Abstract
Ultralarge von Willebrand factor (UL-VWF) multimers are thought to play a central role in pathogenesis of the disease thrombotic thrombocytopenic purpura (TTP); however, experimental evidence in support of this hypothesis has been difficult to establish. Therefore, to examine directly the requirement for VWF in TTP pathogenesis, we generated ADAMTS13-deficient mice on a TTP-susceptible genetic background that were also either haploinsufficient (Vwf+/−) or completely deficient (Vwf−/−) in VWF. Absence of VWF resulted in complete protection from shigatoxin (Stx)–induced thrombocytopenia, demonstrating an absolute requirement for VWF in this model (Stx has been shown previously to trigger TTP in ADAMTS13-deficient mice). We next investigated the requirements for ADAMTS13 and VWF in a murine model of endotoxemia. Unlike Stx-induced TTP findings, LPS-induced thrombocytopenia and mortality were not affected by either VWF or ADAMTS13 deficiency, suggesting divergent mechanisms of thrombocytopenia between these 2 disorders. Finally, we show that VWF deficiency abrogates the ADAMTS13-deficient prothrombotic state, suggesting VWF as the only relevant ADAMTS13 substrate under these conditions. Together, these findings shed new light on the potential roles played by ADAMTS13 and VWF in TTP, endotoxemia, and normal hemostasis.
Introduction
Adhesion molecules and their receptors on platelets, alongside of coagulation factors, are important players in thrombus formation on an injured vessel wall.1 The plasma glycoprotein von Willebrand factor (VWF) is an adhesion molecule that provides the initial link between circulating platelets and sites of vascular injury by binding to components of the extracellular matrix, and to the platelet surface glycoprotein GPIb alpha. VWF also contributes to thrombus formation indirectly by binding and stabilizing coagulation factor VIII.2 The essential role of VWF in hemostasis is illustrated clinically in patients with the bleeding disorder von Willebrand disease; and in mouse models, the role of VWF in occlusive thrombus formation is evident at both arterial and venous shear rates.3,4
VWF is synthesized by endothelial cells and megakaryocytes where it is processed from an initial pro-VWF monomer into larger multimeric forms.5,6 VWF not released constitutively is stored in specialized organelles both in endothelial cells (Weibel-Palade bodies), and platelets (alpha granules).7 Upon cellular activation, stored VWF is released in a form termed ultralarge VWF (UL-VWF), which is thought to represent the most adhesive and thrombogenic form of this molecule.8,9 Soon after release, UL-VWF is processed into smaller and less thrombogenic multimers by the metalloprotease ADAMTS13, and therefore UL-VWF is not typically detected in normal human plasma.10,11
In 1982, Moake et al demonstrated the presence of UL-VWF in the plasma of patients suffering from chronic relapsing thrombotic thrombocytopenic purpura (TTP),12 a disorder of thrombotic microangiopathy (TMA) characterized clinically by microangiopathic hemolytic anemia, thrombocytopenia, neurologic abnormalities, fever, and renal dysfunction.10 These investigators hypothesized that UL-VWF played an important role in the formation of the platelet and VWF-rich thrombi characteristic of this disease. Pathogenesis of TTP has now been linked to deficiency of ADAMTS13, which can either be familial (mutations in the ADAMTS13 gene) or more commonly acquired (inhibitory autoantibodies generated against ADAMTS13).13–16
Investigation into the natural history of TTP has demonstrated that ADAMTS13 deficiency is necessary but not sufficient for the development of this disease, suggesting the requirement for additional genetic and/or environmental factors in TTP pathogenesis (reviewed in Desch and Motto17). Recently, we have developed a mouse model of TTP that recapitulates many of the key clinical features of this disease, including a requirement for further genetic and environmental factors in addition to ADAMTS13 deficiency.18 Immunohistologic studies demonstrated that the thrombi that develop in these mice are remarkably similar to those that are found in patients with TTP, staining strongly for VWF, and relatively weakly for fibrin.18,19 These and other studies suggest that in both humans and mice, VWF is a key component in TTP pathogenesis. In addition, although not yet observed in humans, we and others have found that complete deficiency of ADAMTS13 in mice is prothrombotic, even in the absence of findings consistent with TTP.20,21
Despite the important role postulated for VWF in TTP pathogenesis, direct experimental evidence in support of this hypothesis is lacking. In addition, decreased levels of ADAMTS13 (along with impaired UL-VWF proteolysis) and increased levels of plasma VWF have been hypothesized to be contributory to the pathophysiology of sepsis.22–24 Therefore, to investigate these hypotheses experimentally, we undertook a genetic approach and generated mice deficient for both ADAMTS13 and VWF on 2 genetic backgrounds: (1) a mixed-strain background to study shigatoxin (Stx)–induced TTP pathology, and (2) a C57BL/6 background to investigate LPS-induced pathology. In addition, to date, VWF is the only known substrate of ADAMTS13. We realized that that generation of a mouse line deficient for both ADAMTS13 and VWF on the C57BL/6 background could be used to investigate whether other substrates exist for ADAMTS13. We reasoned that if VWF indeed is the only relevant substrate for ADAMTS13, then complete VWF deficiency should abrogate the prothrombotic state induced by ADAMTS13 deficiency. Conversely, continuing to observe an effect of ADAMTS13 deficiency in the absence of VWF would suggest the existence of an ADAMTS13 substrate(s) in addition to VWF.
