Abstract
Gap-junctional communication between neurons and astrocytes dissociated from rat brain was identified in culture by using dye-transfer assays and electrophysiological measurements. Cell types were identified by using antibodies against β-tubulin III, glial fibrillary acidic protein, and 2′,3′-cyclic-nucleotide phosphohydrolase, which are antigenic determinants of neurons, astroglia, and oligodendrocytes, respectively. Dye coupling was examined as a function of time after dissociated embryonic brain cells were plated onto confluent monolayers of postnatal astrocytes by intracellularly injecting the fluorochrome Lucifer yellow. Coupling of neurons to the astrocytic monolayer was most frequent between 48 h and 72 h in culture and declined over the next 4 days. This gradual uncoupling was accompanied by progressive neuronal maturation, as indicated by morphological measurements in camera lucida drawings. Dye spread was abolished reversibly by octanol, an agent that blocks gap junction channels in other systems. Double whole-cell voltage-clamp measurements confirmed the presence of heterocellular electrical coupling in these cocultures. Coupling was also seen between neurons and astrocytes in cocultures of cells dissociated from embryonic cerebral hemispheres but was rarely detectable in cocultures of postnatal brain cells. These data strongly suggest that junctional communication may provide metabolic and electrotonic interconnections between neuronal and astrocytic networks at early stages of neural development and that such interactions are weakened as differentiation progresses.
Keywords: glial–neuronal interactions, double whole-cell voltage clamp, Lucifer yellow
The nervous system has great complexity in terms of the diversity of cellular contacts and the broad range of information processing performed by neural cells. Thoroughly debated and overwhelmingly accepted nearly a century ago, the neuron doctrine (1) not only established cellular individuality in the brain parenchyma but also designated neurons as the functional elements in signaling in the central nervous system (CNS). As a consequence, research has focused on elucidating the cellular and molecular details of neuronal pathways, whereas glial cells have been regarded as elements of structural and trophic support, with no direct influence in information processing. However, in the past few years, evidence has accumulated indicating that glial networks may provide functional support to neuronal activity and may constitute dynamic pathways for electrical and chemical signaling in the CNS. Studies showing synchronous metabolic and electrical responses in astrocytes mediated by neuronal–glial interactions demonstrate that, in culture, both slow and rapid calcium waves propagate through the functional syncytium provided by the astrocytic network in response to iontophoretic application of glutamate to astrocytes (2) and to firing of glutamatergic neurons (3, 4). Conversely, the propagation of astrocytic calcium waves to neurons has been more recently reported (5–7). Calcium signaling from astrocytes to neurons in culture was proposed to be mediated by Ca2+-dependent glutamate release from nearby astrocytes and its interaction with neuronal membrane receptors (5, 6). Although not established directly, gap-junction-mediated unidirectional propagation of calcium waves from astrocytes to neurons was also suggested to account for such glial–neuronal signaling in forebrain cocultures (7).
Gap junctions have been recognized in all CNS cell types (8, 9). Such membrane complexes are comprised of hemichannels contributed by each adjacent cell and aligned to form intercellular pores, permeable to inorganic ions and many small metabolites. The abundance of gap junctions in glia, and especially between astrocytes, enables these cells to form a functional syncytium with respect to movement of small ions and molecules (9, 10). In addition to these direct intercellular connections, astrocytes seem to communicate by extracellular messengers (11). Gap-junction proteins comprise the connexin family of more than a dozen members. The relative proportions and expression patterns of connexins change during CNS development (12–14), raising the possibility that cellular compartments distinct in terms of metabolic and electrical cooperation could play a role in the functional sculpting of the brain. The perspective of heterocellular cooperativity at various levels in the CNS (5–7, 15–17) and the lack of direct evidence for electrical and metabolic coupling interlacing neuronal and glial networks have led us to evaluate the existence of functional gap junctions between neurons and astrocytes in primary cultures and its possible correlation to cellular maturation in the CNS.
METHODS
Cell Cultures.
Cocultures of astrocytes and neurons were of two types: “isochronic” cultures, in which neurons and astrocytes were of the same age, coming from either embryonic or postnatal brains, and “heterochronic” cultures, in which neurons were derived from embryonic and astrocytes were derived from postnatal brains, as described (18, 19). In general, cerebral hemispheres were removed from Wistar embryonic (E18–E20) or postnatal (P2) rats, and meninges were excised carefully and discarded. Cells were mechanically dissociated, plated on polyornithine-coated coverslips (1.5 μg/ml; GIBCO/BRL), and distributed in 24-well culture dishes (Nunc) with an initial density of ≈300,000 cells per well. Cultures were grown in DMEM–F12 supplemented with 33 mM glucose, 2 mM glutamine, 3 mM sodium bicarbonate, 100 units/ml penicillin, 0.1 mg/ml streptomycin, 2.5 μg/ml fungizone, and 10% (vol/vol) FCS (37°C, 5% CO2 atmosphere).
Isochronic Cultures.
In isochronic cultures, cells were dissociated from brain hemispheres of E18–E20 (embryonic isochronic cultures) or P2 (postnatal isochronic cultures) rats and kept for 15 days in an incubator with minimal mechanical disturbance during medium changes to avoid neuronal detachment.
Heterochronic Cultures.
