Abstract
Voltage-dependent and calcium-sensitive K+ (MaxiK) channels are key regulators of neuronal excitability, secretion, and vascular tone because of their ability to sense transmembrane voltage and intracellular Ca2+. In most tissues, their stimulation results in a noninactivating hyperpolarizing K+ current that reduces excitability. In addition to noninactivating MaxiK currents, an inactivating MaxiK channel phenotype is found in cells like chromaffin cells and hippocampal neurons. The molecular determinants underlying inactivating MaxiK channels remain unknown. Herein, we report a transmembrane β subunit (β2) that yields inactivating MaxiK currents on coexpression with the pore-forming α subunit of MaxiK channels. Intracellular application of trypsin as well as deletion of 19 N-terminal amino acids of the β2 subunit abolished inactivation of the α subunit. Conversely, fusion of these N-terminal amino acids to the noninactivating smooth muscle β1 subunit leads to an inactivating phenotype of MaxiK channels. Furthermore, addition of a synthetic N-terminal peptide of the β2 subunit causes inactivation of the MaxiK channel α subunit by occluding its K+-conducting pore resembling the inactivation caused by the “ball” peptide in voltage-dependent K+ channels. Thus, the inactivating phenotype of MaxiK channels in native tissues can result from the association with different β subunits.
Ca2+-activated K+ channels, also known as BK, MaxiK, or slo channels, are key modulators of cellular excitability. They are characterized by their large single-channel conductance, intrinsic voltage dependence, Ca2+ modulation, and blockade by charybdotoxin (CTX) and iberiotoxin (1–5). In most tissues, MaxiK channels produce noninactivating currents when activated by depolarization and/or an increase in intracellular Ca2+. However, in chromaffin cells of the adrenal gland (6) and hippocampal neurons (7), inactivating MaxiK currents are also observed that otherwise resemble their noninactivating counterparts in their biophysical and pharmacological properties. The mechanism of inactivation in MaxiK channels has been investigated in detail in rat chromaffin cells, which express both inactivating and noninactivating MaxiK channels (8). Inactivation is removed by trypsin application to the cytosolic face of the membrane, suggesting the presence of an associated cytosolic inactivating particle (6, 7). A model developed to explain the biophysical and pharmacological properties of inactivating channels in chromaffin cells suggests that these channels are formed by a tetrameric assembly of inactivating and noninactivating subunits. Interestingly, inactivating channels in chromaffin cells are less sensitive to CTX, and heteromeric channels consisting of inactivating and noninactivating isoforms seem to have intermediate toxin sensitivities (9). Identification of a MaxiK channel variant or a subunit capable of producing fast inactivating MaxiK channel currents has so far been elusive (10).
Here, we report a human β subunit (β2) that yields fast inactivating currents when coexpressed with the human MaxiK channel α subunit (hslo), similar to those found in native cells. Deletion and fusion constructs demonstrate that the inactivating particle is located at the intracellular N terminus of this transmembrane β2 subunit. An N-terminal synthetic peptide of this β subunit induces inactivation of MaxiK channels, which resembles N-type inactivation of voltage-dependent K+ channels by “ball” peptides. Thus, N-type inactivation is conferred to MaxiK channels by a “ball” peptide attached to the intracellular N terminus of their associated β subunits.
MATERIALS AND METHODS
Molecular Biology.
Expressed sequence tag (EST) databases were searched with the blast algorithm with the β1 subunit (GenBank accession no. U25138) as a query sequence. Clone oe72h09 was obtained from the American Type Culture Collection and was sequenced on both strands by using fluorescent sequencing (GenBank accession no. AF099137, β2 subunit). The sequence alignment was performed with the GCG program gap by using default values. Linearized clones were used for in vitro transcription (mMESSAGEmMACHINE, Ambion, Austin, TX). cRNA (0.5 μg) was in vitro translated in 25 μl of reticulocyte lysate in the presence of microsomal membranes (Promega) and [35S]methionine. After a 1-hr incubation at 30°C, the microsomal membranes were collected by centrifugation and an aliquot was treated with N-glycosidase F (NEB, Beverly, MA). Proteins were separated by SDS/PAGE and visualized with a PhosphorImager (Molecular Dynamics).
Dot and Northern blots were hybridized according to the manufacturer’s (CLONTECH) instructions, except that Cot-1 DNA was omitted from the hybridization mix. Briefly, at least 20-min prehybridization was followed by overnight hybridization at 65°C with 32P-labeled β2 DNA fragment. The membranes were washed at 55°C with 0.1 × SSC/0.5% SDS. Internal standards were prepared by spotting dilutions of β1 and β2 cRNA (in 100 ng human placenta total RNA as a carrier) on nylon membranes (Nytran plus, Schleicher & Schuell), following the protocol used for preparation of the commercial dot-blot membranes. After hybridization and washing, the membranes were analyzed using PhosphorImaging (Molecular Dynamics).
Clone β2-IR (inactivation removed) was made by deleting 19 N-terminal amino acids from the β2 subunit and introducing a new start codon at position 19 with a PCR primer. The fusion construct β1 + β2 “ball” was made by using the overlap extension method (11).