Methods
Mouse strains
Mice of strains 129 × 1/Sv, C57BL/6, and CASA/Rk were obtained from The Jackson Laboratory (Bar Harbor, ME). Mice were maintained in a specific pathogen-free facilities at the University of Michigan and the CBR Institute for Biomedical Research. All procedures were approved by the Institutional Animal Care and Use Committee (IACUC) at each institution. To generate mice for the Stx studies, Vwf−/− mice on the C57BL/6 background were crossed with Adamts13+/− mice that had been backcrossed to the CASA/Rk strain 4 generations. The resulting mice that were heterozygous for both Adamts13 and Vwf were then crossed with Adamts13B/CN2−/− mice that we have reported previously.18 Resulting mice that were homozygous null for Adamts13 and heterozygous for Vwf were then intercrossed to obtain the required combination of genotypes. The strain contributions for these mice were approximately 61% CASA/Rk, 32% C57BL/6, and 7% 129 × 1/Sv. As these mice were derived by several generations of matings, they were not genetically identical to those mice that we have studied and reported previously.18
The mice used for the LPS and thrombosis studies were backcrossed 8 generations onto C57BL/6. Wild-type control mice used for the thrombosis studies were obtained from The Jackson Laboratory, otherwise littermate controls were used for all experiments.
Platelet count analysis
Blood was collected into 20-μL EDTA-coated capillary tubes (Drew Scientific, Dallas, TX) by retro-orbital bleeding under isoflurane anesthesia. Analysis was performed with a Bayer Advia 120 whole blood analyzer (Bayer Diagnostics, Tarrytown, NY) using the murine C57BL/6 algorithm.
VWF ELISA analysis
Plasma VWF levels were quantified by enzyme-linked immunosorbent assay (ELISA) as described,25 using anti-VWF antisera A0082 as the primary antibody, and HRP-conjugated VWF antisera (Dako, Carpinteria, CA) as the secondary. Detection was with 1-step Ultra TMB (Pierce, Rockford, IL). Results were expressed in units relative to pooled C57BL/6 control plasma that was designated as 10.
Stx and LPS challenge
Shigatoxin (Stx-2; Sigma-Aldrich, St Louis, MO) was diluted to a final concentration of 25 pg/μL in normal saline, and anesthetized mice (equal numbers of males and females, age 6 to 8 weeks) were injected intravenously with the indicated dose. LPS from E coli serotype 055:B5 (Sigma-Aldrich) was diluted in sterile PBS to a final concentration of 8 mg/mL, and anesthetized mice (equal numbers of males and females, age 15 to 30 weeks) were injected intraperitoneally with the indicated dose.
Platelet preparation and intravital microscopy
The mice used were 3 to 4 weeks old, both male and female, weighing 12 to 15 g. Blood was harvested from the retro-orbital venous plexus and collected in 1.5-mL polypropylene tubes containing 300 μL heparin (30 U/mL). Infused platelets were isolated from 3- to 5-month-old mice of the same genotype and prepared as described previously.20 Thrombosis in the injured arterioles was studied by intravital microscopy as described previously.20 Briefly, mice were anesthetized with 2.5% tribromoethanol (0.15 mL/10 g), and fluorescent platelets (2.5 × 109 platelets/kg) were infused through the retro-orbital plexus. Mesenteric arterioles of approximately 100-μm diameter were studied. The centerline velocity was measured using a optical Doppler velocity meter (Microcirculation Research Institute, Texas A&M University, College Station, TX) and shear rate was calculated as previously described.26 Whatman paper (Whatman, Clifton, NJ) saturated with FeCl3 (10%) solution was applied topically for 5 minutes, which induced denudation of the endothelium. Subsequently vessels were monitored for 40 minutes after injury, or until occlusion. One arteriole was chosen per mouse.
Quantitative analysis of arteriolar thrombus formation
All analyses were performed blinded to genotype. We evaluated (1) the time required for formation of a thrombus larger than 20 μm, (2) the occlusion time of the vessel, (ie, the time required for blood to stop flowing for 30 seconds), and (3) thrombus growth (fold increase), which was calculated by dividing the diameter of the thrombus at time (n) by the diameter of the same thrombus at time (0).
