, O. D., and F. C. Greene, 1989. The characterization and comparative analysis of HMW glutenin genes from genomes A and B of hexaploid bread wheat. Theor. Appl. Genet. 77: 689–700. [DOI] [PubMed] [Google Scholar]
- Anderson, O. D., F. C. Greene, R. E. Yip, N. G. Halford, P. R. Shewry et al., 1989. Nucleotide sequences of the two high-molecular-weight glutenin genes from the D-genome of a hexaploid bread wheat, Triticum aestivum L. cv Cheyenne. Nucleic Acids Res. 17: 461–462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson, O. D., C. Rausch, O. Moullet and W. S. Lagudah, 2003. The wheat D-genome HMW-glutenin locus: BAC sequencing, gene distribution, and retrotransposon clusters. Funct. Integr. Genomics 3: 56–68. [DOI] [PubMed] [Google Scholar]
- Andrews, J. L., R. L. Hay, J. H. Skerritt and H. Sutton, 1994. HPLC and immunoassay-based glutenin subunit analysis: screening for dough properties in wheats grown under different environmental conditions. J. Cereal Sci. 20: 203–215. [Google Scholar]
- Blatter, R. H. E., S. Jacomet and A. Schlumbaum, 2004. About the origin of European spelt (Triticum spelta L.): allelic differentiation of the HMW glutenin B1-1 and A1-2 subunit genes. Theor. Appl. Genet. 108: 360–367. [DOI] [PubMed] [Google Scholar]
- Caldwell, K. S., J. Dvorak, E. S. Lagudah, E. Akhunov, M. C. Luo et al., 2004. Sequence polymorphism in polyploidy wheat and their D-genome ancestor. Genetics 167: 941–947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cozzolino, R., G. S. Di, S. Fisichella, D. Garozzo, D. Lafiandra et al., 2001. Matrix-assisted laser desorption/ionization mass spectrometric peptide mapping of high molecular weight glutenin subunits 1Bx7 and 1Dy10 in Cheyenne cultivar. Rapid Commun. Mass Spectrom. 15: 778–787. [DOI] [PubMed] [Google Scholar]
- Cunsolo, V., S. Foti, R. Saletti, S. Gilbert, A. S. Tatham et al., 2004. Structural studies of the allelic wheat glutenin subunits 1Bx7 and 1Bx20 by matrix-assisted laser desorption/ionization mass spectrometry and high-performance liquid chromatography/electrospray ionization mass spectrometry. J. Mass Spectrom. 39: 66–78. [DOI] [PubMed] [Google Scholar]
- Devos, K. M., J. K. M. Brown and J. L. Bennetzen, 2002. Genome size reduction through illegitimate recombination counteracts genome expansion in Arabidopsis. Genome Res. 12: 1075–1079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- D'Ovidio, R., D. Lafiandra and E. Porceddu, 1996. Identification and molecular characterization of a large insertion within the repetitive domain of a high-molecular-weight glutenin subunit gene from hexaploid wheat. Theor. Appl. Genet. 93: 1048–1053. [DOI] [PubMed] [Google Scholar]
- Dvorak, J., M. C. Luo, Z. L. Yang and H. B. Zhang, 1998. The structure of the Aegilops tauschii genepool and the evolution of hexaploid wheat. Theor. Appl. Genet. 97: 657–670. [Google Scholar]
- Gianibelli, M. C., M. Echaide, O. R. Larroque, J. M. Carillo and J. Dubcovsky, 2002. Biochemical and molecular characterisation of Glu-1 loci in Argentinian wheat cultivars. Euphytica 128: 61–73. [Google Scholar]
- Giles, R. J., and T. A. Brown, 2006. GluDy allele variations in Aegilops tauchii and Triticum aestivum: implications for the origins of hexaploid wheat. Theor. Appl. Genet. 112: 1563–1572. [DOI] [PubMed] [Google Scholar]