In a first step, confluent astrocytic monolayers were obtained at the end of 2 weeks by culturing cells dissociated from P2 cerebral hemispheres. Alternatively, confluence was prevented by culturing astrocytes for up to 1 week before plating embryonic cells (see below). Medium was changed every 2 days, preceded by rinsing with PBS, a procedure that washes away neurons. Cellular purity was evaluated by immunostaining with antibodies to glial fibrillary acidic protein (GFAP), β-tubulin III, and 2′,3′-cyclic-nucleotide phosphohydrolase. In a second step, freshly dissociated embryonic cells (E18–E20) were plated onto astrocytic monolayers at a density of ≈50,000 cells per well. These cultures were grown for up to 1 week; medium was changed 24 h after adding the embryonic cells. Antigen expression, the extent of neuronal arborization, and junctional communication were assayed at different periods in these cultures, as detailed below.
Immunocytochemistry.
Isochronic and heterochronic cultures were fixed with 4% (vol/vol) paraformaldehyde in PBS (pH 7.4; 30 min at room temperature) before blocking [0.1% BSA or 5% (vol/vol) normal rabbit serum, as pertinent] and overnight incubation with primary antibodies (polyclonal anti-GFAP, Dako; monoclonal anti-β-tubulin III, Sigma; monoclonal anti-2′,3′-cyclic-nucleotide phosphohydrolase, Sigma). FITC-conjugated and rhodamine isothiocyanate (RITC)-conjugated secondary antibodies (Sigma) were employed for visualization under an epifluorescence Zeiss microscope. Cell nuclei were stained with bisbenzimide (Sigma). Horseradish peroxidase-labeled secondary antibody (Sigma) was detected by reaction with 0.3% (vol/vol) H2O2 and 0.1% (wt/vol) diaminobenzidine (Sigma) in solution.
Neuronal Morphological Measurements.
For morphological analysis, ≈100 β-tubulin III-positive cells chosen at random from six or seven microscope fields were studied on days 1–4 and 7 in the heterochronic cultures. Neurons were drawn by means of a camera-lucida apparatus attached to an Axioplan Zeiss microscope. Measurements at each time point were taken directly from drawings to evaluate (i) the number of primary processes, i.e., those arising directly from the soma, (ii) the total number of branching points in the neuritic tree, and (iii) neuritic field perimeter, obtained by tracing straight lines connecting outermost neurite endings. Data were compared as described later by means of the Mann–Whitney ranking test, with statistical significance assigned when P < 0.01.
Dye-Coupling Assays.
For dye transfer studies, the small fluorochrome Lucifer yellow CH (LY; Mr = 443; Sigma) was injected into morphologically identified neuronal cells and less frequently into astrocytes, through microiontophoresis, enabling us to visualize, whenever present, junctional coupling during and after microinjections. The apparatus for intracellular injection of fluorescent probes consisted of a microelectrode holder connected to a battery-operated stimulator; 5% (wt/vol) LY in 150 mM LiCl was injected intracellularly by application of brief hyperpolarizing voltage pulses through glass microelectrodes (A-M System, Everett, WA). Coinjections of LY and the gap-junction-impermeant molecule rhodamine B isothiocyanate–dextran (RITC–dextran, Mr ≈ 40,000; Sigma) were also performed to mark the injected cell. In some instances, isochronic and heterochronic cultures assayed for dye coupling were fixed with 4% (vol/vol) paraformaldehyde, allowing later identification of the injected cell type by antibody staining. As a further test for the involvement of gap junctions in intercellular spread of LY, octanol (Sigma) was added to the medium (final concentration of 1 mM). Dye injections were performed in the presence of the alcohol and after successive medium changes to determine whether uncoupling was reversible. All functional experiments, including dye microiontophoresis and electrophysiological assays (below), were carried out at room temperature (≈25°C). At least 80 cells were injected with the fluorochrome in each experimental condition, and the number of cells to which LY had spread was counted at the end of a ≈1-min period after each dye injection. Data were organized in frequency histograms, as detailed below, and compared by means of the χ2 test (statistical significance between data sets was assigned when P < 0.01).
Electrophysiological Assays.
Junctional and nonjunctional currents were recorded by using AXOPATCH1-D amplifiers and analyzed by pclamp software (Axon Instruments, Foster City, CA). Polished-patch-type electrodes were used with resistance of about 5 MΩ. External solution contained (in mM) 165 NaCl, 5 KCl, 2 CaCl2, 10 d-glucose, and 5 Hepes [330 milliosmolar; pH 7.3]; internal solution contained (in mM) 145 KCl, 4.5 NaCl, 1 CaCl2, 1 MgCl2, 11 EGTA, and 5 Hepes (330 milliosmolar; pH 7.3). Neuronal excitability was assessed at various times in culture by means of the conventional whole-cell voltage-clamp technique (20). Records were obtained at potentials that ranged between −80 mV and 80 mV, stepped by 10-mV increments from holding potentials between −80 and −90 mV. Tetrodotoxin (TTX) at 0.1 μM was applied directly to the recording chamber leading to blockade of inward currents and its reversal after washout. Dual whole-cell voltage clamp was employed for electrical-coupling assays (21, 22) in pairs of neurons and astrocytes from both isochronic and heterochronic cultures. Electrotonic spread of current through the well coupled monolayer of astrocytes was minimized by use of subconfluent cocultures, as described above. Small voltage pulses (−10 mV) were alternately applied into a neuron and an astrocyte of each pair and junctional currents were detected as resulting deflections in the current recorded in the nonpulsed cell.