Statistical analysis
Results are reported as the mean plus or minus SEM, unless stated otherwise. The statistical significance of the difference between means was assessed by unpaired Student t test (for comparison of 2 groups) or ANOVA (for more than 2 groups), followed by Bonferroni comparison test. A value of P less than .05 was considered statistically significant.
Results and discussion
A threshold level of VWF is required for Stx-induced thrombocytopenia
We have demonstrated previously that mice comprising a genetic contribution from the CASA/Rk, C57BL/6, and the 129 × 1/Sv strains exhibit thrombocytopenia and other TTP findings in response to Stx, but mice comprising only the C57BL/6 and 129 × 1/Sv backgrounds do not.18 Therefore to investigate the requirement for VWF in Stx-induced thrombocytopenia, we crossed the VWF-null allele3 onto the CASA/Adamts13−/− background, ultimately resulting in mice that were all ADAMTS13 deficient, and either wild-type, heterozygous-null, or homozygous-null for Vwf(Vwf+/+, Vwf+/−, or Vwf−/−, respectively). From birth through the time of study, these mice did not become ill appearing or demonstrate blood count findings consistent with TTP.
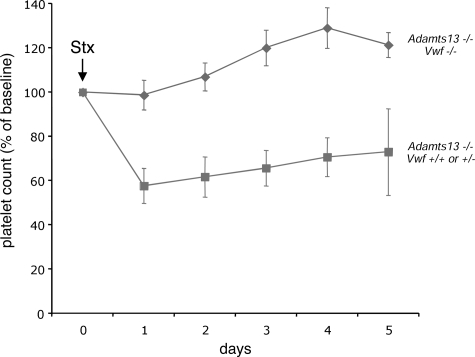
Challenge of the VWF-expressing (Vwf+/+ or Vwf+/−) CASA/Adamts13−/− mice with Stx resulted in significant thrombocytopenia (Figure 1 dashed line), similar to what we have observed previously.18 In contrast, littermate CASA/Adamts13−/− mice deficient for VWF (Vwf−/−) did not respond to the same dose of Stx (Figure 1 solid line). In addition, 6-day mortality in the VWF-expressing mice (81% [13 of 16]) exceeded that of the VWF-deficient mice (56% [5 of 9], P < .02). These results demonstrate the clear requirement for VWF in Stx-induced thrombocytopenia and TMA-related mortality in this system, and provide formal experimental evidence supporting the crucial role of VWF in the pathogenesis of TTP. The 56% mortality observed in the VWF-deficient mice was not due to TMA, as these mice did not develop thrombocytopenia, and likely is secondary to the nonspecific toxic effects of Stx that have been observed in a wide range of animal models.27
Figure 1.
VWF is required for Stx-induced thrombocytopenia. CASA/Adamts13−/− mice that were either deficient for VWF (Vwf−/−, solid line with diamonds) or expressing VWF (Vwf+/+ or Vwf+/−, dashed line with squares) were injected via the lateral tail vein with Stx (250 pg/g body weight) and followed for 5 days. Blood from the retro-orbital plexus was obtained daily for automated platelet count analysis. For each group of mice, the platelet count data were averaged and expressed as the percentage change from baseline values obtained before injection of Stx. The number of mice injected was 16 for the Vwf+/+ and Vwf+/− group and 9 for the Vwf−/− group. The error bars represent the standard error of the mean.
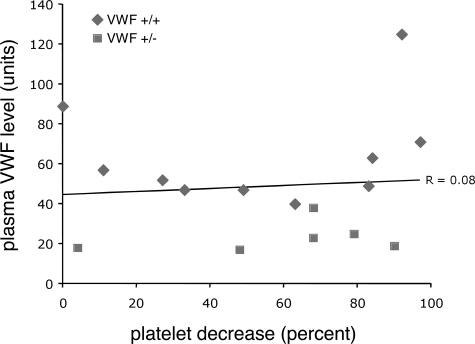
We have also demonstrated previously that plasma VWF levels did not correlate with Stx-induced pathology, suggesting that an elevated VWF level is not a TTP risk factor in mice.18 In this current experiment, the VWF-expressing mice also exhibited a wide range of plasma VWF, with the Vwf+/− animals not surprisingly possessing the lowest levels. Thus we were again able to compare prechallenge plasma VWF level with degree of response to Stx, and similarly, no correlation was observed (Figure 2). Together, these results suggest that a threshold level of VWF is required for induction of Stx-induced thrombocytopenia (and therefore likely TTP), but that further increases may not result in worsening disease. Although these results do not demonstrate the absolute threshold of VWF required for Stx-induced thrombocytopenia, we can infer this value must at least be as low as 20 units (2 times the C57BL/6 reference of 10 units), as several mice with VWF of approximately 20 units responded to Stx (Figure 2). Although remaining to be confirmed in humans, these data suggest that the wide variation in plasma VWF levels in the human population alone may not be a primary determinant of TTP susceptibility or disease severity.
