Abstract
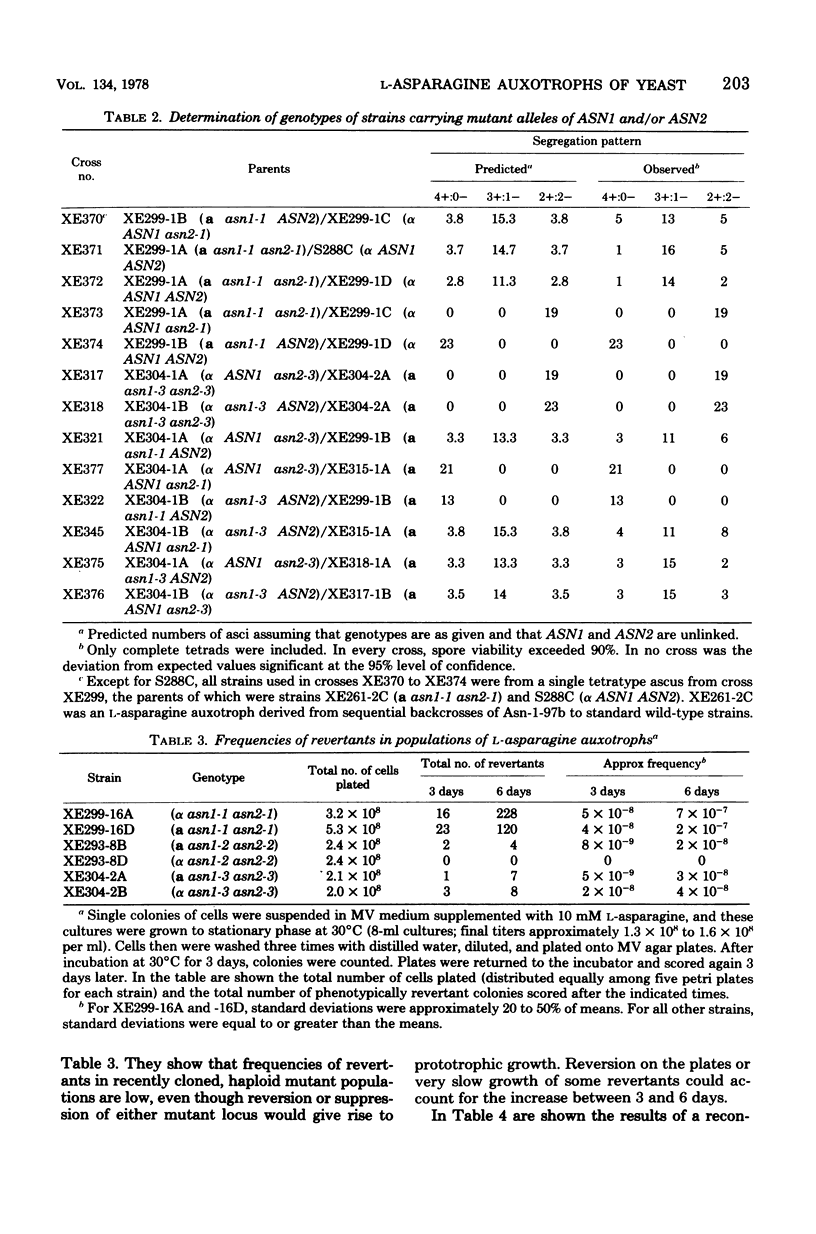
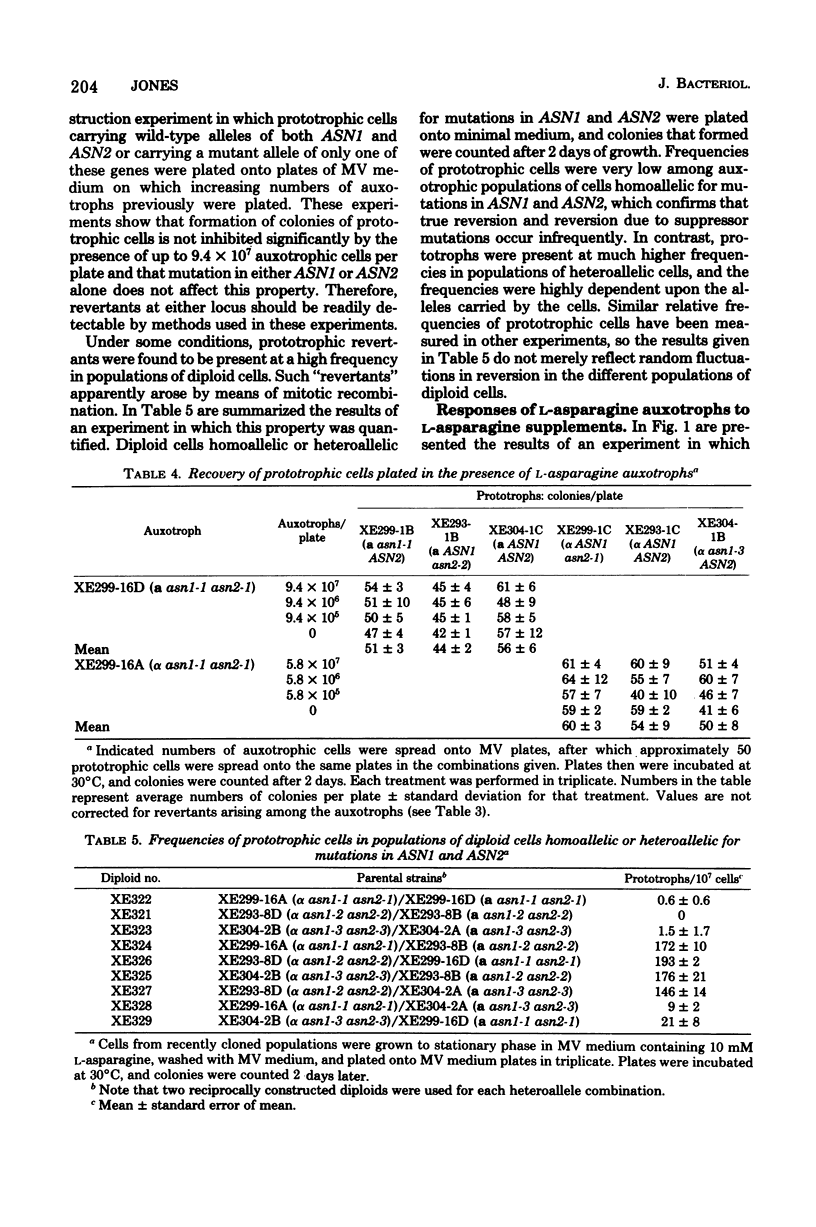
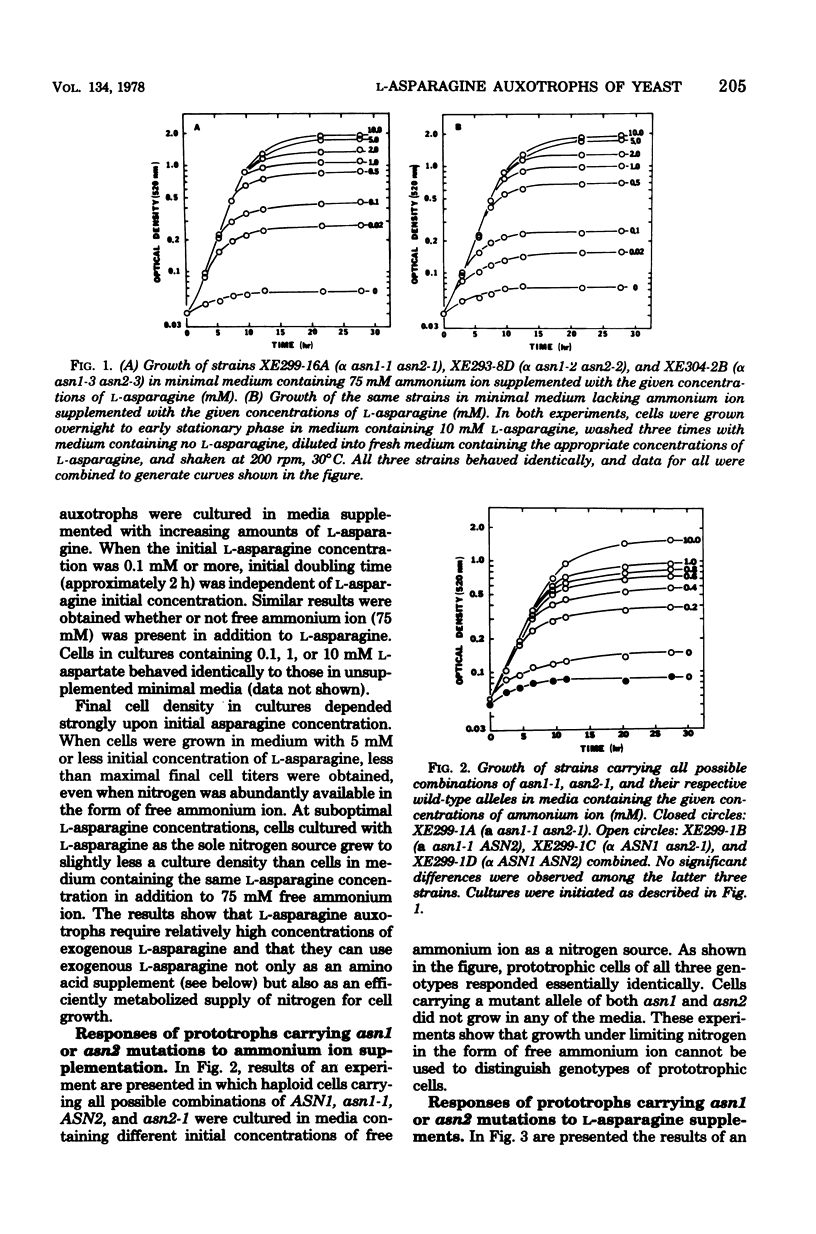
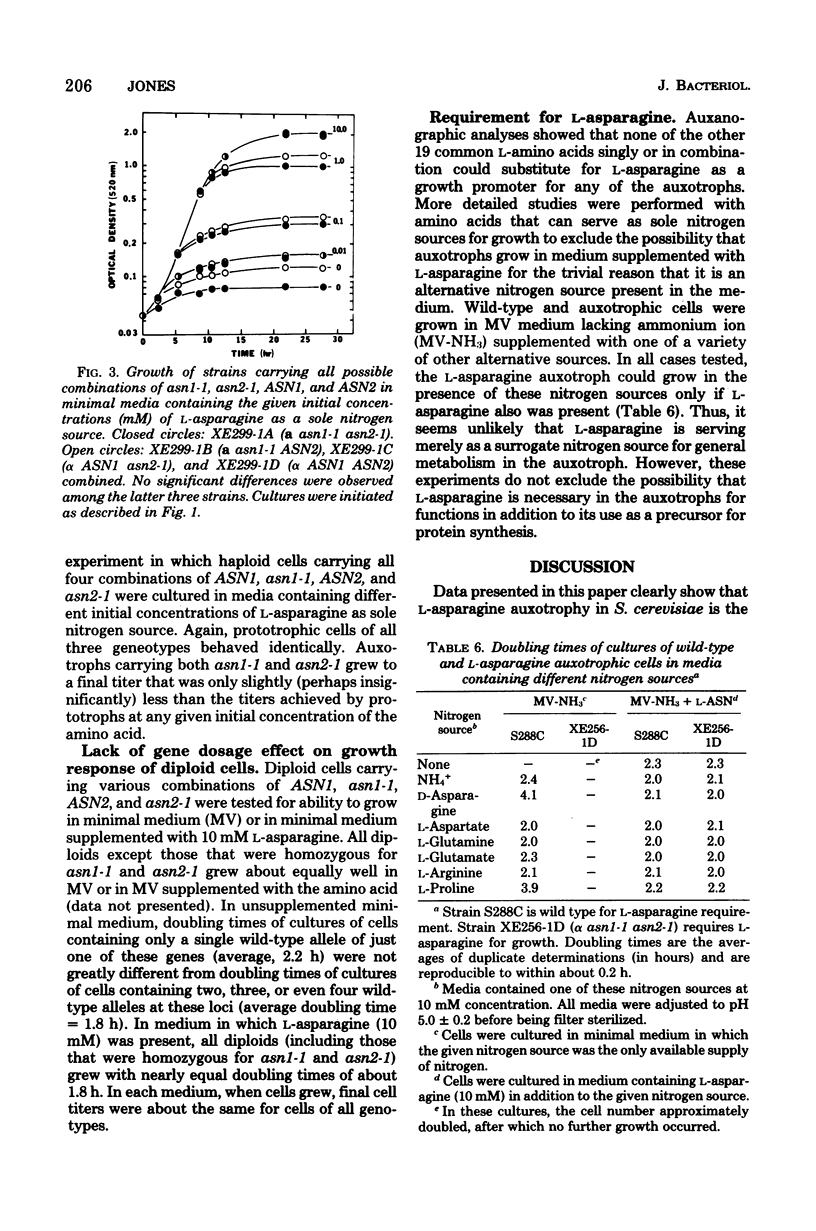
L-Asparagine auxotrophy in Saccharomyces cerevisiae is the result of mutation in each of two unlinked cistrons, ASN1 and ASN2. Mutation in only one of these cistrons yields growth indistinguishable from that of wild-type cells under a variety of nutritional stresses. Relatively high concentrations of L-asparagine are required to permit maximal growth of the auxotrophs, and the amino acid requirement cannot be satisfied by a variety of other amino acids that serve as nitrogen sources for cell growth. Although reversion of the mutations can occur, haploid populations of cells containing only low frequencies of prototrophs can be maintained easily. In diploid cells heteroallelic for certain combinations of alleles of the two genes, mitotic recombination gives rise to prototrophic cells that accumulate to high frequency in populations of the cells.