SUMMARY
Lewis x (Lex, CD15), also known as SSEA-1 (stage specific embryonic antigen-1), is a trisaccharide with the structure Galβ(1-4)Fucα(1-3)GlcNAc, which is expressed on glycoconjugates in human polymorphonuclear granulocytes and various tumors such as colon and breast carcinoma. We have investigated the role of Lex in the adhesion of MCF-7 human breast cancer cells and PMN to human umbilical endothelial cells (HUVEC) and the effects of two different anti-Lex mAbs (FC-2.15 and MCS-1) on this adhesion. We also analyzed the cytolysis of Lex+-cells induced by anti-Lex mAbs and complement when cells were adhered to the endothelium, and the effect of these antibodies on HUVEC. The results indicate that MCF-7 cells can bind to HUVEC, and that MCS-1 but not FC-2.15 mAb inhibit this interaction. Both mAbs can efficiently lyse MCF-7 cells bound to HUVEC in the presence of complement without damaging endothelial cells. We also found a Lex-dependent PMN interaction with HUVEC. Although both anti-Lex mAbs lysed PMN in suspension and adhered to HUVEC, PMN aggregation was only induced by mAb FC-2.15. Blotting studies revealed that the endothelial scavenger receptor C-type lectin (SRCL), which binds Lex-trisaccharide, interacts with specific glycoproteins of Mr ∼ 28 kD and 10 kD from MCF-7 cells. The interaction between Lex+-cancer cells and vascular endothelium is a potential target for cancer treatment.
Keywords: Lewis x (CD15), monoclonal antibodies FC-2.15 and MCS-1, MCF-7, PMN, scavenger receptor C-type lectin
INTRODUCTION
Lewis x (Lex, CD15), also known as SSEA-1 (stage specific antigen-1) is a trisaccharide with the structure Galβ(1-4)Fucα(1-3)GlcNAc [1, 2] which is expressed on glycoconjugates in human polymorphonuclear granulocytes (PMN) [3, 4] and in different tumors including breast and colon carcinoma [4, 5], Hodgkin's lymphoma [6], and acute promyelocytic leukemia [7]. It has been suggested that Lex is involved in cellular recognition during fertilization, embryogenesis and neural development [8-10]. The adhesive functions of Lex glycans and Lex-specific C-type lectins have gained recent interest because of the finding that C-type lectin DC-SIGN (dendritic cell-specific ICAM [intercellular adhesion molecule]-3-grabbing nonintegrin) and related lectins specifically bind to Lex glycans on pathogens such as Schistosoma mansoni and Helicobacter pylori, and possibly on cellular targets, allowing them to serve as adhesion molecules [11]. Furthermore, it has been recently proposed that tumor cells can interact with DC-SIGN in dendritic cells through Lex glycans present in tumor Ags, such as CEA, leading to suppression of dendritic cell functions [12]. Initial adhesion of cancer cells to endothelium can be mediated by the E-selectin-sLex/sLea system. Sialyl-Lewis x (sLex), (NeuNAcα2-3Galβ1-4(Fucα1-3)GlcNAc) on cancer cells and leukocytes is the major ligand for endothelial E-selectin [13, 14], along with sLea, mediating tethering and rolling on activated endothelial cells. To stably adhere to endothelial cells, cancer cells may potentially follow two known routes: an integrin-mediated firm adhesion pathway as shown for melanoma cells [15], or a non-integrin-mediated pathway based on clustering of endothelial E-selectin or E-selectin ligands, which was proposed for the firm arrest of colon carcinoma cells [16, 17]. For example, Burdick et al. [17] demonstrated that sLea-negative, sLex/di-sLex positive metastatic LS174T colon adenocarcinoma cells adhere to human umbilical vein endothelial cells (HUVEC) through sLex-glycolipids and sLex-glycoproteins that bind to E-selectin. However, numerous rolling cells were still observed after treatment with neuraminidase, suggesting the presence of nonsialylated non-integrin ligands. As it refers to normal PMN, in which sLex has also been implicated as a counterreceptor for selectins in adhesion to endothelium [14, 18], glycoproteins containing Lex such as CD11b/CD18 integrin [19-21] and carcinoembryonic antigen-related cellular adhesion molecule 1 (CEA-CAM1) [22] have also been detected.
Recently, the scavenger receptor C-type lectin (SRCL), another C-type lectin that is highly specific for Lex-trisaccharide was found expressed on endothelial cells [23]. The restricted glycan binding of SRCL and its distribution on vascular endothelial cells suggest that it might have a role in cell adhesion analogous to the leukocyte-endothelial interactions mediated by selectins.
We have previously described the mouse mAb FC-2.15 (IgM/κ), which specifically recognizes Lex and induces complement (C′) mediated lysis of Lex+-cells, such as the human breast cancer cell line MCF-7 [4, 24, 25]. The mAb FC-2.15 has been assayed in a Phase I clinical trial performed in 11 patients with advanced cancer whose tumors expressed Lex [26]. In this trial, a sustained >50% reduction of breast carcinoma liver metastases was observed. The main toxicity found in 10/11 patients was a profound neutropenia that occurred within 1 h after the start of each mAb infusion and was reversed within 1 h after its discontinuation. FC-2.15 effects on MCF-7 cells and PMN were analyzed both in vitro and in ex vivo circulating blood [27]. FC-2.15 induced in vitro PMN homotypic aggregation and lysis when C′ was added. However, homotypic aggregation was not common to all Lex+-cells, since MCF-7 breast cancer cells were lysed in the presence of C′ but were not aggregated.
The aim of this study is to investigate the interrelationship between Lex+-tumor cells and vascular endothelium, and the role of Lex epitopes in this interaction. For this, we compared the effects of two different anti-Lex mAbs on this interaction. We have also analyzed the cytolysis of Lex+-cells adhered to endothelium in the presence of anti-Lex mAbs and C′, and the possible direct effect of anti-Lex mAbs on endothelial cells. Finally, we have investigated if the endothelial scavenger receptor C-type lectin (SRCL) could be implicated in the interaction between Lex+-tumor cells and vascular endothelium.
MATERIALS AND METHODS
Antibodies
Anti-Lex mAb FC-2.15 (IgM) was purified as previously described [4]. Anti-Lex mAb MCS-1 (IgG3) was obtained from Cytognos (Salamanca, Spain). Anti-sLex mAb CSLEX1 (IgM) was obtained from hybridoma HB-8580, American Type Culture Collection (ATCC, Rockville, MD, USA). Anti-CD18-activating mAb KIM185 (IgG1) was provided by Dr. Martyn Robinson (Celltech Therapeutics, Berkshire, UK) [28]; anti-CD18-blocking mAb TS1/18 [29] was used as mouse ascites and the hybridoma was obtained from ATCC; mAb anti-CD18 MEM-48 (IgG1) [30] and anti-CD11b MEM-170 were kindly provided by Dr.Václav Horejsi (Prague,Czech Republic). Other antibodies used were rabbit anti-human von Willebrand factor (DAKO, Glostrup, Denmark), mouse mAbs anti-CD34 (IgG1)(DAKO), anti-human CD31 (PECAM-1) (IgG1) (Novocastra Lab. Ltd., Newcastle, UK), and anti-human smooth muscle actin (IgG2a) (DAKO). In control experiments, normal rabbit serum or different isotype-matched control mouse antibodies (Sigma, St. Louis, MI, USA) were used.