Figure 2.
VWF level does not correlate with the degree of Stx-induced thrombocytopenia. Plasma VWF levels were obtained for the Vwf+/+ and Vwf+/− mice shown in Figure 1, and were plotted against the percentage platelet decrease from baseline for each mouse. The R value was calculated using Microsoft Excel (Microsoft, Redmond, WA). Diamonds indicate Vwf+/+ mice; squares, Vwf+/− mice.
LPS-induced thrombocytopenia occurs independently of VWF, and ADAMTS13 deficiency does not worsen LPS-induced thrombocytopenia or mortality
Intraperitoneal injection of mice with LPS is a well-established animal model of endotoxemia and Gram-negative sepsis, and reliably induces thrombocytopenia in wild-type C57BL/6 mice.28,29 Therefore to investigate the functions of VWF and ADAMTS13 in this process, we crossed the VWF-null allele onto the ADAMTS13-deficient background, ultimately resulting in littermate mice (8 generations backcrossed to C57BL/6) that were deficient for either ADAMTS13 (Adamts13−/−, Vwf+/−), VWF (Adamts13+/−, Vwf−/−), both (Adamts13−/−, Vwf−/−), or neither (Adamts13+/−, Vwf+/−). Equal numbers of mice in each group were challenged individually with LPS, and platelet counts were followed for 4 days. Mice of the mixed CASA genetic background were not used for LPS studies because preliminary experiments in genetically similar mixed background mice resulted in variability sufficient to preclude interpretation (data not shown).
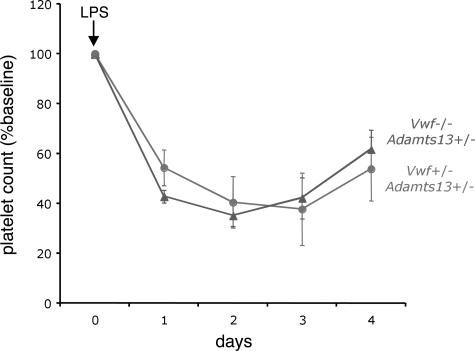
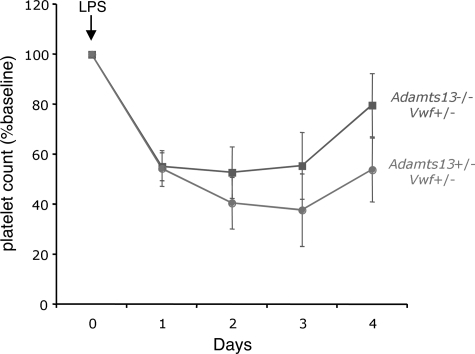
As shown, VWF-expressing and VWF-deficient mice responded similarly to LPS challenge (Figure 3). There also was no significant difference in LPS-induced thrombocytopenia between ADAMTS13-expressing and ADAMTS13-deficient mice (Figure 4). Furthermore, mice deficient for both VWF and ADAMTS13 demonstrated a comparable degree of LPS-induced thrombocytopenia (not shown), and all 4 groups exhibited similar mortality (Adamts13−/−, Vwf−/−, 5/12; Adamts13−/−, Vwf+/−, 5/12; Adamts13+/−, Vwf−/−, 4/12; and Adamts13+/−, Vwf+/−, 4/12).
Figure 3.
VWF is not required for LPS-induced thrombocytopenia. Adamts13+/− mice that were either deficient for VWF (Vwf−/−, dashed line with triangles) or expressing VWF (Vwf+/−, solid line with circles) were injected intraperitoneally with LPS (either 12 or 20 μg/g body weight) and followed for 4 days. Blood from the retro-orbital plexus was obtained daily for automated platelet count analysis. For each group of mice, the daily platelet count data were averaged and expressed as the percentage change from baseline values obtained before injection of LPS. The number of mice injected was 12 for each group (8 mice at 12 μg/g, and 4 mice at 20 μg/g). The error bars represent the standard error of the mean.
Figure 4.
ADAMTS13 deficiency does not worsen LPS-induced thrombocytopenia. VWF-expressing mice that were either deficient for ADAMTS13 (Adamts13−/−, solid line with squares) or expressing ADAMTS13 (Adamts13+/−, dashed line with circles) were injected intraperitoneally with LPS (either 12 or 20 μg/g body weight) and followed for 4 days. Blood from the retro-orbital plexus was obtained daily for automated platelet count analysis. For each group of mice, the daily platelet count data were averaged and expressed as the percentage change from baseline values obtained before injection of LPS. The number of mice injected was 12 for each group (8 mice at 12 μg/g, and 4 mice at 20 μg/g). The error bars represent the standard error of the mean.