RESULTS
Cellular Phenotypic Characterization in Heterochronic and Isochronic Cultures.
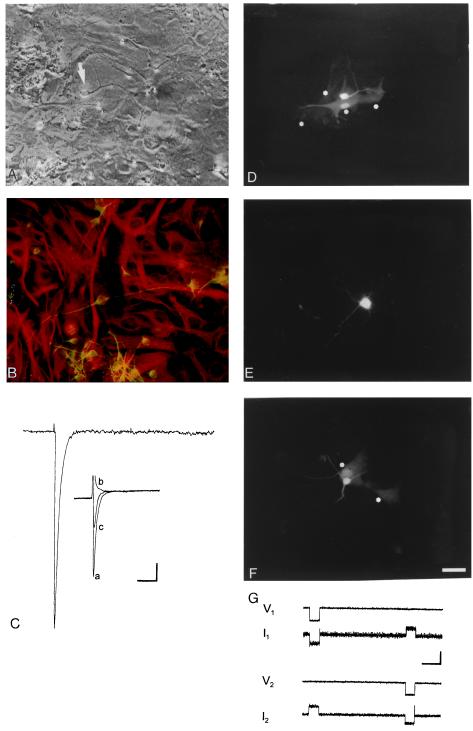
Cellular identity was confirmed in heterochronic and isochronic cultures by double immunocytochemistry to β-tubulin III and GFAP, which are cytoskeletal markers for neurons and astrocytes, respectively (Fig. 1B; see also Fig. 4; refs. 18 and 23). These cultures did not contain significant numbers of other cell types, as judged by immunostaining with the cytoskeletal markers in cultures where nuclei were labeled with bisbenzimide (not shown). Specifically, oligodendrocytes were not present in these cultures, as indicated by the absence of immunoreactivity to the oligodendrocytic marker 2′,3′-cyclic-nucleotide phosphohydrolase (not shown). GFAP-positive cells were large and rather flat, differing markedly from the β-tubulin III-positive cells, which were small, phase bright, and bipolar or multipolar. Immunostaining results thus indicated that astrocytes could be distinguished readily from neurons based on morphological criteria. As an additional test, neuronal excitability was evaluated in morphologically identified neurons; TTX-sensitive inward Na+ currents were present in 21 of 24 recordings of such cells (Fig. 1C).
Figure 1.
Phenotypic characterization and junctional coupling of embryonic neurons and postnatal astrocytes in a heterochronic culture. (A) Phase-contrast micrograph of a 72-h coculture of embryonic neurons and postnatal astrocytes; the arrow points to the LY-injected neuron, shown in the fluorescence micrograph in D. (B) Double immunostaining of a 72-h coculture with anti-β-tubulin III (green-yellow fluorescence due to FITC-conjugated secondary antibody) and anti-GFAP (red fluorescence due to RITC secondary). (C) Inward current recorded in a neuronal cell activated by a depolarizing pulse of 80 mV (capacitive transient and leakage currents were electronically subtracted from original trace). (Insert) a, control; b, after 0.1 μM TTX was added to the bath; c, after washing out TTX—inward current partially recovered (original records—not compensated). [Bars: horizontal = 5 ms (main trace) and 10 ms (Insert); vertical = 0.3 nA (main trace) and 1 nA (Insert)]. Outward K+ currents were not discernible in the range of potentials tested. (D) LY transfer between the neuron (arrow in A) and four neighboring astrocytes, two of which are barely seen (indicated by asterisks). (E) Absence of dye coupling from neuron to astrocytes after a 10-min exposure to 1 mM octanol. (F) Recovery of dye coupling in the same dish after repeated rinses to remove octanol, exemplified between an injected neuron and at least two astrocytes (asterisks). (Magnification = ×400; bar = 20 μm.) (G) Macroscopic junctional currents recorded in a heterotypic cell pair at 72 h; voltage pulses, V1 and V2, were applied alternately to a neuron and an astrocyte, respectively, and junctional currents were recognized as upward current steps in I1 in response to the V2 step and in I2 in response to V1. Junctional conductance (I2/V1 or I1/V2) in this example is about 5 nS. [Horizontal bar = 1 s; vertical bar = 10 mV (V1 and V2) or 50 pA (I1 and I2].
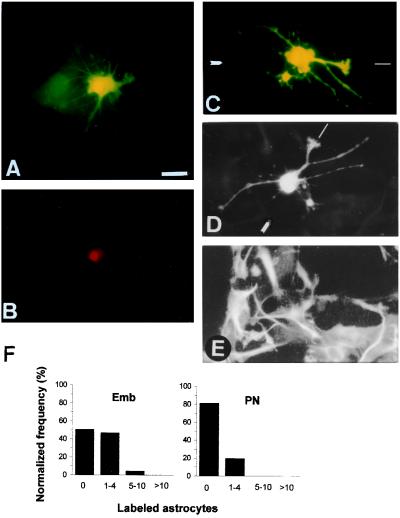
Figure 4.
Dye coupling between antigenically identified neurons and astrocytes in embryonic and postnatal isochronic cultures. Dye coupling between neuron and surrounding astrocytes in embryonic isochronic culture coinjected with LY (A) and RITC–dextran (B), which is confined to the injected cell. (C) Neuron injected with LY in postnatal isochronic culture. (D and E) Same field after staining with antibodies to β-tubulin III and GFAP, respectively. Orientation marks relative to C are presented in D. (F) Frequency histograms of extent of dye coupling in embryonic (Emb, Left) and postnatal (PN, Right) isochronic cultures. Abscissa as in Fig. 2. (Magnification = ×400; bar = 20 μm.)