Cell Cultures
Human umbilical vein endothelial cells (HUVEC) were isolated from cord segments from normal women with negative serology for Hepatitis B and C, HIV and CMV, and submitted to cesarean for medical reasons. The umbilical cords use for this research was authorized by the Institutional Review Boards of the Hospital Naval Pedro Mallo and the Hospital Rivadavia, Buenos Aires, Argentina, and the patients gave informed consent. Umbilical cords were treated with 0.5 mg/ml collagenase according to Jaffé et al. [31]. Primary cultures were grown in RPMI-1640 medium supplemented with 20% heat-inactivated AB human serum, 100 U/ml penicillin, 100 μg/ml streptomycin, 2 mM L-glutamine, 100 U/ml heparin, and 150 μg/ml endothelial cell growth supplement (Sigma) in tissue culture flasks (25 cm2). HUVEC were typically selected for experimental use at passages 2-4. In most experiments, monolayers were pretreated with different concentrations (1-10 μg/ml) of bacterial lipopolysaccharide (LPS, Sigma,) for 4 h at 37°C, to induce expression of adhesion molecules.
The human breast cancer cell line MCF-7 [32] was grown in Dulbecco's modified-Eagle's medium (DMEM)/Ham's nutrient mixture F-12 (1:1) supplemented with 10% fetal bovine serum, 2 mM glutamine, 10 μg/ml insulin, 100 U/ml penicillin, and 100 μg/ml streptomycin. Exponentially growing cells were harvested by treatment with 0.25% trypsin-0.038% EDTA.
Isolation of PMN
PMN were obtained from fresh human blood of normal volunteers, who gave informed consent. Briefly, heparinized whole blood was fractionated by dextran sedimentation, and the upper fraction was submitted to Ficoll-Hypaque gradient centrifugation. PMN were recovered from the pellet, and the remaining erythrocytes were lysed in 0.1 mM EDTA, 10 mM KCO3H, 155 mM NH4Cl during 10 min. After washing in PBS, PMN (purity: 93%) were resuspended in Hanks' balanced salt solution and kept on ice until used.
Immunofluorescence
HUVEC were grown on untreated cover slides (diameter: 12 mm) placed in 24-well microplates. In some experiments, PMN or MCF-7 were adhered to HUVEC and immunostaining was performed after fixation in 3% paraformaldehyde. When necessary, permeabilization was carried out with 0.05% saponin in PBS. HUVEC characterization was performed using the following primary antibodies: polyclonal antibody anti-von Willebrand factor, anti-CD34, and anti-muscle α-actin. Normal rabbit serum or different isotype-matched mouse immunoglobulins were used as controls. Secondary antibodies were Cy2-conjugated goat anti-rabbit IgG (DAKO), R-phycoerythrin-conjugated F(ab′)2 fragment of goat anti-mouse immunoglobulins (DAKO) or FITC-conjugated F(ab')2 fragment of goat anti-mouse immunoglobulins (DAKO).
Flow cytometry assay
PMN, HUVEC or MCF-7 cells were incubated with mAb anti-Lex FC-2.15, anti-Lex MCS-1 or anti-sLex CSLEX1 (50 μg/ml) for 1 h at 0°C, washed three times with PBS, and incubated for 1 h at 0°C with FITC-conjugated F(ab′)2 fragment of goat anti-mouse immunoglobulins (DAKO). Finally, cells were washed three times with PBS and fixed with 3% paraformaldehyde. Lex expression data were acquired on a fluorescence-activated cell sorter (FACScan, Becton Dickinson, Mountain View, CA, USA) and analyzed by using CellQuest software. In control experiments, isotype-matched mouse immunoglobulins were used instead of primary antibodies.
Adhesion of MCF-7 to HUVEC
HUVEC (104/well) were seeded into 96-well microplates precoated with 1% gelatin and incubated for 2 days. After removing growth medium, fresh medium (100 μl/well) was added containing different concentrations of LPS (1-10 μg/ml), and plates were incubated at 37°C for 4 h in a CO2 incubator. The medium was gently removed and replaced with fresh medium (150 μl/well). MCF-7 cells were previously labeled with 3H-thymidine [3H-dT] (1 μCi/ml; sp. act: 20 Ci/mmol) (Perkin Elmer, Life Sci. Inc., Boston, MA, USA) for 24 h, detached with EDTA, and washed twice with PBS. [3H] labeled-MCF-7 cells (2.5- 20 ×104 cells/well) were added to each well, and incubated at 37°C for 30 min in a CO2 incubator. Unbound cells were washed twice with PBS (200 μl/well). Bound cells were lysed with 0.2 N NaOH (200 μl/well); the medium from each well was collected and filtered through GFA papers (Whatman, Middlesex, UK), and the radioactivity was counted in a beta counter (Wallac, Pharmacia). For neuraminidase treatment, MCF-7 cells were digested with neuraminidase from Clostridium perfringens (5 U/ml) in 50 mM sodium citrate, pH 4.5 for 1 h at 37°C.
Adhesion of PMN to HUVEC
Assays were measured by the myeloperoxidase method [33]. Exponentially growing HUVEC (104/well) were cultured to confluence on 96-well microplates precoated with 1% gelatin (Sigma) and incubated for 2 days. After removing growth medium, fresh medium (100 μl/well) containing various concentrations of LPS (1-10 μg/ml) were added and incubated at 37°C for 4 h in a CO2 incubator. PMN (25 μl/well) were added to the wells and the plate was incubated for 5 min at 37°C in a CO2 incubator. Unbound cells were removed by washing three times in PBS containing 2% fetal bovine serum (200 μl/well). Citrate buffer (0.1 M, pH 4.5) containing 0.1% Nonidet P-40 (100 μl/well) was added to each well. After 10 min, a 2-fold o-phenylenediamine (OPDA, Sigma) solution (8 mg OPDA, 8 μl of H2O2 in 8 ml citrate buffer) (100 μl/well) was added to each well. After incubation at room temperature for 30 min, 4 N H2SO4 (100 μl/well) was added to stop the reaction, and OD450nm was measured. For blocking experiments, LPS-treated HUVEC were incubated with different concentrations of mAbs (100 μl/well) for 1 h at 37°C in a CO2 incubator; then, PMN resuspended in each mAb were added to the corresponding well. Isotype-matched irrelevant antibodies or PBS were used as controls. For neuraminidase treatment, PMN were digested with neuraminidase from Clostridium perfringens (5 U/ml) in 50 mM sodium citrate, pH 4.5 for 1 h at 37°C.
Complement-dependent cytotoxicity (CDC)
A 51Cr-release assay was used to test the ability of FC-2.15 to mediate complement-dependent cytotoxicity. Experiments were performed on isolated target cells (PMN or MCF-7) or on target cells adhered to monolayers of HUVEC. For experiments with isolated target cells, PMN or MCF-7 cells (2.5 × 106/ml) were labeled with 51Cr (Na251CrO4, sp. act.: 445.75 mCi/mg; 150 μCi/ml; Perkin Elmer, Boston, MA, USA) for 2 h at 37°C, and washed three times with PBS. 51Cr-labeled cells (105) in 100 μl culture medium plus 100 μl human serum as the source of complement were incubated with mAb FC-2.15 (0, 2.5, 10, 20 μg/ml) for different times at 37°C [27]. Parallel incubations containing target cells resuspended in 200 μl medium alone or in 200 μl 0.1 M HCl were performed to determine spontaneous and total 51Cr release, respectively. At the end of the incubation period, the cells were centrifuged, and the supernatant radioactivity was determined in a gamma counter. Negative control experiments were performed with mouse IgM (20 μg/ml, DAKO) instead of FC-2.15. Each point was assayed in triplicate and the percentage of specific cytolysis was calculated with the formula:
To study FC-2.15-mediated CDC on 51Cr-labeled PMN or MCF-7 adhered to HUVEC monolayers, unlabeled HUVEC (104/well) were incubated in 96-well microplates coated with 1% gelatin for 2 days, and they were stimulated with LPS (1 μg/ml) for 4 h at 37°C. Isolated 51Cr-labeled PMN (5 × 105/well) or 51Cr-labeled MCF-7 cells (5 × 104/well) were loaded on unlabeled HUVEC monolayers, and adhesion was conducted for 30 min at 37°C. After adhesion, the wells were washed twice to eliminate unbound cells, and 100 μl of FC-2.15 (0-20 μg/ml) plus 100 μl of human serum as C′ source were added. Incubations were performed for 90 min at 37°C and the percentage of specific cytolysis was determined as described above.