β2 “ball” peptides consisting of 19 or 26 N-terminal amino acids were synthesized (Genemed Biotechnologies, South San Francisco, CA). The peptides were dissolved to a concentration of 10 mM in 50 mM Tris⋅Cl and adjusted to pH 7. Peptides were further diluted in the bath solution and perfused to the cytoplasmic side of excised patches.
Electrophysiology.
Xenopous laevis oocytes were used as an expression system and injected with 2 ng of α subunit (hslo, GenBank accession no. U11058) and 4 ng of β subunit cRNAs, respectively. Pipette and bath solution were (mM): 110 K-methanesulfonate, 5 KCl, 10 Hepes, 5 N-hydroxyethylethylenediaminetriacetic acid (HEDTA), pH 7.4; pipettes were of 1–3 MΩ. Free Ca2+ concentrations were calculated by using the program chelator (12) and were measured with a Ca2+ electrode (World Precision Instruments, Sarasota, FL). Data analysis was done as described elsewhere (2).
Chemicals.
Dehydrosoyasaponin I (DHS-I) was dissolved in H2O. CTX was from Sigma, Research Biochemicals, or was a kind gift of M. L. Garcia from Merck.
RESULTS AND DISCUSSION
Molecular Characteristics of the Inactivating β Subunit (β2).
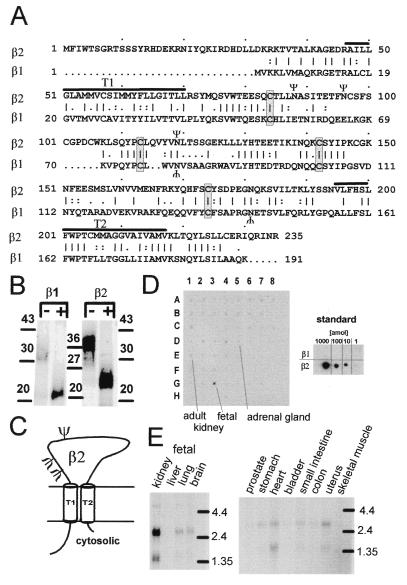
The human β subunit (referred here as β2) was identified in the EST database by using the smooth muscle MaxiK channel β subunit (referred to here as β1) as a probe. Sequencing revealed that an EST clone (I.M.A.G.E. consortium clone ID 1417217), found in a neuroendocrine lung carcinoid library, carried the complete coding sequence with an upstream in-frame stop codon. This human β2 subunit shows 43% protein sequence identity and 53% sequence similarity when compared with the regulatory noninactivating smooth muscle β1 subunit of human MaxiK channels (2). The β2 subunit is distinguished by an N-terminal extension of 31 amino acids, a slightly longer extracellular loop, and five additional amino acids at the C terminus (Fig. 1A). The sequence alignment of β1 and β2 suggests common structural characteristics with two putative transmembrane regions (T1, T2), a long extracellular loop containing three consensus sequences for N-linked glycosylation (ψ), and four conserved cysteine residues that might form structurally important disulfide bridges. Similar to the β1 subunit, in vitro translation of β2 subunit in the presence of microsomal membranes leads to a membrane-incorporated glycosylated protein (Fig. 1B). These features are consistent with the proposed membrane topology shown in Fig. 1C (13). To investigate the tissue distribution of β2 subunit mRNA, we used dot blot membranes, which allow a simultaneous evaluation of gene expression in 50 different human tissues. This dot blot analysis indicates high levels of β2 subunit transcripts in fetal kidney, with no, or weak signals in other tissues including adult kidney and adrenal gland (Fig. 1D). To confirm these findings and to obtain the transcript sizes, we performed Northern blot analysis, which revealed two sizes of β2 transcripts, one at 2.5 kb and the other at 1.5 kb (Fig. 1E). The presence of weak signals of the same sizes in some smooth muscle tissues (stomach, small intestine, colon, uterus) and in heart, fetal lung, and brain suggests that low levels of the β2 subunit message are present in many other tissues.
Figure 1.