Gap-Junctional Coupling Between Neurons and Astrocytes in Heterochronic Cultures.
Embryonic neurons, identified by using either morphological or morphological/immunostaining criteria, were very frequently dye coupled to the astrocytic monolayer in the first 3 days after setting up the heterochronic cultures, as exemplified in Fig. 1D. Coupling between neurons was not detectable in our culture conditions. When coinjected with LY, the large gap-junction impermeant fluorescent conjugate RITC–dextran (Mr ≈ 40,000) was restricted to the impaled cell in 96% of the microinjections (139 RITC–dextran single cells staining of 144 coinjections in two heterochronic cultures). LY coupling was reversibly abolished by 1 mM octanol, a blocker of junctional communication (Fig. 1 E and F; refs. 24 and 25). A total of 122 microinjections was done in two different heterochronic cultures, at 24 h and 48 h after plating neurons. Before adding octanol, detectable coupling was found in 57% of the dye injections; the incidence of coupling was reduced to 5% after a 10-min exposure to octanol and partially recovered (36% of the injections showing dye spread) after rinsing the cultures with normal medium. As a further proof of gap-junction-mediated heterocellular communication in these cultures, we recorded electrical coupling in pairs of neurons and astrocytes by using the dual whole-cell voltage-clamp technique (21, 22). In these experiments, identification of neurons was confirmed further by the presence of inward Na+ currents (Fig. 1C). As illustrated in Fig. 1G, bidirectional junctional currents were recorded from pairs of neurons (I1) and astrocytes (I2; five cases of coupling in 19 pairs in heterochronic cultures between 24 h and 96 h). Although bidirectional electrical coupling predicts bidirectional dye coupling, spread of LY from confluent astrocytes to neurons was rarely observed (in 3 of 252 microinjections into astrocytes) in typical heterochronic cultures. In contrast, when performed in heterochronic cultures that were set up before astrocytes had reached confluence, ≈15% of dye injections into astrocytes resulted in spread of LY to an adjacent neuron, confirming the existence of bidirectional communication between these two cell types and suggesting that dye coupling from astrocytes to neurons in confluent cultures may be, at least partially, undetectable by dilution of the fluorochrome through the well coupled astrocytic monolayer. Also evident is the lower incidence of electrical coupling in subconfluent heterochronic cultures (up to 26%, evaluated between 24 h and 96 h) compared with incidence of dye coupling in confluent ones (up to 83%, in the same period). Such contrast may be ascribed to differences in astrocytic maturation (astrocytes were ≈1 week younger in subconfluent cultures) or simply to different cell densities; whereas several cells are available for functional dye coupling to neurons in confluent astrocytic monolayers, only one potential partner is available for electrical coupling in recordings from cell pairs.
Neuronal Morphological Differentiation Parallels Loss of Dye Coupling Between Neurons and Astrocytes in Heterochronic Cultures.
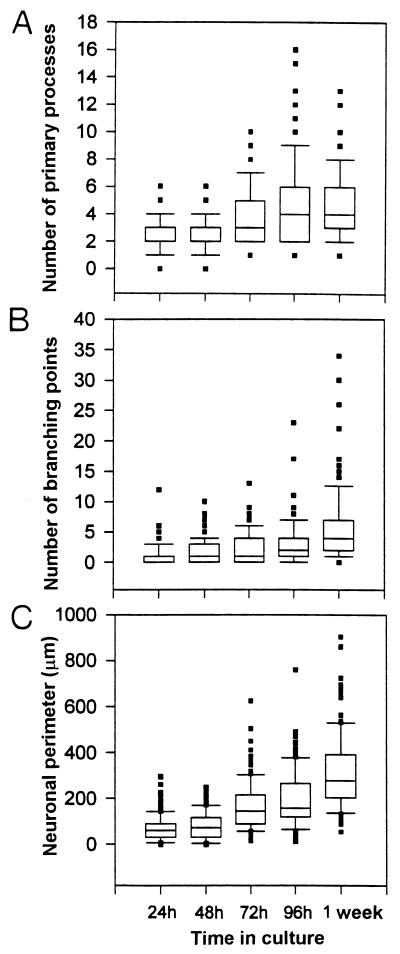
Enriched in astrocytes and virtually devoid of other glial cell types, cocultures of acutely dissociated neural cells on previously established astrocytic monolayers allowed us to investigate a possible correlation between neuronal maturation in vitro and the phenomenon of heterocellular coupling that we observed during the first days of heterochronic culture, as described above. For this investigation, we performed neuronal morphological measurements as well as dye-coupling assays at 24-h intervals up to 96 h and then 1 week after adding neural cells to the astrocyte monolayer. Neuronal arborization was indicated by immunostaining with peroxidase-conjugated secondary antibodies reacted against β-tubulin III monoclonal antibodies (Fig. 2 A–E). Camera-lucida drawings of about 100 neurons at each time point (546 neurons in total) were used for measurements of the number of primary processes, the number of branching points, and the neuritic field perimeter (Fig. 3 A–C). Increases in each of these parameters with time in heterochronic cultures are in accordance with previous reports of progressive neurite elongation, branching, and morphological differentiation in other culture types (26, 27). Data derived from morphological measurements were compared by means of the nonparametric Mann–Whitney ranking test in successive pairs, corresponding to the periods studied. Results indicated no significant differences between 24 h and 48 h or between 72 h and 96 h, whereas all the remaining comparisons indicated significant differences (P < 0.01).