To assess the lytic effect of bound cells undergoing FC-2.15-dependent CDC on HUVEC monolayers, HUVEC (104/well) were cultured in 96-well microplates to confluence, and then stimulated with LPS (1 μg/ml) for 4 h at 37°C. Each HUVEC monolayer was labeled with 51Cr (5 μl/well) for 2 h at 37°C, and washed three times with PBS. 51Cr-labeled HUVEC were incubated with unlabeled PMN or unlabeled MCF-7 cells for 30 min at 37°C. After adhesion, unbound cells were washed twice with PBS, and 100 μl of FC-2.15 (20 μg/ml) plus 100 μl of human serum were added. Incubations were performed for 90 min at 37°C. Parallel incubations in medium alone or in 0.1 M HCl were performed to determine spontaneous and total 51Cr release, respectively. Negative control experiments were performed with mouse IgM (20 μg/ml, DAKO) instead of FC-2.15. Each point was assayed in triplicate, and the specific cytolysis percentage was calculated as described above. When indicated, untreated PMN or fMLP-activated PMN (10−7 M fMLP) were tested for FC-2.15-dependent CDC on HUVEC monolayers. Cytotoxicity on MCF-7 and PMN in suspension mediated by anti-Lex mAbs (FC-2.15 or MCS-1) plus C′ was evaluated by the MTT assay. This method is based on the reduction of the tetrazolium salt MTT into a dark blue formazan crystal which production is proportional to the number of viable cells. Control cells were treated with human serum as a source of C′ in the absence of anti-Lex mAbs. Treated cells were incubated in the presence of anti-Lex mAbs plus C′. Briefly, MTT (2.5 mg/ml) was added to the cells and incubated 4 h at 37°C. Cells were centrifuged, supernatants were discarded, and the colored formazan crystals were dissolved in 0.01N H2>SO4. Absorbance (A) was read in a microplate reader at 540 nm using absorbance at 630 nm as a reference. Data expressed as lysis percentages were calculated with the formula: %Lysis = 100% - [A value of sample/A value of control] × 100 [34].
In vitro aggregation assay
Suspensions of PMN (1.5 × 106 cells/ml) were incubated for 1 h at 37°C with gentle agitation in the presence of different concentrations of FC-2.15. Samples were observed under a phase contrast microscope (Axioplan, Carl Zeiss, Göttingen, Germany). The percentage of aggregated PMN was determined as: (aggregated/non-aggregated × 100). To analyze the kinetics of FC-2.15 induced PMN aggregation, samples were taken at different times, fixed with 4% formaldehyde, and the percentage of aggregated PMN was determined as above.
In other experiments, aggregation assays were performed by incubating PMN under different conditions such as 4°C, 0.04% sodium azide, 25 mM 2-deoxy-D-glucose (2dGlc) (4 min, at 37°C), 1 mM iodoacetic acid (IAA), or fixation in 4% formaldehyde (10 min). PMN aggregation induced by fMLP (4 × 10−7M, Sigma) or by the activating mAb against human β2 integrin KIM185 (20 μg/ml) were also determined. When indicated, a preincubation of PMN with the blocking mAb against human β2 integrin TS1/18 (ascites, 1/75) during 1 h at 37°C was performed, and this mAb was maintained during the aggregation assay in the presence of FC-2.15 or KIM185 (both at 20 μg/ml).
Western blots
Membrane extracts were prepared as follows. The cells were homogenized in 10 mM Tris-HCl, 1.5 mM EDTA, 1 mM PMSF, pH 7.4 with a polytron (IKA Werk, Germany). After ultracentrifugation (100,000 × g, 1 h) of crude extracts, membrane pellets were solubilized in NP-40 buffer (1 % NP-40, 0.1 M Tris-HCl pH 8.2, 0.15 M NaCl, 5 μg/ml pepstatin A, 10 μg/ml aprotinin, 5 μg/ml leupeptin, 1 mM PMSF) for 30 min on ice. Insoluble material was removed by centrifugation, and the supernatant was used for further work. Protein samples were run in SDS-PAGE and transferred to nitrocellulose membrane (Sigma, 0.45 μm). After blocking with 3% dried skim milk in PBS for 1 h, blots were incubated with primary antibodies (overnight at 4°C), washed in 0.05% Tween 20-PBS, and developed using alkaline phosphatase-conjugated goat anti-mouse antibodies (Jackson Immuno Research, West Grove, PA, USA). Finally, blots were visualized using nitroblue tetrazolium/5-Bromo-4-chloro-3-indolylphosphate (Promega, Madison, WI, USA).
Immunoprecipitation
Eight hundred μg of 1% NP-40 membrane fraction preparation were used for each immunoprecipitation and incubated with 6 μg of mAb MEM-48, MEM-170, or isotype-matched mouse immunoglobulins overnight at 4°C. Complexes were bound to Protein G-Sepharose (Amersham Pharmacia Biotech AB, Uppsala, Sweeden) for 2 h at 4°C. After washing with NP-40 buffer, immunoprecipitates were eluted under non-reducing conditions with SDS-PAGE sample buffer, run in 7.5% SDS-PAGE and blotted onto nitrocelullose. Western blots were developed with MEM-48, MEM-170, FC-2.15 mAbs or with isotype-matched mouse immunoglobulins (20 μg/ml).
Blotting with the scavenger receptor C-type lectin (SRCL)
The trimeric extracellular domain of human SRCL was prepared and labeled as previously described [23]. Membrane extracts from MCF-7 cells were prepared as described before, separated on 17.5% gels which were transferred to nitrocellulose and probed with the labeled receptor following the procedure previously used to study purified glycoproteins [23].
RESULTS
Comparative expression of Lex in MCF-7, PMN, and HUVEC
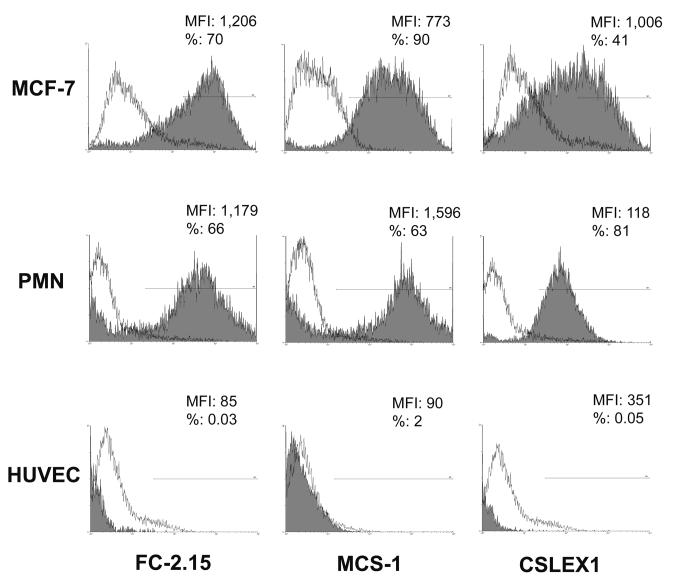
To characterize our experimental model, we first investigated by immunofluorescence if Lex was expressed in HUVEC. Primary cultures of HUVEC grew as monolayers with a cobblestone-like morphology, high levels of von Willebrand factor (vWF) and lack of Lex epitope expression (Figure 1a); PMN were added to HUVEC to serve as positive controls of FC-2.15 reactivity (Figure 1b), as previously demonstrated [27]. Flow cytometry studies with mAbs FC-2.15 and MCS-1, or CSLEX1, respectively, confirmed the lack of Lex and sLex expression in HUVEC (Figure 2) [35]. On the contrary, MCF-7 and PMN showed high levels of Lex and sLex surface expression (Figure 2).