Sequence analysis and tissue distribution of the hslo β2 subunit. (A) Sequence comparison of β2 and β1 subunits. Vertical bar, identical; colon, highly conserved; period, conserved amino acids; Ψ, consensus sequences for N-linked glycosylation; T1 and T2, putative transmembrane regions (black lines). (B) In vitro translation and deglycosylation of smooth-muscle β1 and β2 subunits. +, treated; −, untreated with N-glycosidase F. Consistent with the number of N-linked glycosylation sites, deglycosylation reduces the apparent molecular mass to a value corresponding with the calculated molecular mass of β1 (22 kDa) and β2 (27 kDa) proteins. Numbers, standard molecular mass in kDa. (C) Proposed membrane topology of the β2 subunit with two membrane-spanning regions separated by a glycosylated extracellular loop. (D and E) Expression profile of β2 mRNA. Poly(A)+ RNA Dot blot (CLONTECH) of 50 human tissues and human Northern blots (CLONTECH) were hybridized at high stringency with a 32P-labeled β2 probe (758-bp AccI, NcoI fragment). Hybridization was visualized by phosphor imaging. Tissues on the human Dot blots are: whole brain (1A), amygdala (2A), caudate nucleus (3A), cerebellum (4A), cerebral cortex (5A), frontal lobe (6A), hippocampus (7A), medulla oblongata (8A), occipital lobe (1B), putamen (2B), substantia nigra (3B), temporal lobe (4B), thalamus (5B), nucleus accumbens (6B), spinal cord (7B), heart (1C), aorta (2C), skeletal muscle (3C), colon (4C), bladder (5C), uterus (6C), prostate (7C), stomach (8C), testis (1D), ovary (2D), pancreas (3D), pituitary gland (4D), adrenal gland (5D), thyroid gland (6D), salivary gland (7D), mammary gland (8D), kidney (1E), liver (2E), small intestine (3E), spleen (4E), thymus (5E), peripheral leukocyte (6E), lymph node (7E), bone marrow (8E), appendix (1F), lung (2F), trachea (3F), placenta (4F), no sample (5F-8F, G8), fetal brain (G1), fetal heart (G2), fetal kidney (G3), fetal liver (G4), fetal spleen (G5), fetal thymus (G6), fetal lung (G7), 100 ng of yeast total RNA (H1), 100 ng yeast tRNA (H2), 100 ng Escherichia coli rRNA (H3), 100 ng E. coli DNA (H4), 100 ng poly(rA) (H5), 100 ng human Cot 1 DNA (H6), 100 ng human DNA (H7), 500 ng human DNA (H8). The amount of poly(A)+ RNA varies from 89 to 514 ng because of normalization with respect to ubiquitously expressed mRNAs. (D Inset) Internal Dot blot standard of β1 and β2 transcripts show a detection limit of 6 × 105 β2 subunit transcripts.
β2 Subunit Induces Fast Inactivating MaxiK Currents.
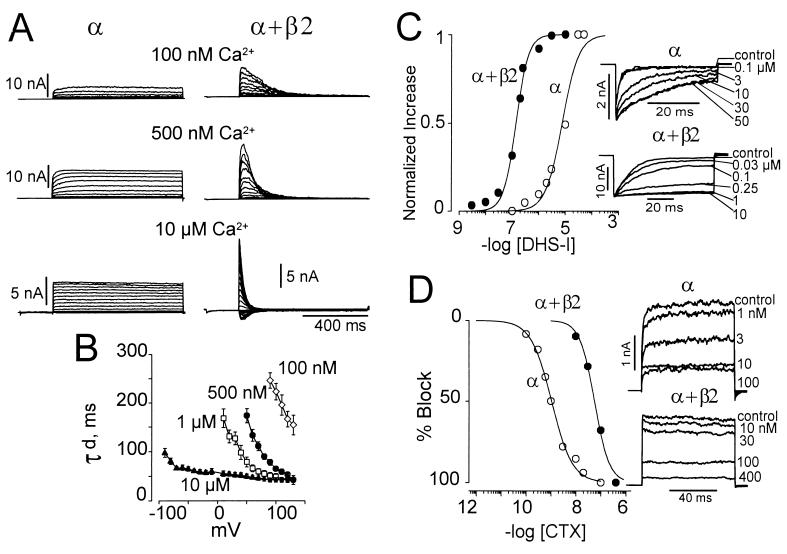
Coexpression of the human β2 subunit along with the pore-forming α subunit of MaxiK channels (hslo) induced fast inactivating currents (Fig. 2A) that resemble those found in native rat chromaffin cells (6). In contrast, the α subunit alone produced sustained currents that do not inactivate when identical pulse protocols are applied. The time constant of current decay (τd) induced by the β2 subunit is increased by voltage and intracellular Ca2+ (Fig. 2B). Under conditions where MaxiK channels have a low open probability and activate slowly (100 nM, 500 nM, and 1 μM Ca2+, voltages below ≈100 mV), the steep voltage dependence of the macroscopic current decay induced by the β2 subunit most likely reflects the voltage dependence of the activation process. In contrast, under high open probability conditions, where channel activation is much faster than inactivation (at 10 μM Ca2+, voltages above 0 mV), the macroscopic current decay reflects primarily the rate of inactivation. In this case, the speed of inactivation increases minimally with depolarizing voltages. These results indicate that the inactivation caused by the β2 subunit is weakly voltage dependent and is favored by open channels, similar to N-type inactivation found in voltage dependent K+ channels (14).
Figure 2.