- Goldsbrough, A. P., N. J. Bulleid, R. B. Freedman and R. B. Flavell, 1989. Conformational differences between two wheat (Triticum aestivum) ‘high-molecular-weight’ glutenin subunits are due to a short region containing six amino acid differences. Biochem. J. 263: 837–842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gu, Y. Q., C. D. Devin, X. Y. Kong and O. D. Anderson, 2004. Rapid genome evolution revealed by comparative sequence analysis of orthologous regions from four Triticeae genomes. Plant Physiol. 135: 459–470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hickman, D. R., P. Roepstorff, P. R. Shewry and A. S. Tatham, 1995. Molecular weights of high molecular weight subunits of glutenin determined by mass spectrometry. J. Cereal Sci. 22: 99–103. [Google Scholar]
- Huang, S., A. Sirikhachornkit, X. J. Su, J. Faris, B. Gill et al., 2002. Genes encoding plastid acetyl-CoaA carboxylase and 3-phosphoglycerate kinase of the Triticum/Aegilops complex and evolutionary history of polyploid wheat. Proc. Natl. Acad. Sci. USA 99: 8133–8138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lagudah, E. S., and G. M. Halloran, 1988. Phylogenetic relationships of Triticum tauschii the D genome donor to hexaploid wheat. 1. Variation in HMW subunits of glutenin and gliadin. Theor. Appl. Genet. 75: 592–598. [DOI] [PubMed] [Google Scholar]
- Lelley, T., M. Stachel, H. Grausgruber and J. Vollmann, 2000. Analysis of relationships between Ae. tauschii and the D genome of wheat utilizing microsatellite. Genome 43: 661–668. [PubMed] [Google Scholar]
- Li, W., Y. Wan, Z. Liu, K. Liu, B. Li et al., 2004. Molecular charaterization of HMW glutenin subunit allele 1Bx14: further insights into the evolution of Glu-B1-1 alleles in wheat and related species. Theor. Appl. Genet. 109: 1093–1104. [DOI] [PubMed] [Google Scholar]
- Ma, W., R. Appels, F. Bekes, O. Larroque, M. K. Morell et al., 2005. Genetic characterisation of dough rheological properties in a wheat doubled haploid population: additive genetic effects and epistatic interactions. Theor. Appl. Genet. 111: 410–422. [DOI] [PubMed] [Google Scholar]
- Masci, S., R. D'Ovidio, D. Lafiandra and D. D. Kasarda, 1998. Characterization of a low-molecular-weight glutenin subunit gene from bread wheat and the corresponding protein that represents a major subunit of the glutenin polymers. Plant Physiol. 118: 1147–1158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McFadden, E. S., and E. R. Sears, 1946. a The origin of Triticum spelta and its free-threshing hexaploid relatives. J. Hered. 37: 81–89. [DOI] [PubMed] [Google Scholar]
- McFadden, E. S., and E. R. Sears, 1946. b The origin of Triticum spelta and its free-threshing hexaploid relatives. J. Hered. 37: 107–116. [DOI] [PubMed] [Google Scholar]
- Payne, P. I., 1987. Genetics of wheat storage proteins and the effect of allelic variation on bread-making quality. Annu. Rev. Plant Physiol. 38: 141–153. [Google Scholar]
- Razin, A., and A. D. Riggs, 1980. DNA methylation and gene function. Science 210: 604–610. [DOI] [PubMed] [Google Scholar]
- Shewry, P. R., and N. G. Halford, 2002. Cereal seed storage proteins: structures, properties and role in grain utilization. J. Exp. Bot. 53: 947–958. [DOI] [PubMed] [Google Scholar]
- Shewry, P. R., N. G. Halford and A. S. Tatham, 1992. The high molecular weight subunits of wheat glutenin. J. Cereal Sci. 15: 105–120. [Google Scholar]