Figure 1. HUVEC express von Willebrand factor but not Lex epitopes.
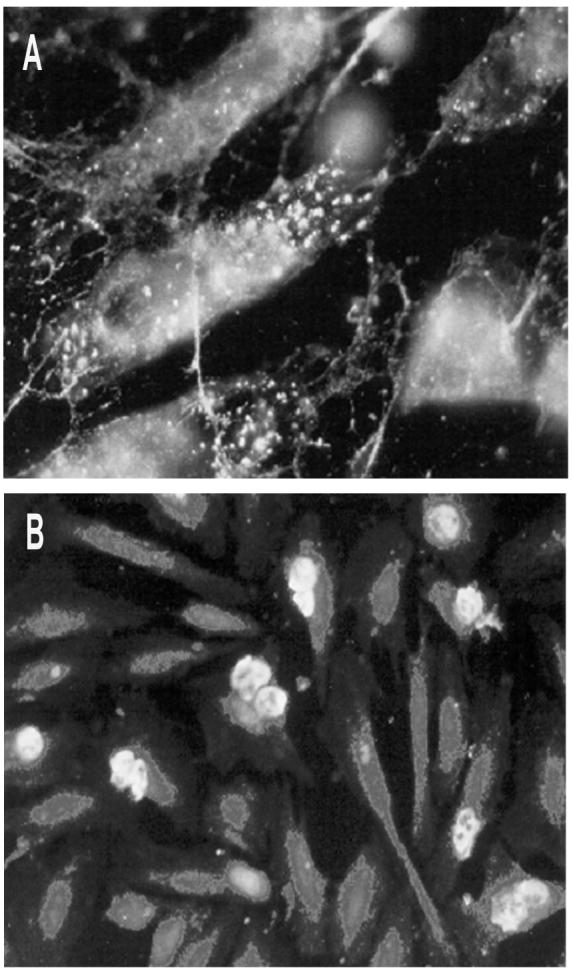
a) Immunofluorescence study of HUVEC endothelial cells immunostained with anti-von Willebrand factor polyclonal antibodies to confirm endothelial phenotype. Cells exhibit a homogeneous staining pattern localized in endothelial-specific organelles called Weibel-Palade bodies. Magnification: × 400. b) Immunofluorescence study performed on PMN adhered to HUVEC monolayers with mAb FC-2.15. A high level of Lex expression can be seen on PMN, while HUVEC did not stain. Magnification: × 200.
Figure 2. Expression of Lex epitopes in MCF-7 breast carcinoma cells, PMN, and HUVEC.
Lex expression was analyzed by flow cytometry using two mAbs against nonsialylated-Lex, FC-2.15 and MCS-1, and a mAb anti-sialylated-Lex, CSLEX1, followed by incubation with FITC-conjugated goat anti-mouse Ig. Data are taken from one representative experiment. Filled peaks represent staining with the primary anti-Lex or anti-sLex antibodies followed by FITC-secondary antibody. Staining by isotype-matched mouse immunoglobulins is indicated in white (each non-specific isotype control followed by FITC-conjugated goat anti-mouse Ig). Data are represented as histograms of fluorescence intensity for 10,000 light scatter-gated events. Median values of fluorescence intensity (MFI) are indicated in each figure.
Adhesion of MCF-7 cells and PMN to HUVEC. Comparative effects of antibodies anti-Lex
Since the mechanism of PMN adhesion to human endothelia involves both sugar-ligand interaction (selectin-mediated rolling) and integrin activation (strong adhesion), we tested if Lex plays a role in the adhesion of both MCF-7 tumor cells and PMN to LPS-activated or non-activated HUVEC. To establish the optimal experimental setting, the effect of HUVEC activation on PMN adhesion was first measured. The adhesion of PMN to HUVEC augmented after LPS activation, attaining a maximum increase of three-fold at 1 μg/ml LPS (Figure 3b). Similarly, MCF-7 cells binding to non-activated and LPS-activated HUVEC was measured, and as shown in Figure 3a the percentages of cell binding were 60% and 80%, respectively (Figure 3a). LPS did not increase binding of MCF-7 cells to endothelial cells to an appreciable extent (Figure 3a, lower panel) when compared to PMN binding to LPS-activated HUVEC (Figure 3b). However, since PMN binding was increased after LPS-treatment, it seemed reasonable to maintain LPS activation in all experiments performed thereafter. To investigate the role of Lex epitopes on this adhesion, we incubated MCF-7 cells (5 × 104 cells/well) in the presence of two different anti-Lex mAbs, FC-2.15 (IgM) and MCS-1 (IgG3). Adhesion was blocked by approximately ∼57% (remaining adhesion: 43 ± 8.5%) in the presence of 50 μg/ml anti-Lex MCS-1, whereas no inhibition was observed with FC-2.15, again at 50 μg/ml (Figure 3c). When the effects of anti- Lex mAbs on PMN adhesion to HUVEC were measured, it was also found that only MCS-1 significantly reduced (∼63-95%) the level of PMN adhesion, whereas no inhibition was observed with FC-2.15 (Figure 3d). The anti-sLex mAb CSLEX1 was also tested, and an inhibition of ∼57% was found at 50 μg/ml (remaining adhesion: 43 ± 9.8%). To confirm the role of sialic acid residues on PMN adhesion to HUVEC, assays were performed after PMN treatment with neuraminidase. In Figure 3e, it may be observed that neuraminidase treatment inhibited PMN adhesion. For instance, when 2 × 105 PMN/well were loaded, adhesion was inhibited by approximately ∼45% in neuraminidase-treated as compared to control PMN (Figure 3e). Therefore, in spite of the removal of sialic acid by neuraminidase treatment, ∼55 % of PMN adhesion to HUVEC is still retained. This residual adhesion might be due to integrin-dependent mechanisms and/or to the existence of other interactions such as those mediated by Lex determinants, since low affinity binding of Lex-residues to E-selectin has been previously demonstrated [36]. Regarding sLex mediated-MCF-7 binding to HUVEC, although mAb CSLEX1 did not block adhesion (data not shown), neuraminidase treatment of MCF-7 cells partially diminished this binding (42 ± 10%) (Figure 3c, right panel), which might indicate that the selectin pathway could also participate in this adhesion.
Figure 3. Effect of anti-Lex mAbs on adhesion of MCF-7 cells and PMN to activated and non-activated HUVEC.