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Atkins C. A., Pate J. S., Sharkey P. J. Asparagine metabolism-key to the nitrogen nutrition of developing legume seeds. Plant Physiol. 1975 Dec;56(6):807–812. doi: 10.1104/pp.56.6.807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cedar H., Schwartz J. H. The asparagine synthetase of Escherhic coli. I. Biosynthetic role of the enzyme, purification, and characterization of the reaction products. J Biol Chem. 1969 Aug 10;244(15):4112–4121. [PubMed] [Google Scholar]
- Dunlop P. C., Roon R. J., Even H. L. Utilization of D-asparagine by Saccharomyces cerevisiae. J Bacteriol. 1976 Mar;125(3):999–1004. doi: 10.1128/jb.125.3.999-1004.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dunlop P. C., Roon R. J. L-Asparaginase of Saccharomyces cerevisiae: an extracellular Enzyme. J Bacteriol. 1975 Jun;122(3):1017–1024. doi: 10.1128/jb.122.3.1017-1024.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldfarb P. S., Carritt B., Hooper M. L., Slack C. The isolation and characterization of asparagine-requiring mutants of Chinese hamster cells. Exp Cell Res. 1977 Feb;104(2):357–367. doi: 10.1016/0014-4827(77)90101-x. [DOI] [PubMed] [Google Scholar]
- Henry S. A., Donahue T. F., Culbertson M. R. Selection of spontaneous mutants by inositol starvation in yeast. Mol Gen Genet. 1975 Dec 30;143(1):5–11. doi: 10.1007/BF00269415. [DOI] [PubMed] [Google Scholar]
- Jones G. E. A fine-structure map of the yeast l-asparaginase gene. Mol Gen Genet. 1973;121(1):9–14. doi: 10.1007/BF00353689. [DOI] [PubMed] [Google Scholar]
- Jones G. E. Genetic and physiological relationships between L-asparaginase I and asparaginase II in Saccharomyces cerevisiae. J Bacteriol. 1977 Apr;130(1):128–130. doi: 10.1128/jb.130.1.128-130.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones G. E. Genetics of expression of asparaginase II activity in Saccharomyces cerevisiae. J Bacteriol. 1977 Feb;129(2):1165–1167. doi: 10.1128/jb.129.2.1165-1167.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones G. E., Mortimer R. K. Biochemical properties of yeast L-asparaginase. Biochem Genet. 1973 Jun;9(2):131–146. doi: 10.1007/BF00487443. [DOI] [PubMed] [Google Scholar]
- Jones G. E., Mortimer R. K. L-asparaginase-deficient mutants of yeast. Science. 1970 Jan 9;167(3915):181–182. doi: 10.1126/science.167.3915.181. [DOI] [PubMed] [Google Scholar]
- Wriston J. C., Jr, Yellin T. O. L-asparaginase: a review. Adv Enzymol Relat Areas Mol Biol. 1973;39:185–248. doi: 10.1002/9780470122846.ch3. [DOI] [PubMed] [Google Scholar]