Thus complete deficiency of either VWF or ADAMTS13 (relative to heterozygous levels) has no effect on thrombocytopenia or mortality in this model of endotoxemia. These results are in clear contrast to the Stx-induced TTP model, where both ADAMTS13 deficiency and a threshold level VWF expression are prerequisites for disease pathogenesis. Although the mice used for the Stx and LPS studies were of different genetic backgrounds (which can result in variability and/or an absence of responses to LPS and Stx), as the underlying pathophysiology is unlikely to differ among strains of the same animal species, these results suggest that the mechanisms of thrombocytopenia may differ (at least in part) between TTP and endotoxemia.
The observation that absolute VWF deficiency has no effect on LPS-induced thrombocytopenia is not consistent with previous hypotheses that elevated plasma VWF may be an important contributing factor to the thrombotic tendencies observed in multiple clinical syndromes, including sepsis (reviewed in Blann24). However, Camerer et al have demonstrated recently that murine LPS-induced thrombocytopenia also does not require platelet PAR4 or fibrinogen,28 and Andonegui et al showed that neutrophils and the LPS receptor (TLR4) expression (both systemically and on platelets) are required for thrombocytopenia secondary to LPS injection.30 Together with our results, these findings suggest that LPS-induced thrombocytopenia may occur primarily through an inflammatory mechanism, rather than via a consumptive process secondary to disseminated intravascular coagulation (DIC). Finally, despite evidence from recent clinical studies associating reduced ADAMTS13 activity with severity of sepsis-induced DIC,22,23 our observation that ADAMTS13 deficiency has no effect in this model perhaps is not surprising in light of the finding that VWF also is not required for LPS-induced thrombocytopenia.
VWF deficiency abrogates the ADAMTS13-deficient prothrombotic state
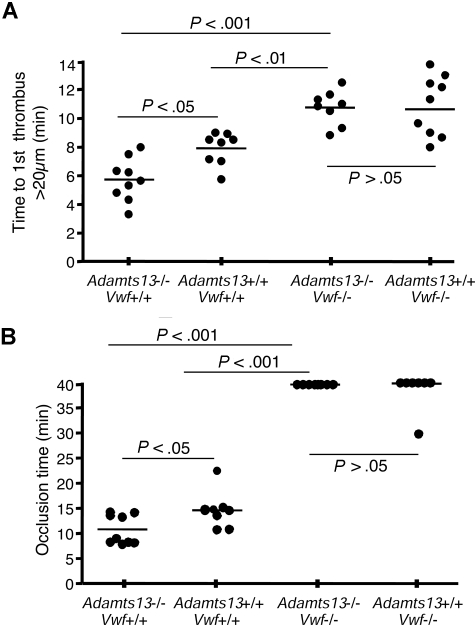
We have previously observed that murine ADAMTS13 deficiency on a mixed genetic background (C57BL/6, 129 × 1/Sv) accelerates thrombosis in injured arterioles.20 Before investigating the role of VWF in this process, we first confirmed our previous findings in Adamts13−/− mice backcrossed 8 generations to the C57BL/6 strain background (Figure 5). Following FeCl3 injury, there was a significant decrease in time needed to form the first thrombus larger than 20 μm in the Adamts13−/− mice (5.7 ± 0.5 minutes) compared with Adamts13+/+ mice (7.9 ± 0.4 minutes, P < .05 [Figure 5A columns 1,2]). In addition, the average time to total FeCl3-induced vessel occlusion was also significantly shortened in Adamts13−/− mice (10.8 ± 1.0 minutes) compared with Adamts13+/+ mice (14.6 ± 1.3 minutes, P < .05 [Figure 5B columns 1,2]). The shear rate and diameter of arterioles studied were similar for both groups of mice (Table 1). These results confirm that the prothrombotic state induced by ADAMTS13 deficiency also occurs in C57BL/6 mice.
Figure 5.
VWF deficiency abrogates the ADAMTS13-deficient prothrombotic state. (A) Quantification of thrombi formation in FeCl3-injured arterioles. In VWF-expressing Adamts13−/− mice, thrombi larger than 20 μm were observed at 5.7 (± 0.5) minutes (column 1), compared with 7.9 (± 0.4) minutes for Adamts13+/+ mice (P < .05, column 2). In contrast, there was no difference in mean time to first thrombus formation larger than 20 μm between mice deficient for both VWF and ADAMTS13 (10.7 ± 0.4 minutes, column 3), compared with VWF-deficient mice expressing ADAMTS13 (10.6 ± 0.7 minutes, column 4). (B) Occlusion time of FeCl3-injured arterioles. VWF-expressing Adamts13−/− mice demonstrated an average occlusion time of 10.8 (± 1.0) minutes (column 1), compared with 14.6 (± 1.3) minutes for Adamts13+/+ mice (P < .05, column 2). In contrast, there was no difference in occlusion time between mice deficient for both VWF and ADAMTS13 (column 3), compared with VWF-deficient mice expressing ADAMTS13 (column 4). Most of the injured arterioles did not occlude in mice of either of these 2 groups. The bars represent the mean of each group.