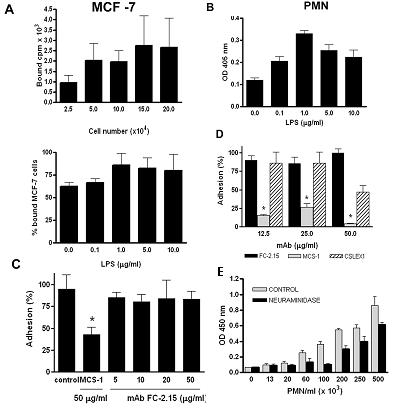
a) Adhesion assay to HUVEC was performed with different numbers of [3H] labeled MCF-7 cells (5×104 loaded cells corresponded to 20,020 cpm) (upper panel). In the lower panel, HUVEC were activated with 0, 0.1, 1, 5, and 10 μg/ml of LPS, before incubation with [3H] labeled MCF-7 cells (5×104 loaded cells corresponded to 22,619 cpm). Data shown are mean of bound cpm ± SD of triplicate determinations in three independent experiments. b) A PMN adhesion assay was performed by the myeloperoxidase technique as described under Methods. HUVEC were previously activated with 0, 0.1, 1, 5, and 10 μg/ml LPS. Paired Student's t test, P<0.05. Data shown are mean ± SD of triplicate determinations in three independent experiments. c) Adhesion assay was performed with [3H] labeled MCF-7. Left panel: Incubations were performed in the presence of PBS (control), MCS-1 (50 μg/ml), and FC-2.15 (5, 10, 20, and 50 μg/ml) (5×104 loaded cells corresponded to 58,114 cpm). Paired Student's t test, P<0.05. Data shown are mean + SD of triplicate determinations in three independent experiments. Right panel: Adhesion assay was performed with [3H] labeled MCF-7 digested with neuraminidase (NANAse) or not (control) (5×104 loaded cells corresponded to 11,210 cpm). d) PMN adhesion to HUVEC was assayed in the presence of the following mAbs: FC-2.15, MCS-1, and CSLEX1 at 12.5, 25, 50 μg/ml. Paired Student's t test, P<0.05. e) PMN adhesion to activated HUVEC was assayed after PMN treatment with 5 U/ml of neuraminidase from Clostridium perfringens or with PBS (control). X-axis represents the number of PMN/ml. Paired Student's t test, P<0.05. Data shown are mean ± SD of triplicate determinations in three independent .
FC-2.15 antibody mediates complement-dependent cytotoxicity (CDC) on MCF-7-cells and PMN. Comparative effects on adhered and isolated cells
To test the ability of FC-2.15 to mediate CDC on MCF-7, PMN and HUVEC, 51Cr-release assays were performed at different FC-2.15 concentrations and incubation times (30, 60, and 90 min). Incubations of 90 min gave the highest cytolysis percentages (data not shown), so all the following experiments were performed at this incubation time. For MCF-7 cells in suspension, the lysis percentages obtained were 84.5 ± 3.8%, 64.0 ± 16.4%, 29.75 ± 0.1% for FC-2.15 concentrations of 20, 10, and 2.5 μg/ml, respectively (Figure 4a). The lysis of control IgM-treated (20 μg/ml) MCF-7 cells in suspension was 3.6 ± 1.9% (data not shown). For FC-2.15 concentrations of 20, 10, and 2.5 μg/ml, the lysis of PMN in suspension were 97 ± 4.2%, 89.5 ± 14.6%, and 59.8 ± 11.7 %, respectively (Figure 4a). The lysis of control IgM-treated (20 μg/ml) PMN was 8.9 ± 0.3% (data not shown). Therefore, low concentrations of FC-2.15 (2.5 μg/ml) produced higher levels of cytolysis on PMN than MCF-7 cells when both were in suspension.
Figure 4. Complement-dependent cytotoxicity (CDC) of 51Cr-MCF-7 and 51Cr-PMN.
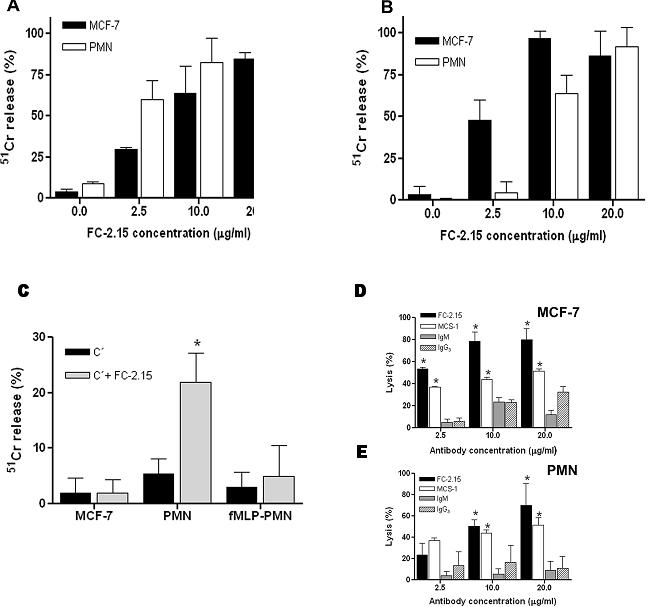
a) 51Cr-release assay was used to test the ability of mAb FC-2.15 (0, 2.5, 10, 20 μg/ml) to mediate CDC on 51Cr-MCF-7 and 51Cr-PMN in suspension. b) 51Cr-release assay was used to test the ability of mAb FC-2.15 (0, 2.5, 10, 20 μg/ml) to mediate CDC on 51Cr-MCF-7 and 51Cr-PMN adhered to HUVEC. c) Evaluation of the lytic effect on 51Cr-HUVEC of bound cells undergoing FC-2.15-mediated CDC. 51Cr-release assay was used to test the ability of complement alone (C') or FC-2.15 (20 μg/ml) plus complement (FC-2.15 + C') to mediate CDC when unlabeled PMN, unlabeled fMLP-activated PMN, and unlabeled MCF-7 are adhered to 51Cr-HUVEC. Data shown are mean ± SD of triplicate determinations in three independent experiments. Paired Student's t test, P<0.05. d) MCS-1 mediates CDC: MTT assay was performed on MCF-7 in suspension after treatment with mAbs FC-2.15 or MCS-1 (2.5, 10, 20 μg/ml) in the presence of C'. Control isotype-matched immunoglobulins used were IgM and IgG3. Paired Student's t test, P<0.05. Data shown are mean ± SD of triplicate determinations in three independent experiments. e) MCS-1 mediates CDC: MTT assay was performed on PMN in suspension after treatment with mAbs FC-2.15 or MCS-1 (2.5, 10, 20 μg/ml) in the presence of C'. Control isotype-matched immunoglobulins used were IgM and IgG3. Paired Student's t test, P<0.05. Data shown are mean ± SD of triplicate determinations in three independent experiments.
Afterwards, we studied FC-2.15-mediated CDC of MCF-7 or PMN once adhered to HUVEC monolayers, for which unlabeled HUVEC were incubated with 51Cr-labeled MCF-7 cells or PMN. The opposite situation to that observed with suspended cells was found, since FC-2.15 produced a marked cytolysis (∼50%) of adherent MCF-7 starting at 2.5 μg/ml and attaining ∼90-100% lysis at 10-20 μg/ml (Figure 4b). Lysis of adherent PMN was only observed at 10 and 20 μg/ml (∼60% and ∼90%, respectively). To summarize, when suspended cells were analyzed, PMN were more sensitive than MCF-7 cells to CDC mediated by low concentrations (2.5 μg/ml) of FC-2.15 while the opposite was observed for adherent cells.
Whether the CDC lysis of adhered cells had any deleterious effect on underlying HUVEC was also evaluated by labeling HUVEC monolayers grown in 96-well microplates with 51Cr, before the addition of unlabeled target cells. When 51Cr-labeled HUVEC were incubated with FC-2.15 (20 μg/ml) and C′ in the absence of PMN or MCF-7, insignificant levels of HUVEC cytolysis were observed (data not shown). In the presence of unlabeled MCF-7 adhering to 51Cr-labeled HUVEC, no lytic effects on HUVEC were observed (Figure 4c) in spite of the fact that adherent cells were undergoing FC-2.15-dependent CDC (Figure 4b). On the contrary, when PMN adhered to 51Cr-labeled HUVEC underwent CDC, ∼20% 51Cr-release from HUVEC was observed, which was statistically significant (P<0.05, Figure 4c).