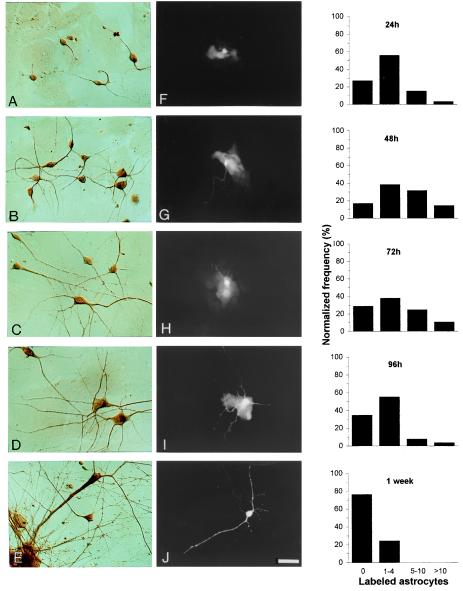
Figure 2.
Neuronal morphological differentiation parallels loss of dye coupling between embryonic neurons and postnatal astrocytes in heterochronic cultures. (A–E) Extent of neuronal arborization as indicated by peroxidase immunoreactivity to anti-β-tubulin III at 24 h (A), 48 h (B), 72 h (C), 96 h (D), and 1 week (E). (F–J) Examples of LY spread into neurons injected at each of the time points indicated for the Left column and the spread from these neurons to neighboring astrocytes, when coupled. In these examples, dye spread to 4 astrocytes at 24 h (F), ≈10 cells at 48 h (G), at least 4 cells at 72 h (H), 1 astrocyte at 96 h (I), and to no astrocyte at 1 week (J). (Right) Frequency distributions of dye coupling in heterochronic cultures at each time point; abscissa indicates the number of astrocytes in which LY was detected at ≈1 min after injection into a neuron. [Magnification = ×400; bars = 20 μm (A–E) and 40 μm (F–J).]
Figure 3.
Multiple box-whisker plots relative to distributions of the three different morphological parameters measured in neurons kept from 24 h to 1 week in heterochronic culture. The number of primary processes (A), the number of branching points (B), and the neuritic field perimeter (C) were presented as non-Gaussian distributions. Data were compared between time points by the Mann–Whitney ranking test, as described in Results.
Dye injections were performed at the same time intervals (total of 569 injections), and representative samples are shown in Fig. 2 F–J. For each LY injection into a neuron, a variable number of astrocytes were labeled, ranging from a lack of dye coupling, in which LY was restricted to the injected neuron, to relatively extensive coupling, when the fluorescent probe was spread to more than 10 astrocytes within about 1 min. Data were then distributed into four different categories of dye-coupling extent [i.e.; (i) cases with no spread to astrocytes, (ii) spread to 1–4, (iii) to 5–10, or (iv) to >10 astrocytes] and plotted as frequency histograms, each corresponding to the different time points assayed after adding neural cells to the astrocytic monolayer (Fig. 2 Right). By pairing these distributions sequentially and applying the χ2 test, changes in the frequency of heterocellular coupling were confirmed. Distributions of dye coupling at 48 h and 72 h presented the highest incidence (i.e., percentage of coupling events) and extent of coupling, and were not statistically different. At 96 h, however, heterocellular dye coupling was less frequent, and after 1 week, neurons and astrocytes were rarely and poorly coupled (P < 0.01). As is evident from Fig. 2, this gradual uncoupling, observed in LY microiontophoresis assays, parallels an enhancement of neuronal morphological complexity in heterochronic cultures. Although maturation of astrocytes was not assayed, we cannot discard its contribution to the reduction of heterocellular coupling observed. Nevertheless, the possibility that this uncoupling was merely reflecting a decrease in junctional communication between elements of the astrocytic syncytium was discarded, because LY injections performed into astrocytes at the same time intervals indicated constant incidence and increasing extent of homocellular coupling in the astrocytic monolayer (252 microinjections; data not shown).
Coupling Between Neurons and Astrocytes in Embryonic and Postnatal Isochronic Cultures.
Previous studies have established a possible involvement of gap junctions in the unidirectional spread of calcium waves from astrocytes to neurons in rat forebrain embryonic cultures (7). Using an analogous primary culture assay, herein named embryonic isochronic culture, we decided to investigate the existence of functional gap junctions between neurons and astrocytes by tracer-coupling measurements. Isochronic cultures from embryonic (E18–E20) brains were assayed for dye spread, and significant levels of heterocellular coupling were detectable, as illustrated in Fig. 4 A and F (80 microinjections, from which 26 were coinjections with LY/RITC–dextran with no detectable spread of the large conjugate to cells other than the injected neuron; Fig. 4B). Data obtained with embryonic isochronic cultures were then compared with those obtained with analogous postnatal cultures. In vitro conditions were the same for both embryonic and postnatal isochronic cultures. In contrast to embryonic cultures, neurons and astrocytes in postnatal (P2) isochronic cultures were predominantly not coupled (Fig. 4C), as judged by dye assays summarized in the corresponding frequency histogram (Fig. 4F; 18% incidence of coupling; 88 microinjections) and double whole-cell voltage-clamp recordings (no coupling in 16 neuron–astrocyte pairs). As pointed out above, antigenicity to β-tubulin III and GFAP were confirmed in these cultures (Fig. 4 D and E), as was neuronal excitability, evaluated by the presence of TTX-sensitive inward Na+ currents (not shown).