Table 1.
Hemodynamic parameters were established prior to application of FeCl3 on arterioles (Figure 5)
| Genotype | Diameter, μm | Centerline velocity, mm/s | Shear rate, s−1 |
|---|---|---|---|
| Adamts13+/+, n = 9 | 128.13 ± 16.02 | 37.09 ± 6.0 | 1455.71 ± 220.88 |
| Adamts13−/−, n = 9 | 131.94 ± 18.87 | 40.44 ± 6.23 | 1568.52 ± 381.11 |
| Adamts13+/+, Vwf−/−, n = 8 | 117.19 ± 13.26 | 36.64 ± 6.76 | 1580.18 ± 321.88 |
| Adamts13−/−, Vwf−/−, n = 8 | 115 ± 12.54 | 37.88 ± 3.31 | 1798 ± 434.39 |
Values are represented as the mean plus or minus SD.
We next performed a similar series of experiments in Adamts13+/+ and Adamts13−/− mice that were also deficient for VWF (Figure 5). We reasoned that if VWF indeed is the only relevant substrate for ADAMTS13, then complete VWF deficiency should abrogate the prothrombotic state induced by ADAMTS13 deficiency. Conversely, continuing to observe an effect of ADAMTS13 deficiency in the absence of VWF would suggest the existence of an ADAMTS13 substrate(s) in addition to VWF.
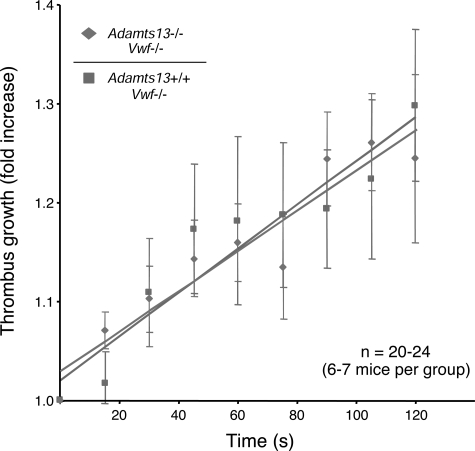
Compared with VWF-expressing mice, VWF deficiency resulted in a significant increase in time to first thrombus larger than 20 μm, which no longer differed between Adamts13−/− mice (10.7 ± 0.4 minutes) and Adamts13+/+ mice (10.6 ± 0.7 minutes [Figure 5A columns 3,4]). Furthermore, the actual rate of individual thrombus growth was nearly identical in the VWF-deficient Adamts13−/− and Adamts13+/+ mice (Figure 6). Finally, regarding the average time to total FeCl3-induced vessel occlusion in the VWF-deficient mice, most of the injured arterioles did not occlude in either the Adamts13+/+ or Adamts13−/− mice (Figure 5B columns 3,4), demonstrating that ADAMTS13 deficiency was not sufficient to overcome the effect of VWF deficiency in this system.
Figure 6.
ADAMTS13 deficiency does not affect the rate of thrombus growth in the absence of VWF. Thrombus growth kinetics were calculated by dividing the diameter of the thrombus at time (n) by the diameter of the same thrombus at time (0). Thrombi that were at least 30 μm in diameter were followed for 2 minutes. The rate of thrombus growth in the VWF-deficient Adamts13−/− mice (dashed line with diamonds) was similar to the VWF-deficient Adamts13+/+ mice (solid line with squares). The slope for both groups of mice was 0.002. The errors bars represent the standard error of the mean.
These results demonstrate that the absence of ADAMTS13 in the setting of VWF deficiency no longer results in a prothrombotic state, suggesting VWF as the only relevant substrate for ADAMTS13 in this model of thrombus growth and stability under arterial shear conditions. However, as with any similar genetic experiment, the effect of ADAMTS13 on other potential substrates could theoretically be masked by an upstream or downstream requirement for VWF.
In conclusion, we have taken a genetic approach to investigate the combined roles of ADAMTS13 and VWF in murine models of TTP, endotoxemia, and thrombosis. Our results provide formal experimental evidence supporting the hitherto putative crucial role for VWF in the pathogenesis of TTP, but conversely, do not support an essential role for ADAMTS13 or VWF in a murine model of endotoxemia-associated thrombocytopenia. When considered together with recent findings from other groups, these results suggest further that LPS-induced thrombocytopenia may not result solely from platelet consumption mediated by activation of coagulation pathways, but rather, may occur secondary to inflammatory processes.28,30 Finally, we also provide initial evidence in support of the notion that VWF is the major, and perhaps only, substrate for ADAMTS13 in the setting of arteriolar thrombosis.