Surprisingly, fMLP-activated adherent PMN showed no lytic effects on 51Cr-labeled HUVEC as compared to adherent non-activated PMN, both undergoing FC-2.15-dependent CDC. In summary, there was no damage to HUVEC in the presence of adherent MCF-7 undergoing CDC and the injury resulted to be not severe in the presence of PMN undergoing CDC.
MCS-1 antibody can mediate CDC
To investigate if the FC-2.15-mediated CDC was shared by other anti-Lex mAbs, isolated MCF-7 cells and PMN in suspension were incubated with different concentrations of MCS-1 and FC-2.15 or the corresponding isotype-matched immunoglobulins (IgG3 and IgM, respectively) plus C′. FC-2.15 produced stronger lysis of MCF-7 cells than MCS-1 (Figure 4d), although the lytic effect on PMN was similar (Figure 4e).
FC-2.15 antibody aggregates PMN but MCS-1 antibody does not
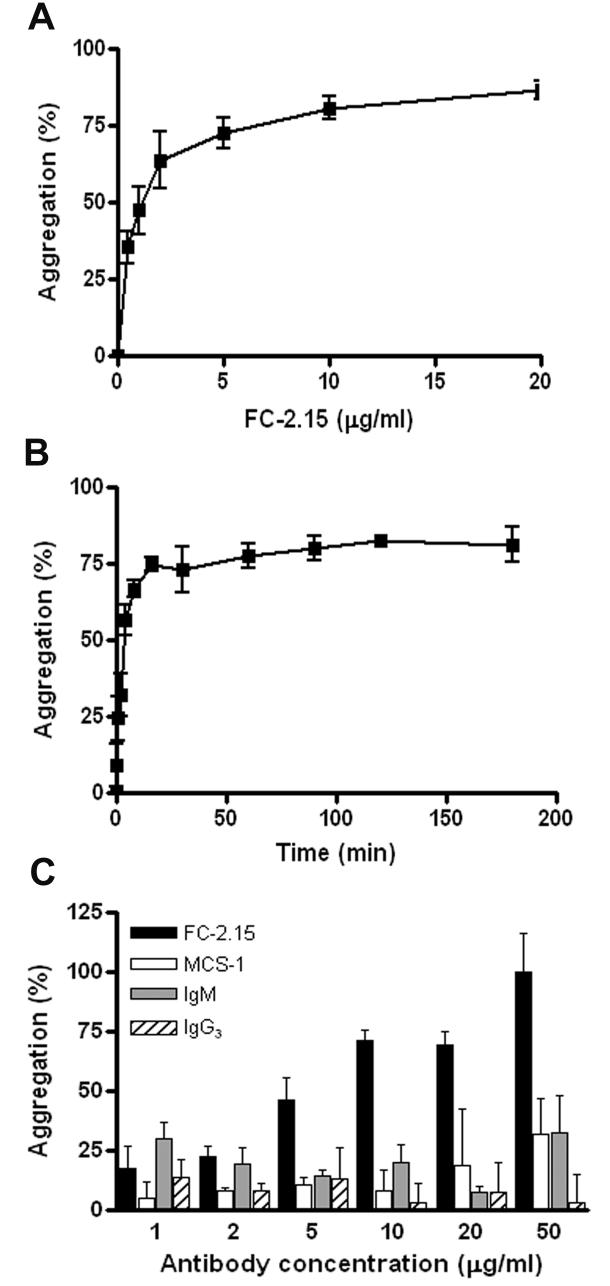
We have previously reported that FC-2.15 induces PMN homotypic aggregation, which could be a cause of the neutropenia observed in clinical trials [26]. In order to disclose if PMN aggregation only required recognition of Lex epitopes or if other factors such as multivalency of IgM or integrins activation were also involved, we performed further experiments. First, different FC-2.15 concentrations (0-20 μg/ml) and incubation periods were tested. FC-2.15 (20 μg/ml) induced a strong and rapid PMN aggregation, with only 25% PMN remaining isolated (Figure 5a). Time-course experiments performed at a saturating concentration of FC-2.15 (20 μg/ml) demonstrated that aggregation was already detected after 30 sec, and maximal effects were obtained after 15 min (Figure 5b). When we tested whether aggregation only depends on Lex-specificity, we found that was not the case since aggregation was not induced by another anti-Lex mAb MCS-1 (IgG3) (Figure 5c). Therefore, additional factors to Lex specificity were necessary. Other experiments performed demonstrated that this aggregation did not require energy, since it was not modified by incubation at 4°C, or with 0.04% sodium azide, 1 mM iodoacetic acid and 25 mM 2-deoxy-D-glucose (data not shown). However, fixation in formaldehyde inhibited aggregation (data not shown). Taken together, these findings suggest that aggregation requires a conformational flexibility of the cell membrane but does not require energy.
Figure 5. In vitro FC-2.15-induced PMN aggregation exhibits dose- and time-dependence.
Suspensions of PMN were incubated in different conditions: a) with different FC-2.15 concentrations for 1 h at 37°C; b) with 20 μg/ml FC-2.15 during different incubation times at 37°C. c) PMN aggregation was evaluated in the presence of different concentrations of FC-2.15, control IgM, MCS-1 or control IgG3. The percentage of aggregated cells was calculated for each condition as described under Methods. Determinations were performed by duplicate (bars: mean ± SD).
FC-2.15 produced PMN aggregation which was partially blocked by a β2 integrin inhibitor antibody
Since β2 integrin (CD11b/CD18) has been shown to be involved in PMN homotypic aggregation [37] and to contain Lex epitopes, the possibility was explored that FC-2.15 could bind to these Lex epitopes and induce the aggregation process due to its multivalency (absent in MCS-1). To explore this possibility, we compared FC-2.15-induced PMN aggregation to that induced by the β2 integrin-activator mAb KIM185. Both KIM185 and FC-2.15 produced PMN aggregation which was partially blocked by the β2 integrin-inhibitory mAb TS1/18 (Figure 6a). Therefore, β2 integrin activation pathway might be partially involved in FC-2.15-mediated PMN aggregation. Afterwards, we explored if both CD11b and CD18 integrins contained Lex oligosaccharides recognized by Lex mAbs as described [19-21]. For this purpose, immunoprecipitation of PMN membrane extracts with anti-CD11b MEM-170 and anti-CD18 MEM-48 followed by Western blots developed with FC-2.15, MEM-170, and MEM-48 were performed and confirmed the presence of Lex in both subunits of this integrin (Figure 6b). The band of 180 kD is CD11b and the one of 95kD is CD18. During immunoprecipitation with each anti-integrin subunit mAb, there was co-precipitation of the other integrin subunit. The CD11b band was more readly detected with mAb MEM-170 although a slight cross-reaction, presumably with CD18, was observed. mAb FC-2.15 recognizes both chains of the integrin, since they both carry Lex epitopes.
Figure 6. In vitro aggregation of PMN by FC-2.15 appears to be partially mediated by β2-integrins, which express Lex.