DISCUSSION
Our data show that functional intercellular junctional channels connect neurons directly to astrocytes in vitro, defining a pathway for bidirectional electrical and metabolic signaling. The question arises as to whether gap junctions couple the neuronal and the glial syncytia in the intact developing brain. Neurons and astrocytes are the two prevalent cell types in the nervous tissue, especially in the gray matter, and form intricate cellular arrays that dominate the brain parenchyma beginning at early stages of the CNS ontogenesis. Prenatal GFAP expression can be attributed to at least two types of cells, both astrocyte precursors. One is the radial glial cell present very early, even before neurogenesis begins. A second one is presumably represented by a nonradial astrocyte precursor that migrates outward from the germinal layers, populates the cerebral wall, and seems to account for in situ generation of many astrocytes. At this stage, mature astrocytes may be present, although these are detected mostly after birth (28, 29). Thus, neurons dissociated from cerebral hemispheres between E18 and E20 may contact astrocytes in situ at several stages of gliogenesis, and our data from in vitro studies indicate one of the multiple ways by which these cells might cross-interact. Our results show that embryonic neurons are frequently coupled to monolayered astrocytes during the first days in heterochronic cultures. These astrocytes are expected to be represented mainly by postnatal ones, plated 2 weeks before adding embryonic cells and left to grow to confluence. Nevertheless, we cannot eliminate the possibility of occasional interactions between neurons and young astrocytes that contribute to the pool of added embryonic cells. Moreover, our data show that embryonic neurons after 1 week in heterochronic cultures are rarely coupled to astrocytes. Using morphological indicators of neuronal maturation, we have established an inverse correlation between the frequency of junctional communication connecting neurons and astrocytes and the extent of neuronal maturation in these cultures. It is well established that neuronal morphological differentiation is accompanied by maturation of chemical synapses, as observed in both dissociated and organotypic primary CNS cultures (30–35). In addition, traditionally, it has been suggested that neuronal differentiation and, more specifically, final maturation of chemical synapses, follow the silencing of electrical ones (36, 37). As chemical synaptic transmission remains to be evaluated in these cocultures, we cannot discard the possibility that some overlap in time exists between the onset of classical ligand-mediated intercellular signaling and the neuron–astrocyte junctional communication described herein.
Although dissection of the cellular pathways that determine LY spreading from neurons to nearby astrocytes was not feasible in these studies, there are two possible extremes: one neuron coupled to many astrocytes and one neuron coupled to one astrocyte, which in turn couples to adjacent astrocytes. Whatever is the preferential route for dye spreading and electrical coupling between neurons and astrocytes, the time profile of heterocellular junctional communication described for heterochronic cultures probably reflects a modulation of gap junctions that directly connect neurons to astrocytes. Supporting this possibility is the evidence that junctional coupling in the astrocyte monolayer does not decay with time in culture.
Although bidirectional, the evidence of heterocellular junctional coupling in embryonic isochronic cultures reinforces Nedergaard’s (7) previous suggestions for the existence of functional gap junctions mediating calcium signaling from astrocytes to neurons in similar culture conditions. Also, we have compared heterocellular coupling measured in embryonic cultures with data obtained from similar postnatal cultures. After 2 weeks in isochronic cultures, neurons and astrocytes from postnatal brains were virtually uncoupled, whereas, in E18–E20 isochronic cultures, these cells were significantly dye coupled. Neurons and astrocytes are probably less mature in the embryonic than in the postnatal brain tissues, and the difference may be maintained during the 2-week period in vitro. Therefore, the lower incidence of heterocellular coupling detected in postnatal isochronic cultures, relative to embryonic ones, indicates the possibility of an inverse correlation between this communication and in situ cellular maturation, as observed with in vitro neuronal maturation in heterochronic cultures.
Heterocellular coupling between elements of neuroglia has been shown for astrocytes with oligodendrocytes and Müller cells (15–17, 38). Coupling between astrocytes and oligodendrocytes has been suggested to increase with time in coculture (17), paralleling the increased coupling over this period seen between oligodendrocytes (39). Our results identify an additional form of heterocellular coupling that interconnects astrocytes to neurons. Although we have not evaluated whether neurons can couple to oligodendrocytes in appropriate cocultures, the inverse relationship found here between neuronal maturation and the frequency of astrocyte–neuronal coupling occurred at developmental stages that precede establishment of coupling between astrocytes and oligodendrocytes (17). It seems likely that functional cooperation mediated by gap junctions between neuronal and astrocytic networks would be restricted to early periods of brain histogenesis.
In recent years, a considerable effort has been made to elucidate the mechanisms that underlie the intercellular propagation of calcium waves in the CNS, as a way to identify additional means of intercellular signaling and their possible consequences for brain physiology. Calcium waves have been shown to propagate between astrocytes (10), at least in some cases mediated by an extracellular signal (11), and also from these cells to neurons in culture (5–7). Communication between neurons and astrocytes (7) and elevation of neuronal calcium levels evoked by glutamate released from adjacent astrocytes (5) have been postulated as routes for calcium-wave propagation from astrocytes to neurons. By a combination of dye-injection assays and electrophysiological records in neuron/astrocyte cocultures, we have established bidirectional junctional coupling, thus substantiating gap-junctional communication as a viable route for the spread of second messengers between neurons and astrocytes, at least in culture.