Acknowledgments
This work was supported by the National Heart, Lung, and Blood Institute of the National Institutes of Health grants R37-HL39693 and P01-HL057346 (D.G.), R37-HL41002 and P01-HL066105 (D.D.W.), and K08-HL076539 (D.G.M.), and by a Sponsored Research Agreement from Baxter, Vienna, Austria (A.K.C. and D.D.W.). D.G.M. is the recipient of an American Society of Hematology Scholar Award, and D.G. in an investigator of the Howard Hughes Medical Institute.
Footnotes
An Inside Blood analysis of this article appears at the front of this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Authorship
Contribution: A.K.C., D.G., D.D.W., and D.G.M. contributed equally to the design of these studies; A.K.C. and D.G.M. wrote the paper, and performed all of the studies with the assistance of M.T.W. and G.Z.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: David G. Motto, University of Iowa, 3269C Carver Biomedical Research Bldg, Iowa City, IA 52242; e-mail: david-motto@uiowa.edu.
References
- 1.Denis CV, Wagner DD. Platelet adhesion receptors and their ligands in mouse models of thrombosis. Arterioscler Thromb Vasc Biol. 2007;27:728–739. doi: 10.1161/01.ATV.0000259359.52265.62. [DOI] [PubMed] [Google Scholar]
- 2.Ruggeri ZM. Von Willebrand factor, platelets and endothelial cell interactions. J Thromb Haemost. 2003;1:1335–1342. doi: 10.1046/j.1538-7836.2003.00260.x. [DOI] [PubMed] [Google Scholar]
- 3.Denis C, Methia N, Frenette PS, et al. A mouse model of severe von Willebrand disease: defects in hemostasis and thrombosis. Proc Natl Acad Sci U S A. 1998;95:9524–9529. doi: 10.1073/pnas.95.16.9524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chauhan AK, Kisucka J, Lamb CB, Bergmeier W, Wagner DD. von Willebrand factor and factor VIII are independently required to form stable occlusive thrombi in injured veins. Blood. 2007;109:2424–2429. doi: 10.1182/blood-2006-06-028241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sporn LA, Chavin SI, Marder VJ, Wagner DD. Biosynthesis of von Willebrand protein by human megakaryocytes. J Clin Invest. 1985;76:1102–1106. doi: 10.1172/JCI112064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wagner DD, Marder VJ. Biosynthesis of von Willebrand protein by human endothelial cells. J Biol Chem. 1983;258:2065–2067. [PubMed] [Google Scholar]
- 7.Wagner DD. The Weibel-Palade body: the storage granule for von Willebrand factor and P-selectin. Thromb Haemost. 1993;70:105–110. [PubMed] [Google Scholar]
- 8.Arya M, Anvari B, Romo GM, et al. Ultralarge multimers of von Willebrand factor form spontaneous high-strength bonds with the platelet glycoprotein Ib-IX complex: studies using optical tweezers. Blood. 2002;99:3971–3977. doi: 10.1182/blood-2001-11-0060. [DOI] [PubMed] [Google Scholar]
- 9.Sporn LA, Marder VJ, Wagner DD. Inducible secretion of large, biologically potent von Willebrand factor multimers. Cell. 1986;46:185–190. doi: 10.1016/0092-8674(86)90735-x. [DOI] [PubMed] [Google Scholar]
- 10.Moake JL. Thrombotic microangiopathies. N Engl J Med. 2002;347:589–600. doi: 10.1056/NEJMra020528. [DOI] [PubMed] [Google Scholar]
- 11.Tsai HM. Von Willebrand factor, ADAMTS13, and thrombotic thrombocytopenic purpura. J Mol Med. 2002;80:639–647. doi: 10.1007/s00109-002-0369-8. [DOI] [PubMed] [Google Scholar]
- 12.Moake JL, Rudy CK, Troll JH, et al. Unusually large plasma factor VIII:von Willebrand factor multimers in chronic relapsing thrombotic thrombocytopenic purpura. N Engl J Med. 1982;307:1432–1435. doi: 10.1056/NEJM198212023072306. [DOI] [PubMed] [Google Scholar]
- 13.Levy GG, Nichols WC, Lian EC, et al. Mutations in a member of the ADAMTS gene family cause thrombotic thrombocytopenic purpura. Nature. 2001;413:488–494. doi: 10.1038/35097008. [DOI] [PubMed] [Google Scholar]