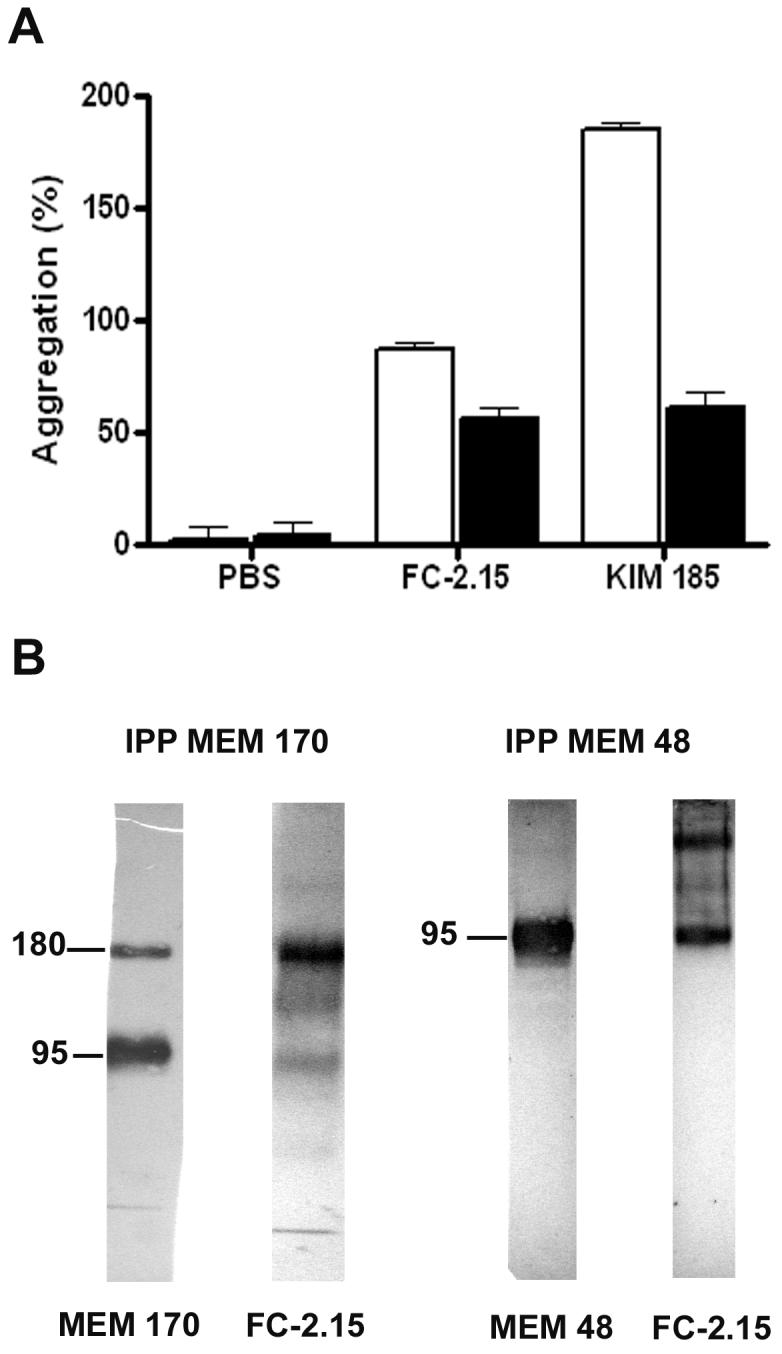
a) Isolated PMN were preincubated in the absence (control) or the presence of mAb TS1/18, a β2-integrin blocking antibody. Then, the agglutination assay was performed in the presence of FC-2.15 (20 μg/ml), KIM185 (20 μg/ml) or PBS (control). Each determination was performed in duplicates in two independent experiments (bars: mean ± SD). b) PMN membrane extracts were immunoprecipitated with either MEM-170 mAb, MEM 48 mAb or isotype-matched immunoglobulins (not shown), and immunoprecipitates were subjected to SDS-PAGE and immunoblotting with either MEM-170, MEM-48 or FC-2.15 mAbs as indicated.
The scavenger receptor C-type lectin may provide a mechanism for MCF-7 cell binding to HUVEC
The ability of MCS-1 mAb to inhibit MCF-7 binding to HUVEC confirmed the importance of the Lex epitope in this interaction. Since the scavenger receptor C-type lectin (SRCL) is found on endothelial cells and binds Lex with high selectivity, it is a strong candidate for mediating this tumor cell-endothelium interaction. In order to confirm the ability of SRCL to bind Lex on MCF-7 cells, 125I-labeled extracellular domain of the receptor was used to probe membrane proteins from MCF-7 cells. The results showed that the receptor bound efficiently two main glycoproteins, Mr ∼ 28 kD and 10 kD, in the cell membranes (Figure 7b). Thus, MCF-7 cells express Lex on specific surface glycoproteins. These low Mr glycoproteins that bind to the SRCL receptor were not detected by anti-Lex mAbs by immunoblotting (Figure 7a), probably reflecting differences in the preferred spacing and presentation of sugar epitopes for the antibodies and SRCL.
Figure 7. Glycoproteins of MCF-7 cells are bound by the scavenger receptor C-type lectin.
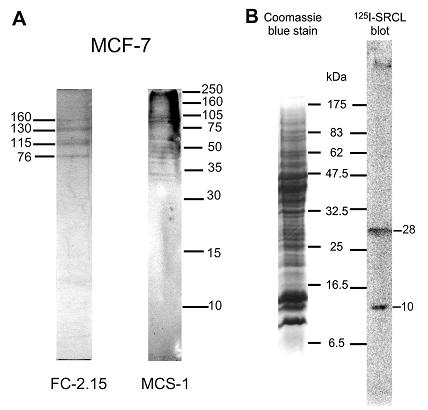
a) Immunoblotting analysis of MCF-7 cells glycoproteins containing Lex epitopes. Membrane extracts of MCF-7 cells were submitted to SDS-PAGE (15%) and immunoblotted with FC-2.15 and MCS-1 mAbs. b) Membrane extracts of MCF-7 cells were subjected to SDS-PAGE (17.5%) and parallel lanes were stained with Coomassie blue or blotted onto a nitrocellulose membrane that was probed with 125I-labeled extracellular domain of SRCL as described under Methods.
DISCUSSION
In this work, we have confirmed that MCF-7 cancer cells and PMN bind to HUVEC, as previously shown [33, 38]. Subsequently, we have demonstrated that the adhesion of both cell types to LPS-activated HUVEC was partially dependent on Lex epitopes. This conclusion was based on the fact that adhesion could be blocked by the anti-Lex mAb MCS-1, but not by FC-2.15, suggesting different specificities. Since both mAbs react with different glycoprotein species (Figure 7a), we suggest that MCS-1 recognizes molecules essential for adhesion, whereas FC-2.15 does not. Other authors also showed differential binding of anti-Lex mAbs [22, 39]. For instance, three anti-Lex mAbs which bind to granulocyte glycolipids have been studied, and their binding revealed differential reactivity [40]. Comparison of fine specificities between MCS-1 and FC-2.15 will require further studies, such as crystallographic analysis of each antibody in complex with its Lex antigen as reported by van Roon et al. [41]. The adhesive function of Lex, partially related to P-selectin binding [6], has also been demonstrated in Hodgkin's lymphoma-derived L428 cells.