Recently, we have shown that neurons are able to induce differentiation of young astrocytes (40). Our in vitro manipulations suggest that coupling between neurons and astrocytes is a transient phenomenon, as it correlates inversely to cellular maturation. This inverse relationship favors the hypothesis that coupling would involve young neurons and astrocytes at early periods of brain histogenesis, and this coupling may be one of the possible ways by which neurons could induce astrocytic maturation. Certainly, a direct pathway for the exchange of nutrients and cell messengers between neuronal and glial cell types is an appealing mechanism for metabolic cooperativity and excitability modulation in the developing nervous tissue. Furthermore, it includes astrocytes as potential transient mediators of local excitability, a circuit previously presumed to be built exclusively by neurons. The establishment of direct low-resistance pathways for electrical and metabolic interplay between neurons and astrocytes could be a necessary early step for cell-specific recognition and differentiation in the CNS. Additional studies are required to establish the functional significance and the developmental regulation of such direct pathways for intercellular signaling in brain slices. These experiments may prove fundamental in unveiling the cellular and molecular rules that govern CNS ontogenesis.
Acknowledgments
We thank Dr. Michael Bennett for communicating this manuscript and especially for his complete and enriching review, Drs. J. Menezes, R. Reis, J. H. Nascimento, M. Mallat, H. Chneiweiss, and C. Giaume for valuable comments and suggestions, and Dr. R. Raggio Luiz for statistical assistance. This work was supported by grants from Conselho Nacional de Pesquisas (Brazil), Coordenaçao de Aperfeiçoamento de Pessoal de Nível Superior-Comité Français d’Évaluation de la Coopération Universitaire avec le Brésil, Programa Avançado de Neurociências—Universidade Federal do Rio de Janeiro, Conselho de Ensino para Graduados—Universidade Federal do Rio de Janeiro, Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro, Financiadora de Estudos e Projetos, and Programa de Núcleos de Excelência-052/97-Ministério da Ciência e Technologia to A.C.C.C. and V.M.N., and National Institutes of Health Grants NS10428 and NS34931 to D.C.S.
ABBREVIATIONS
- CNS
central nervous system
- En
embryonic day n
- Pn
postnatal day n
- LY
Lucifer yellow
- GFAP
glial fibrillary acidic protein
- RITC
rhodamine isothiocyanate
- TTX
tetrodotoxin
References
- 1.Ramón y Cajal S. Neuron Theory or Reticular Theory. 1935. trans. Purkiss, M. U. & Fox, C. A. (1954) (Consejo Superior de Investigaciones Científicas, Inst. Ramón y Cajal, Madrid). [Google Scholar]
- 2.Cornell-Bell A H, Finkbeiner S M, Cooper M S, Smith S J. Science. 1990;247:470–473. doi: 10.1126/science.1967852. [DOI] [PubMed] [Google Scholar]
- 3.Dani J W, Chernjavsky A, Smith S J. Neuron. 1992;8:429–440. doi: 10.1016/0896-6273(92)90271-e. [DOI] [PubMed] [Google Scholar]
- 4.Murphy T H, Blatter L A, Wier W G, Baraban J M. J Neurosci. 1993;13:2672–2679. doi: 10.1523/JNEUROSCI.13-06-02672.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Parpura V, Basarsky T A, Liu F, Jeftinija F, Jeftinija K, Haydon P G. Nature (London) 1994;369:744–747. doi: 10.1038/369744a0. [DOI] [PubMed] [Google Scholar]
- 6.Hassinger T D, Atkinson P B, Strecker G J, Whalen L R, Dudek F E, Kossel A H, Kater S B. J Neurobiol. 1995;28:159–170. doi: 10.1002/neu.480280204. [DOI] [PubMed] [Google Scholar]
- 7.Nedergaard M. Science. 1994;263:1768–1771. doi: 10.1126/science.8134839. [DOI] [PubMed] [Google Scholar]
- 8.Dermietzel R, Spray D C. Trends Neurosci. 1993;16:186–192. doi: 10.1016/0166-2236(93)90151-b. [DOI] [PubMed] [Google Scholar]
- 9.Spray D C. In: Gap Junctions in the Nervous System. Spray D C, Dermietzel R, editors. Georgetown, TX: Landes; 1996. pp. 1–11. [Google Scholar]
- 10.Giaume C, McCarthy K D. Trends Neurosci. 1996;19:319–325. doi: 10.1016/0166-2236(96)10046-1. [DOI] [PubMed] [Google Scholar]