Human β2 induces inactivation of the α subunit of MaxiK (hslo) channels. (A) Noninactivating currents from MaxiK α subunit alone and fast inactivating currents from α + β2 subunits at different voltages and [Ca2+]i. Currents at 100 nM and 500 nM Ca2+ were elicited after a 200-ms prepulse to −60 and −80 mV, respectively; step potentials: −20 mV to +120 mV in 10 mV increments. In this and following figures, currents are from inside-out patches (except in Fig. 2D), the prepulse duration was 200 ms, and the holding potential was 0 mV. In 10 μM Ca2+, the prepulse was to −140 mV. Step potentials: −100 mV to 110 mV for α subunit, −150 mV to 110 mV for α + β2 subunits in increments of 10 mV. Prepulse potentials were used that relieve inactivation and give maximal currents. Note that the β2 subunit leads to a slower current activation, similar to β1 subunit (2); (compare Middle to Fig. 3C). (B) Time constants of current decay (τd) as a function of voltage at various [Ca2+]i. Values were obtained by fitting the current decay to a single exponential function. (C and D) Pharmacological changes induced by coexpression of β2. β2 subunit increases the sensitivity of the α subunit to DHS-I from an EC50 of 8 μM to 139 nM. Data were normalized to the maximum effect. To allow activation of MaxiK channels by DHS-I, data were obtained at potentials where the open probability is below 0.1 (−140 mV for α + β2, −60 mV for α). (D) β2 decreases the sensitivity of MaxiK channels for CTX from a Kd of 1 nM to 56 nM (at +40 mV, 110 mM symmetrical K+), outside-out patch. The apparent low Kd value (1 nM, in 110 mM symmetrical K+) for CTX on the α subunit alone as compared with the reported 20 nM (in 250 mM symmetrical K+) (1) may be explained by the strong effect of ionic strength on the CTX dissociation constant (35). The β2 subunit construct used for DHS-I and CTX experiments was the deletion Δ2–19 (β2-IR); see Fig. 3.
β2 Subunit Alters the Pharmacology of the MaxiK α Subunit.
Besides causing inactivation, the β2 subunit also changed the pharmacology of the pore-forming α subunit. It has been thought that DHS-I, a compound isolated from a Ghanese medicinal herb (15), specifically activates MaxiK channels formed by α and β1 subunits (16). Here we show that DHS-I also activates channels coexpressed with the β2 subunit in the nanomolar range (mean EC50 = 139 nM ± 36, n = 2), similar to the effect seen on hslo + β1 channels (16, 17). We also found that DHS-I, at higher doses, is capable of activating channels formed by the α subunit alone with an EC50 of 7.6 μM (Fig. 2C). The amount of DHS-I required for an increase in voltage sensitivity of 25 mV is 250 nM if α + β2 subunits are coexpressed, whereas it is 40 μM for α subunit alone (n = 2). Thus, β1 and β2 subunits increase the DHS-I sensitivity of the MaxiK channel α subunit rather than conferring it. This finding also suggests that the α subunit by itself contains a DHS-I binding site and either the binding or the functional effect of DHS-I is enhanced by the presence of the β subunit. Apart from studying the effect of the activator DHS-I, we also tested the effect of a typical pore blocker, CTX. Coexpression of the inactivating β2 subunit lowered the sensitivity of the α subunit to CTX from an EC50 of 1 nM (α subunit alone) to 58 nM (α + β2) (Fig. 2D). This change in CTX sensitivity is similar to the finding in chromaffin cells where inactivating MaxiK currents show a lower CTX sensitivity than noninactivating MaxiK currents (9). Thus, it is likely that an inactivating MaxiK channel β subunit is responsible for the phenotypic variations of MaxiK channels in chromaffin cells rather than possible inactivating splice variants of the pore-forming α subunit.
β2 Subunit N Terminus Carries an Inactivating “Ball” Peptide.
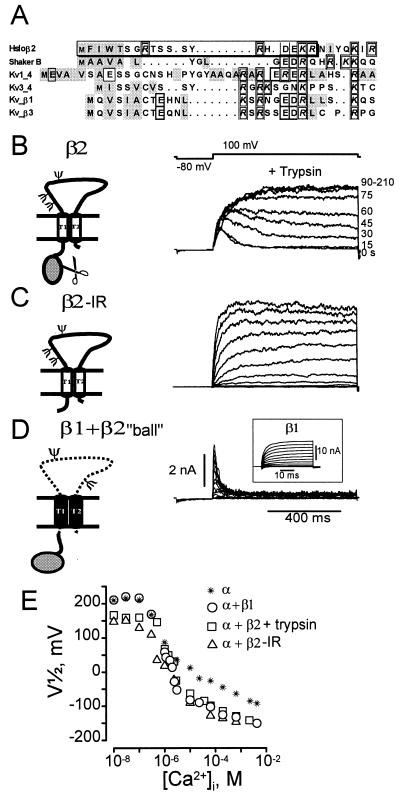
Fast inactivation, also called N-type inactivation, in voltage-dependent K channels occurs by means of a “ball and chain” mechanism (18), mediated by about 20 N-terminal amino acids, with the “ball” located either on the α subunit or on cytosolic modulatory β subunits (Fig. 3A) (19–22). Because “ball”-inactivating peptides are located at the N terminus of α and β subunits, we compared the N-terminal sequences of the β2-subunit with those of other inactivating K+ channel subunits (Fig. 3A). The sequence comparison revealed that, despite the overall low sequence conservation among all these inactivating “balls,” the β2 N terminus shares two features with other “ball” sequences: the presence of hydrophobic amino acids following the methionine start site and charged residues at the end of the “ball.” To test whether the amino terminus of the β2 forms an intracellular inactivating “ball peptide,” we proceeded with the following strategy: (i) removal of inactivation by trypsin treatment, (ii) coexpression of a β2 deletion mutant lacking 18 N-terminal amino acids (Δ2–19, β2-IR), and (iii) coexpression of a fusion construct where the inactivating “ball” (residues 1–19) is fused to the N terminus of the noninactivating β1 subunit (construct β1 + β2 “ball”).