- Shewry, P. R., Y. Popineau, D. Lafiandra and P. Belton, 2001. Wheat glutenin subunits and dough elasticity findings of the EUROWHEAT project. Trends Food Sci. Technol. 11: 433–441. [Google Scholar]
- Sugiyama, T., A. Rafalski, D. Peterson and D. Sol, 1985. A wheat HMW glutenin subunit gene reveals a highly repeated structure. Nucleic Acid Res. 13: 8729–8737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun, M. M., Y. M. Yan, Y. Jiang, Y. H. Xiao, Y. K. Hu et al., 2004. Molecular cloning and comparative analysis of a y-type inactive HMW glutenin subunit gene from cultivated emmer wheat (Triticum dicoccum L.). Hereditas 141: 46–54. [DOI] [PubMed] [Google Scholar]
- Sun, X., S. Hu, X. Liu, W. Qian, S. Hao et al., 2006. Characterization of the HMW glutenin subunits from Aegilops searsii and identification of a novel variant HMW glutenin subunit. Theor. Appl. Genet. 113: 631–641. [DOI] [PubMed] [Google Scholar]
- Talbert, L. E., L. Y. Smith and N. K. Blake, 1998. More than one origin of hexaploid wheat is indicated by sequence comparison of low-copy DNA. Genome 41: 402–407. [Google Scholar]
- Tatham, A. S., and P. R. Shewry, 1985. The conformation of wheat gluten proteins. The secondary structures and thermal stabilities of alpha-, beta-, gamma- and omega-gliadins. J. Cereal Sci. 3: 103–113. [Google Scholar]
- Thompson, J. D., D. G. Higgins and T. J. Gibson, 1994. Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 22: 4673–4680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wan, Y., D. Wang, P. R. Shewry and N. G. Halford, 2002. Isolation and characterization of five novel high molecular weight subunit genes from Triticum timopheevi and Aegilops cylindrica. Theor. Appl. Genet. 104: 828–839. [DOI] [PubMed] [Google Scholar]
- Wan, Y., Z. Yan, K. Liu, Y. Zheng, R. D'Ovidio et al., 2005. Comparative analysis of D genome-encoded high-molecular weight subunits of glutenin. Theor. Appl. Genet. 111: 1183–1190. [DOI] [PubMed] [Google Scholar]
- Wicker, T., N. Yahiaoui, R. Guyot, E. Schlagenhauf, Z. D. Liu et al., 2003. Rapid genome divergence at orthologous low molecular weight glutenin loci of A and Am genomes of wheat. Plant Cell 15: 1186–1197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wrigley, C. W., 1996. Giant proteins with flour power. Nature 381: 738–739. [DOI] [PubMed] [Google Scholar]
- Yan, Y., S. L. K. Hsam, J. Z. Yu, Y. Jiang and F. J. Zeller, 2003. a Allelic variation of the HMW glutenin subunits in Aegilops tauschii accessions detected by sodium dodecyl sulphate (SDS-PAGE), acid polyacrylamide gel (A-PAGE) and capillary electrophoresis. Euphytica 130: 377–385. [Google Scholar]
- Yan, Y., S. L. K. Hsam, J. Yu, Y. Jiang, I. Ohtsuka et al., 2003. b HMW and LMW glutenin alleles among putative tetraploid and hexaploid T. spelta progenitors. Theor. Appl. Genet. 107: 1321–1330. [DOI] [PubMed] [Google Scholar]
- Yan, Y., J. Zheng, Y. Xiao, J. Yu, Y. Hu et al., 2004. Identification and molecular characterization of a novel y-type Glu-Dt1 glutenin gene of Aegilops tauschii. Theor. Appl. Genet. 108: 1349–1358. [DOI] [PubMed] [Google Scholar]
- Zhang, Y. Z., Q. Y. Li, Y. M. Yan, J. G. Zheng, X. L. An et al., 2006. Molecular characterization and phylogenetic analysis a novel glutenin gene (Dy10.1t) from Aegilops tauschii. Genome 49: 735–745. [DOI] [PubMed] [Google Scholar]