- 11.Hassinger T D, Guthrie P B, Atkinson P B, Bennett M V, Kater S B. Proc Natl Acad Sci USA. 1996;93:13268–13273. doi: 10.1073/pnas.93.23.13268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dermietzel R, Traub O, Hwang T K, Beyer E, Bennett M V, Spray D C, Willecke K. Proc Natl Acad Sci USA. 1989;66:10148–10152. doi: 10.1073/pnas.86.24.10148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Willecke K, Hennemann H, Dahl E, Jungbluth S, Heynkes R. Eur J Cell Biol. 1991;56:1–7. [PubMed] [Google Scholar]
- 14.Peinado A, Yuste R, Katz L C. Neuron. 1993;10:103–114. doi: 10.1016/0896-6273(93)90246-n. [DOI] [PubMed] [Google Scholar]
- 15.Ransom B R, Kettenmann H. Glia. 1990;3:258–266. doi: 10.1002/glia.440030405. [DOI] [PubMed] [Google Scholar]
- 16.Robinson S R, Hampson E C G M, Munro M N, Vaney D I. Science. 1993;262:1072–1074. doi: 10.1126/science.8093125. [DOI] [PubMed] [Google Scholar]
- 17.Venance L, Cordier J, Monge M, Zalc B, Glowinski J, Giaume C. Eur J Neurosci. 1995;7:451–461. doi: 10.1111/j.1460-9568.1995.tb00341.x. [DOI] [PubMed] [Google Scholar]
- 18.Garcia-Abreu J, Cavalcante L A, Moura Neto V. NeuroReport. 1995;6:761–764. doi: 10.1097/00001756-199503270-00014. [DOI] [PubMed] [Google Scholar]
- 19.Moura Neto V, Mallat M, Jeantet C, Prochiantz A. EMBO J. 1983;2:1243–1248. doi: 10.1002/j.1460-2075.1983.tb01576.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hamill O P, Marty A, Neher E, Sakmann B, Sigworth F J. Pflügers Arch. 1981;391:85–100. doi: 10.1007/BF00656997. [DOI] [PubMed] [Google Scholar]
- 21.White R L, Spray D C, Campos de Carvalho A C, Wittenberg B A, Bennett M V L. Am J Physiol. 1985;24:C447–C445. doi: 10.1152/ajpcell.1985.249.5.C447. [DOI] [PubMed] [Google Scholar]
- 22.Neyton J, Trautmann A. Nature (London) 1985;317:331–335. doi: 10.1038/317331a0. [DOI] [PubMed] [Google Scholar]
- 23.Ferreira A, Caceres A. J Neurosci Res. 1992;32:516–529. doi: 10.1002/jnr.490320407. [DOI] [PubMed] [Google Scholar]
- 24.Johnston M F, Simon S A, Ramon F. Nature (London) 1980;286:498–500. doi: 10.1038/286498a0. [DOI] [PubMed] [Google Scholar]
- 25.Dermietzel R, Hertzberg E L, Kessler J A, Spray D C. J Neurosci. 1991;11:1421–1432. doi: 10.1523/JNEUROSCI.11-05-01421.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Banker G A. Science. 1980;209:809–810. doi: 10.1126/science.7403847. [DOI] [PubMed] [Google Scholar]
- 27.Bartlett W P, Banker G A. J Neurosci. 1984;4:1944–1953. doi: 10.1523/JNEUROSCI.04-08-01944.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Skoff R P, Knapp P A. In: Neuroglia. Kettenmann H, Ransom B R, editors. London: Oxford Univ. Press; 1995. pp. 135–148. [Google Scholar]
- 29.Fedoroff, S. (1986)in Astrocytes, eds. Fedoroff, S. & Vernadakis, A. (Academic, London), pp. 35–67.
- 30.van Huizen F, Romijn H J, Habets A M. Brain Res. 1985;351:67–80. doi: 10.1016/0165-3806(85)90232-9. [DOI] [PubMed] [Google Scholar]
- 31.Kim S U, Osborne D N, Kim M W, Spiegelman I, Puil E, Shin D H, Eisen A. Neuroscience. 1988;25:659–670. doi: 10.1016/0306-4522(88)90266-7. [DOI] [PubMed] [Google Scholar]
- 32.Legendre P, Tixier-Vidal A, Brigant J L, Vincent J D. Brain Res. 1988;471:273–285. doi: 10.1016/0165-3806(88)90105-8. [DOI] [PubMed] [Google Scholar]
- 33.Kraszewski K, Grantyn R. J Neurobiol. 1992;23:766–781. doi: 10.1002/neu.480230613. [DOI] [PubMed] [Google Scholar]
- 34.Gottmann K, Pfrieger F W, Lux H D. Brain Res Dev Brain Res. 1994;81:77–88. doi: 10.1016/0165-3806(94)90070-1. [DOI] [PubMed] [Google Scholar]
- 35.Basarsky T A, Parpura V, Haydon P G. J Neurosci. 1994;14:6402–6411. doi: 10.1523/JNEUROSCI.14-11-06402.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Fischbach G D. Dev Biol. 1972;28:407–429. doi: 10.1016/0012-1606(72)90023-1. [DOI] [PubMed] [Google Scholar]
- 37.Rozental R, Mehler M F, Morales M, Andrade-Rozental A F, Kessler J A, Spray D C. Dev Biol. 1995;167:350–362. doi: 10.1006/dbio.1995.1029. [DOI] [PubMed] [Google Scholar]
- 38.Zahs K R, Newman E A. Glia. 1997;20:10–22. [PubMed] [Google Scholar]
- 39.Kettenmann H, Ransom B R. Glia. 1988;1:64–73. doi: 10.1002/glia.440010108. [DOI] [PubMed] [Google Scholar]
- 40.Gomes F A C, Garcia-Abreu J, Galou M, Paulin D, Moura Neto V. Glia. 1999;26:97–108. doi: 10.1002/(sici)1098-1136(199904)26:2<97::aid-glia1>3.0.co;2-z. [DOI] [PubMed] [Google Scholar]