Figure 3.
Molecular determinant of MaxiK channel fast inactivation. (A) Sequence comparison of β2 N terminus with other N-terminal inactivating “balls.” Charged amino acids, individually boxed. Hydrophobic amino acids, shaded gray. Nineteen amino acid “ball” peptide, boxed. (B) Trypsin eliminates inactivation. Trypsin (0.2 mg/ml, [Ca2+]i = 1 μM) applied to the intracellular side of oocytes expressing α + β2 channels. Numbers at right, seconds after trypsin addition. (C) Inactivation is removed by deletion of residues 2–19 from the β2 subunit (β2-IR). β2-IR coexpressed with α subunit of MaxiK channels yields noninactivating currents. Voltages were −20 mV to 110 mV, 10 mV intervals; [Ca2+]i = 500 nM Ca2+. (D) Transfer of inactivation by fusion of 19 N-terminal amino acids of β2 subunit to the N terminus of noninactivating β1 subunit (β1 + β2 “ball”). Inactivating currents are from α coexpressed with β1 + β2 “ball.” Voltages: −20 mV to 110 mV in 10-mV increments, prepulse to −80 mV, [Ca2+]i = 1 μM. Inset: Noninactivating currents from α + β1 subunit, 1 μM Ca2+. Test potentials: −100 to 122 mV in steps of 12 mV. (E) Half activation potentials (V1/2) as a function of [Ca2+]i. Channels formed by α subunit alone (*), α + β1 subunits (○), α + β2 subunits after trypsin treatment (□), α+ β2-IR (Δ). β2 subunit increases Ca2+ sensitivity in the channel’s Ca2+ independent phase, e.g., V1/2 at 10 nM Ca2+ for α, α + β1, α + β2 (trypsin), and α + β2-IR is 209 ± 14, 211 ± 6, 159 ± 10, 146 ± 2, respectively (± SD, n ≥ 2). V1/2 values were obtained from voltage activation curves (2).
As expected for an intracellular domain involved in the inactivation process, trypsin (0.2 mg/ml) applied to excised inside-out patches expressing hslo (α subunit) and β2 subunits abolished inactivation within 2 min and produced sustained currents (Fig. 3B). This loss of MaxiK channel inactivation resembles the one observed in chromaffin cells and hippocampal neurons after trypsin treatment (6, 7). Trypsin action led to a gradual slowing of the time constants of inactivation, consistent with previous reports that more than one β subunit is associated with a tetrameric channel (9). The action of trypsin was quite specific for the removal of inactivation, because we did not observe any obvious changes in other channel properties after trypsin treatment (Fig. 3B and E). This trypsin specificity is surprising considering the fact that the putative Ca2+-binding sites of the MaxiK channel α subunit reside in the long intracellular C terminus (23), which contains 84 possible trypsin cleavage sites. A possible explanation for this remarkable specificity of trypsin is that the β2 “ball” peptide may have a flexible and low-ordered structure (24, 25) and is therefore accessible for trypsin cleavage, whereas a tightly packed ordered structure of the long C terminus of MaxiK channels protects its potential trypsin cleavage sites.
Consistent with our hypothesis that the amino terminus of the β2 subunit is responsible for fast inactivation, deletion of 18 N-terminal amino acids of the β2 subunit (β2-IR) produced noninactivating currents with properties very similar to those encountered in channels coexpressing β2 after trypsin treatment (Fig. 3C). The loss of inactivation in the β2-IR is caused by the loss of a functional domain and not by general disruption of the protein, as this β2-IR is still able to sensitize the α subunit for Ca2+ and voltage in a way similar to β1 (2) (see Fig. 3E). This feature is also noticeable when the complete β2 is coexpressed with the α subunit; e.g., at 10 μM Ca2+, currents from α + β2 activate at much lower voltages than the α subunit alone (Fig. 1A, Bottom).
Fusion of the N-terminal inactivating “ball” of the β2 subunit (19 amino acids) to the amino terminus of the noninactivating β1 subunit produces fast inactivating currents (Fig. 3D). For comparison, the inset shows the currents induced by the wild-type noninactivating β1 subunit. This gain-of-function experiment demonstrates that the first 19 amino acids of the β2 subunit are sufficient to produce the inactivating phenotype. As expected, trypsin treatment of channels coexpressed with the β1 + β2 “ball” fusion construct abolished inactivation and produced noninactivating currents with similar Ca2+-voltage dependencies as channels coexpressed with β1 subunit (not shown). It is striking that both β2-IR and trypsin-treated β2 subunits increased the apparent Ca2+/voltage sensitivity of MaxiK channels, analogous to the action of the smooth-muscle β1 subunit (Fig. 3E). For example, at 10 μM Ca2+, the voltage activation curves of channels coexpressed with β1, β2-IR, or trypsin-treated β2 had almost identical V1/2 values, which are ≈100 mV left shifted when compared with the α subunit alone. In contrast to β1, β2 increases the Ca2+/voltage sensitivity of MaxiK channels (diminution in V1/2 ≈50 mV) at Ca2+ concentrations ≤100 nM (Fig. 3E), in the range where the channel open probability is independent of intracellular Ca2+ (2). Similar to the β1 subunit (2), the β2 subunit leads to slower current activation of the α subunit that is difficult to assess in inactivating currents (see Fig. 2A Top and Middle), but becomes much more evident after removal of inactivation (compare traces in Fig. 3C with Fig. 2A at 500 nM Ca2+). All these results indicate that the β2 subunit carries at least two independent functional domains: the N-terminal inactivation domain and a (not yet localized) voltage or Ca2+ sensitization domain.