- 14.Levy GG, Motto DG, Ginsburg D. ADAMTS13 turns 3. Blood. 2005;106:11–17. doi: 10.1182/blood-2004-10-4097. [DOI] [PubMed] [Google Scholar]
- 15.Tsai H, Lian EC. Antibodies to von Willebrand factor-cleaving protease in acute thrombotic thrombocytopenic purpura. N Engl J Med. 1998;339:1385–1394. doi: 10.1056/NEJM199811263392203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Furlan M, Robles R, Galbusera M, et al. von Willebrand factor-cleaving protease in thrombotic thrombocytopenic purpura and the hemolytic-uremic syndrome. N Engl J Med. 1998;339:1578–1584. doi: 10.1056/NEJM199811263392202. [DOI] [PubMed] [Google Scholar]
- 17.Desch KC, Motto DG. Thrombotic thrombocytopenic purpura in humans and mice. Arterioscler Thromb Vasc Biol. 2007;27:1901–1908. doi: 10.1161/ATVBAHA.107.145797. [DOI] [PubMed] [Google Scholar]
- 18.Motto DG, Chauhan AK, Zhu G, et al. Shigatoxin triggers thrombotic thrombocytopenic purpura in genetically susceptible ADAMTS13-deficient mice. J Clin Invest. 2005;115:2752–2761. doi: 10.1172/JCI26007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Asada Y, Sumiyoshi A, Hayashi T, Suzumiya J, Kaketani K. Immunohistochemistry of vascular lesion in thrombotic thrombocytopenic purpura, with special reference to factor VIII related antigen. Thromb Res. 1985;38:469–479. doi: 10.1016/0049-3848(85)90180-x. [DOI] [PubMed] [Google Scholar]
- 20.Chauhan AK, Motto DG, Lamb CB, et al. Systemic antithrombotic effects of ADAMTS13. J Exp Med. 2006;203:767–776. doi: 10.1084/jem.20051732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Banno F, Kokame K, Okuda T, et al. Complete deficiency in ADAMTS13 is prothrombotic, but it alone is not sufficient to cause thrombotic thrombocytopenic purpura. Blood. 2006;107:3161–3166. doi: 10.1182/blood-2005-07-2765. [DOI] [PubMed] [Google Scholar]
- 22.Nguyen TC, Liu A, Liu L, et al. Acquired ADAMTS-13 deficiency in pediatric patients with severe sepsis. Haematologica. 2007;92:121–124. doi: 10.3324/haematol.10262. [DOI] [PubMed] [Google Scholar]
- 23.Ono T, Mimuro J, Madoiwa S, et al. Severe secondary deficiency of von Willebrand factor-cleaving protease (ADAMTS13) in patients with sepsis-induced disseminated intravascular coagulation: its correlation with development of renal failure. Blood. 2006;107:528–534. doi: 10.1182/blood-2005-03-1087. [DOI] [PubMed] [Google Scholar]
- 24.Blann AD. Plasma von Willebrand factor, thrombosis, and the endothelium: the first 30 years. Thromb Haemost. 2006;95:49–55. [PubMed] [Google Scholar]
- 25.Mohlke KL, Nichols WC, Westrick RJ, et al. A novel modifier gene for plasma von Willebrand factor level maps to distal mouse chromosome 11. Proc Natl Acad Sci U S A. 1996;93:15352–15357. doi: 10.1073/pnas.93.26.15352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Frenette PS, Moyna C, Hartwell DW, Lowe JB, Hynes RO, Wagner DD. Platelet-endothelial interactions in inflamed mesenteric venules. Blood. 1998;91:1318–1324. [PubMed] [Google Scholar]
- 27.Laszik ZG, Blakey GL. Searching for a valid animal model of hemolytic uremic syndrome. Am J Clin Pathol. 2002;118:323–325. doi: 10.1309/TC75-EECC-TU3X-FDL9. [DOI] [PubMed] [Google Scholar]
- 28.Camerer E, Cornelissen I, Kataoka H, Duong DN, Zheng YW, Coughlin SR. Roles of protease-activated receptors in a mouse model of endotoxemia. Blood. 2006;107:3912–3921. doi: 10.1182/blood-2005-08-3130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kerlin BA, Yan SB, Isermann BH, et al. Survival advantage associated with heterozygous factor V Leiden mutation in patients with severe sepsis and in mouse endotoxemia. Blood. 2003;102:3085–3092. doi: 10.1182/blood-2003-06-1789. [DOI] [PubMed] [Google Scholar]
- 30.Andonegui G, Kerfoot SM, McNagny K, Ebbert KV, Patel KD, Kubes P. Platelets express functional Toll-like receptor-4. Blood. 2005;106:2417–2423. doi: 10.1182/blood-2005-03-0916. [DOI] [PubMed] [Google Scholar]