N-Terminal β2 Synthetic Peptides Induce Inactivation of MaxiK Currents.
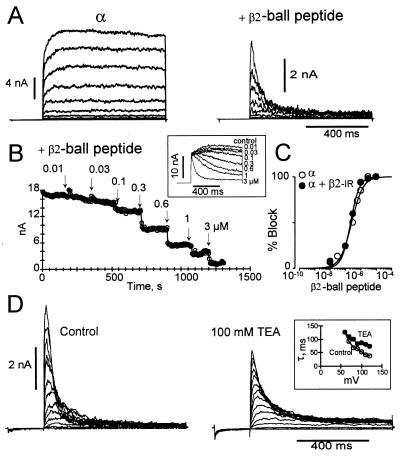
To test further the molecular identity of the inactivating domain of the β2 subunit, we investigated whether synthetic peptides could act as inactivating “balls.” Peptides consisting of the first 26 (Fig. 4) or 19 amino acids of the β2 subunit are able to induce fast inactivating currents in channels formed by the α subunit alone (apparent Kd for both peptides ≈200 nM, Hill coefficient, n ≈1, at +90 mV) (A and B), or coexpressed with the β2 subunit lacking the amino terminus, β2-IR (apparent Kd of ≈200 nM, n ≈1, at +90 mV; C). The rate of peptide-induced inactivation depends on the β2-“ball”peptide concentration and, as predicted for a simple bimolecular reaction, gives a linear relationship with an estimated on-rate (kon) of 4.6 × 106 M−1·s−1. This value is equivalent to the association rate constant of the Shaker B-“ball” peptide for binding to MaxiK channels and to Shaker K channels (26, 27). Assuming a bimolecular reaction, the residence time (τr) of the peptide on MaxiK channels can be calculated from the Kd values (Kd = [kon *τr]−1), which gives ≈1 s for the β2-“ball” peptide vs. ≈5 ms for the Shaker B “ball” peptide (26). Thus, the β2-“ball” peptide binds to its MaxiK channel receptor with a much higher affinity than the Shaker B “ball” peptide. We have proposed that MaxiK channels must possess a hydrophobic “pocket” in its internal vestibule since increasing hydrophobicity of Shaker K+-channel “ball” peptide at its N-terminal end (positions 2, 3, 5) increases the residence time of the peptide in its receptor (28). It is striking that amino acids 2, 3, 4 in the MaxiK β2-inactivating “ball” are more hydrophobic than in other “ball” peptides (Fig. 3A); it is tempting to suggest that these residues are responsible for a longer residence time in its receptor, leading to fast-decaying MaxiK currents.
Figure 4.
N-terminal synthetic peptide of β2 subunit induces inactivation. (A) Currents before and after intracellular application of 3-μM β2-“ball” peptide on MaxiK channel α subunit. Voltages −20 to 110 mV in steps of 10 mV, prepulse −60 mV, Ca2+ = 500 nM. Nineteen or 26 N-terminal amino acid peptides produced similar results. (B) Current blockade vs. time. Test pulses every 4 s, +90 mV (800 ms); prepulse, −60 mV (200 ms), 500 nM [Ca2+]i. Inset: Current blockade at various β2-“ball” peptide concentrations. (C) Kd is 215 nM (α subunit, □) and 250 nM (α + β2-IR, □). Currents were measured at the end of the test pulse. (D) TEA competes with β2-“ball” peptide for blockade of MaxiK channels. Control currents: MaxiK α + β2-IR subunit after intracellular application of 3 μM β2-“ball” peptide. TEA currents: Intracellular application of 100 mM TEA to the same patch slows down inactivation induced by β2-“ball” peptide. Similar results were obtained by using α + β2 channels after trypsin treatment. Pulse protocol was as in A, 500 nM [Ca2+]i. Inset: Time constant of decay (τ) before and after TEA. (A–D) Experiments using the 26-residue NH2-terminal “ball” peptide.
Similar to tetraethylammonium (TEA) (29), “ball” peptides block open channels (14, 26, 30, 31), as if the receptor for the inactivating domain becomes accessible only when the channel gate is open. It has been suggested that TEA might compete with inactivating “balls” for the same binding site (32). Consistent with an open-channel blocking mechanism of β2 subunit, intracellular application of TEA interfered with the β2 subunit “ball” peptide, resulting in a slowdown of inactivation (Fig. 4D). Furthermore, raising extracellular K+ from 5 to 110 mM facilitated the recovery from inactivation (not shown), suggesting that K+ influx destabilizes the interaction of the β2-“ball” with its receptor. Therefore, it is possible that “ball” peptides find their way into the aqueous cavity of the pore (33) and are stabilized by hydrophobic interactions in the inner vestibule of K+ channels.
Our results provide a molecular mechanism to explain MaxiK channel inactivation in native tissues. Currents induced by β2 subunit (α + β2) have a striking resemblance to inactivating MaxiK currents from rat chromaffin cells, including removal of inactivation by trypsin (6), time constant of decay (6), and CTX sensitivity (9) (Fig. 2D). They also share properties with currents from hippocampal neurons, like increased apparent Ca2+ sensitivity (Fig. 3E) and inactivation by means of an open channel block mechanism (Fig. 4D) (7). Yet, α + β2 currents, unlike inactivating currents in chromaffin cells, are sensitive to DHS-I and have a higher Ca2+ sensitivity (10) (Figs. 2D and 3E); and compared to hippocampal neurons their time constant of current decay is faster. In addition, in poly(A)+ RNA dot blots very weak β2 signals were observed in human adrenal gland (Fig. 1D), and in total RNA Northern blots from human and rat adrenal glands β2 signals were undetectable (data not shown). Therefore, it is likely that another inactivating β subunit isoform may be responsible for inactivation in these cells. The presence of an inactivating β2-like subunit may also provide an explanation for the bell-shaped Ca2+ dependence of MaxiK channels in parathyroid cells (34), since channels formed by α + β2 show this behavior if hyperporalizing prepulses, which relieve inactivation, are omitted (data not shown).
Northern and Dot blots show low β2-subunit mRNA levels in many human tissues, including brain (see Fig. 1 D and E). Depending on mRNA and protein turnover, low amounts of β2 mRNA may be sufficient to produce a substantial amount of β2 subunit proteins. In addition, phenotypic expression of inactivating MaxiK currents may also depend on the presence of other noninactivating β subunits that are likely to compete for the same binding site on the α subunit.
Inactivation of MaxiK channels would lead to less hyperpolarizing K+ efflux and therefore is expected to increase Ca2+ entry through voltage-dependent Ca2+ channels in vivo, affecting a variety of functions such as secretion and cell division. During development (β2 subunit mRNA is found in the fetal kidney), the transient expression of inactivating MaxiK channels may allow cell proliferation and tissue development by allowing more Ca2+ to enter the cell. In line with this view, inactivating MaxiK channel β2 subunit EST clones are derived from a neuroendocrine tumor where β2 expression may contribute to cell proliferation.
In summary, the β2 subunit reported here induces MaxiK currents that resemble phenotypic characteristics of the native inactivating MaxiK channels and thus may be the first identified member of a family of inactivating MaxiK channel β subunits.
Acknowledgments
We thank Drs. Ramon Latorre, Francisco Bezanilla, and Enrico Stefani for helpful discussions and suggestions. We also thank Dr. Owen McManus of Merck Research Laboratories for DHS-I. This work was supported by National Institutes of Health grant HL54970 (L.T.) and American Heart Association National Center Grant in-Aid 9750745N (P.M.). L.T. is an Established Investigator of the American Heart Association.
ABBREVIATIONS
- MaxiK
large conductance, voltage and Ca2+-activated K+ channel
- hslo
human MaxiK channel α subunit
- DHS-I
dehydrosoyasaponin I
- CTX
charybdotoxin
- TEA
tetraethylammonium
- β2
transmembrane β subunit
- EST
expressed sequence tag
- IR
inactivation removed
Footnotes
This paper was submitted directly (Track II) to the Proceedings Office.
Data deposition: The sequence reported in this paper has been deposited in GenBank database (accession no. AF099137).
References
- 1.Wallner M, Meera P, Ottolia M, Kaczorowski G, Latorre R, Garcia M L, Stefani E, Toro L. Recept Channels. 1995;3:185–199. [PubMed] [Google Scholar]
- 2.Meera P, Wallner M, Jiang Z, Toro L. FEBS Lett. 1996;382:84–88. doi: 10.1016/0014-5793(96)00151-2. [DOI] [PubMed] [Google Scholar]
- 3.Stefani E, Ottolia M, Noceti F, Olcese R, Wallner M, Latorre R, Toro L. Proc Natl Acad Sci USA. 1997;94:5427–5431. doi: 10.1073/pnas.94.10.5427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cui J, Cox D H, Aldrich R W. J Gen Physiol. 1997;109:647–673. doi: 10.1085/jgp.109.5.647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Toro L, Meera P, Wallner M, Tanaka Y. News Physiol Sci. 1998;13:112–117. doi: 10.1152/physiologyonline.1998.13.3.112. [DOI] [PubMed] [Google Scholar]
- 6.Solaro C R, Lingle C J. Science. 1992;257:1694–1698. doi: 10.1126/science.1529355. [DOI] [PubMed] [Google Scholar]
- 7.Hicks, G. A. & Marrion, N. V. (1998) J. Physiol. (London)508 (Pt. 3), 721–734. [DOI] [PMC free article] [PubMed]
- 8.Solaro C R, Ding J P, Li Z W, Lingle C J. Biophys J. 1997;73:819–830. doi: 10.1016/S0006-3495(97)78114-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ding J P, Li Z W, Lingle C J. Biophys J. 1998;74:268–289. doi: 10.1016/S0006-3495(98)77785-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Saito M, Nelson C, Salkoff L, Lingle C J. J Biol Chem. 1997;272:11710–11717. doi: 10.1074/jbc.272.18.11710. [DOI] [PubMed] [Google Scholar]
- 11.Horton R M, Cai Z L, Ho S N, Pease L R. BioTechniques. 1990;8:528–535. [PubMed] [Google Scholar]
- 12.Schoenmakers T J, Visser G J, Flik G, Theuvenet A P. BioTechniques. 1992;12:870–4. , 876–879. [PubMed] [Google Scholar]
- 13.Knaus H G, Folander K, Garcia-Calvo M, Garcia M L, Kaczorowski G J, Smith M, Swanson R. J Biol Chem. 1994;269:17274–17278. [PubMed] [Google Scholar]
- 14.Demo D D, Yellen G. Neuron. 1991;7:743–753. doi: 10.1016/0896-6273(91)90277-7. [DOI] [PubMed] [Google Scholar]
- 15.McManus O B, Harris G H, Giangiacomo K M, Feigenbaum P, Reuben J P, Addy M E, Burka J F, Kaczorowski G J, Garcia M L. Biochemistry. 1993;32:6128–6133. doi: 10.1021/bi00075a002. [DOI] [PubMed] [Google Scholar]
- 16.McManus O B, Helms L M, Pallanck L, Ganetzky B, Swanson R, Leonard R J. Neuron. 1995;14:645–650. doi: 10.1016/0896-6273(95)90321-6. [DOI] [PubMed] [Google Scholar]
- 17.Tanaka Y, Meera P, Song M, Knaus H-G, Toro L. J Physiol. 1997;502:545–557. doi: 10.1111/j.1469-7793.1997.545bj.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bezanilla F, Armstrong C M. J Gen Physiol. 1977;70:549–566. doi: 10.1085/jgp.70.5.549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hoshi T, Zagotta W N, Aldrich R W. Science. 1990;250:533–538. doi: 10.1126/science.2122519. [DOI] [PubMed] [Google Scholar]
- 20.Zagotta W N, Hoshi T, Aldrich R W. Science. 1990;250:568–571. doi: 10.1126/science.2122520. [DOI] [PubMed] [Google Scholar]
- 21.Rettig J, Heinemann S H, Wunder F, Lorra C, Parcej D N, Dolly J O, Pongs O. Nature (London) 1994;369:289–294. doi: 10.1038/369289a0. [DOI] [PubMed] [Google Scholar]
- 22.Latorre R, Stefani E, Toro L. In: Calcium and Cellular Metabolism. Sotelo J R, Benech J C, editors. New York: Plenum; 1997. pp. 59–71. [Google Scholar]
- 23.Schreiber M, Salkoff L. Biophys J. 1997;73:1355–1363. doi: 10.1016/S0006-3495(97)78168-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Antz C, Geyer M, Fakler B, Schott M K, Guy H R, Frank R, Ruppersberg J P, Kalbitzer H R. Nature (London) 1997;385:272–275. doi: 10.1038/385272a0. [DOI] [PubMed] [Google Scholar]
- 25.Schott M K, Antz C, Frank R, Ruppersberg J P, Kalbitzer H R. Eur Biophys J. 1998;27:99–104. doi: 10.1007/s002490050115. [DOI] [PubMed] [Google Scholar]
- 26.Toro L, Stefani E, Latorre R. Neuron. 1992;9:237–245. doi: 10.1016/0896-6273(92)90163-8. [DOI] [PubMed] [Google Scholar]
- 27.Zagotta W N, Hoshi T, Aldrich R W. Science. 1990;250:568–571. doi: 10.1126/science.2122520. [DOI] [PubMed] [Google Scholar]
- 28.Toro L, Ottolia M, Stefani E, Latorre R. Biochemistry. 1994;33:7220–7228. doi: 10.1021/bi00189a026. [DOI] [PubMed] [Google Scholar]
- 29.Yellen G. J Gen Physiol. 1984;84:157–186. doi: 10.1085/jgp.84.2.157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Foster C D, Chung S, Zagotta W N, Aldrich R W, Levitan I B. Neuron. 1992;9:229–236. doi: 10.1016/0896-6273(92)90162-7. [DOI] [PubMed] [Google Scholar]
- 31.Murrell-Lagnado R D, Aldrich R W. J Gen Physiol. 1993;102:949–975. doi: 10.1085/jgp.102.6.949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Choi K L, Aldrich R W, Yellen G. Proc Natl Acad Sci USA. 1991;88:5092–5095. doi: 10.1073/pnas.88.12.5092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Doyle D A, Cabral J M, Pfuetzner R A, Kuo A, Gulbis J M, Cohen S L, Chait B T, MacKinnon R. Science. 1998;280:69–77. doi: 10.1126/science.280.5360.69. [DOI] [PubMed] [Google Scholar]
- 34.Jia M, Ehrenstein G, Iwasa K. Proc Natl Acad Sci USA. 1988;85:7236–7239. doi: 10.1073/pnas.85.19.7236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Anderson C S, MacKinnon R, Smith C, Miller C. J Gen Physiol. 1988;91:317–333. doi: 10.1085/jgp.91.3.317. [DOI] [PMC free article] [PubMed] [Google Scholar]