In this paper, we also described that MCF-7 cells possess, among other glycoproteins, ligands for the endothelial scavenger receptor C-type lectin (SRCL) [23], which could mediate adhesion of cancer cells to vascular endothelium in a manner analogous to the leukocyte-endothelial interactions mediated by selectins. Further characterization of the specific ligands present in breast cancer MCF-7 cells remains to be performed. We then examined the effect of anti-Lex mAbs on lysis of tumor or normal cells. HUVEC did not suffer CDC in the presence of anti-Lex mAbs, as expected due to their undetectable Lex surface expression [35]. In this paper, we demonstrated that both anti-Lex mAbs efficiently lysed Lex+ MCF-7 cells in the presence of C′, both in suspension and adhered to HUVEC. Moreover, we have shown that the lysis of adherent MCF-7 did not damage endothelial cells. As it regards to PMN lysis, it was achieved by FC-2.15 and MCS-1, either in suspension as already described for FC-2.15 [27] or adhered to HUVEC. However, in the latter case, lysis of PMN by anti-Lex mAb induced a certain degree of HUVEC lysis, which might be due to the discharge of different enzymes present in PMN granules, whose potential clinical significance is unknown. Different anti-Lex antibodies have been used in the immunotherapy of hematological malignancies [42] and solid tumors [26]. When assayed in a Phase I clinical trial, FC-2.15 induced the remission of liver metastases in a breast cancer patient, albeit at the expense of a neutropenia which was rapidly reversible after the end of the infusion. We do not know if the observed neutropenia was due to PMN lysis, PMN aggregation or a combination of both mechanisms. Homotypic neutrophil aggregation mediated by anti-Lex mAb has already been observed by us [27] and other authors [43]. In this paper, we have demonstrated that it is induced by FC-2.15 (IgM) but not by MCS-1 (IgG3), which suggests that FC-2.15-mediated aggregation would depend on the combined effects of Lex-specificity and multivalency. A compounding factor could be that the PMN integrins CD18 and CD11b carry Lex epitopes, and anti-Lex mAbs binding could induce integrin activation and PMN aggregation. This assumption is supported by the fact that FC-2.15-mediated PMN aggregation was partially blocked by the β2 integrin inhibitor mAb TS1/18. If PMN aggregation contributes significantly to neutropenia, MCS-1, which does not induce it, could hold more therapeutic promise than FC-2.15.
The interaction between tumor cells and endothelium through Lex epitopes could provide a target for inhibition by mAbs such as those investigated in this study, and it could therefore have implications in the prevention of tumor cell metastasis. Although the release of cancer cells during surgery has not been extensively studied, due in great part to technical difficulties in identifying circulating tumor cells, in a few cases it has been documented. For instance, Wong et al. [44] have demonstrated that, after hepatocellular carcinoma surgery, tumor cells are detached and may spread hematogeneously. Similarly, in laparoscopic lobectomy, Yamashita et al. [45] have demonstrated that cancer cells were released into circulation. Based on these results, one could speculate that peri-surgical infusion of anti-Lex mAbs in patients submitted to surgery of Lex-expressing tumors, such as breast and colorectal carcinoma, could be useful to prevent tumor metastases, either by lysing tumor cells in circulation detached from large tumors, or by preventing their attachment to endothelia. In summary, MCS-1 would appear to be a better candidate for therapeutic purposes than FC-2.15, since it efficiently mediates Lex+-tumor cell CDC, it blocks binding of Lex+-cells to vascular endothelia and it does not induce PMN aggregation.
ACKNOWLEDGEMENTS
This work has been supported by grants from Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), Agencia Nacional de Promoción Científica y Tecnológica (ANPCyT), Fundación Sales, Fundación para la Investigación y Prevención del Cancer (FUCA), Fundación Pedro F. Mosoteguy, Fundación María Calderón de la Barca, Argentina, the Wellcome Trust and the Biotechnology and Biological Sciences Research Council. JM and MTE are members of CONICET. We are grateful to the Obstetric Department, Hospital Rivadavia, Argentina, for providing umbilical cords from normal volunteers. We also acknowledge the Hematology and Obstetric Departments, Hospital Naval Pedro Mallo, Argentina, for the provision of blood/human serum, and umbilical cords, respectively, from normal volunteers.
Abbreviations
- Ags
antigens
- C′
complement
- CD
Cluster of Differentiation
- CDC
complement-dependent cytotoxicity
- CEA
carcinoembryonic antigen
- CEA-CAM-1
carcinoembryonic antigen-related cell adhesion molecule 1
- DC
dendritic cell
- DC-SIGN
dendritic cell-specific [intercellular adhesion molecule]-3- grabbing nonintegrin
- 2dGlc
2-deoxy-D-glucose
- FACS
Fluorescence-activated cell sorting
- FITC
fluorescein isothiocyanate
- fMLP
N-formyl-methionyl-leucyl-phenylalanine
- HUVEC
human umbillical vein endothelial cells
- IAA
iodoacetic acid
- ICAM-3
intercellular adhesion molecule-3
- LPS
lipopolyssaccharide
- Lex
Lewis x; Galβ1,4(Fucα1,3)GlcNAc
- Lea
Lewis a, Galβ1,3(Fucα1,4)GlcNAc
- sLex
sialyl Lewis x, NeuAcα2-3Galβ1,4(Fucα1,3)GlcNAc
- mAb
monoclonal antibody
- PBS
phosphate buffered saline
- PMN
polymorphonuclear neutrophils
- PMSF
phenyl-methylsulfonyl-fluoride
- SRCL
scavenger receptor C-type lectin
- SSEA-1
stage specific embryonic antigen 1
REFERENCES
- 1.Gooi HC, Feizi T, Kapadia A, Knowles BB, Solter D, Evans MJ. Stage-specific embryonic antigen involves α1→3 fucosylated type 2 blood group chains. Nature. 1981;292:156–158. doi: 10.1038/292156a0. [DOI] [PubMed] [Google Scholar]
- 2.Hakomori S, Nudelman E, Levery S, Solter D, Knowles BB. The hapten structure of a developmentally regulated glycolipid antigen (SSEA-1) isolated from human erytrocytes and adenocarcinoma: a preliminary note. Biochem Biophys Res Commun. 1981;100:1578–1586. doi: 10.1016/0006-291x(81)90699-9. [DOI] [PubMed] [Google Scholar]
- 3.Spooncer E, Fukuda M, Klock JC, Oates JE, Dell A. Isolation and characterization of polyfucosylated lactosaminoglycan from human granulocytes. J Biol Chem. 1984;259:4792–4801. [PubMed] [Google Scholar]
- 4.Mordoh J, Leis S, Bravo AI, Podhajcer OL, Ballaré C, Capurro M, Kairiyama C, Bover L. Description of a new monoclonal antibody, FC-2.15, reactive with human breast cancer and other human neoplasia. Int J Biological Markers. 1994;9:125–134. doi: 10.1177/172460089400900301. [DOI] [PubMed] [Google Scholar]
- 5.Hittelet A, Camby I, Nagy N, Legendre H, Bronckart Y, Decaestecker C, Kaltner H, Nifant'ev NE, Bovin NV, Pector JC, Salmon I, Gabius HJ, Kiss R, Yeaton P. Binding sites for Lewis antigens are expressed by human colon cancer cells and negatively affect their migration. Lab Invest. 2003;83:777–787. doi: 10.1097/01.lab.0000073129.62433.39. [DOI] [PubMed] [Google Scholar]
- 6.Ohana-Malka O, Benharroch D, Isakov N, Prinsloo I, Shubinsky G, Sacks M, Gopas J. Selectins and anti-CD15 (Lewis x/a) antibodies transmit activation signals in Hodgkin's lymphoma-derived cell lines. Exp Hematol. 2003;31:1057–1065. doi: 10.1016/s0301-472x(03)00237-6. [DOI] [PubMed] [Google Scholar]
- 7.Rizzatti EG, Portieres FL, Martins SL, Rego EM, Zago MA, Falcao RP. Microgranular and t(11;17)/PLZF-RARalpha variants of acute promyelocytic leukemia also present the flow cytometric pattern of CD13, CD34 and CD15 expression characteristic of PML-RARalpha gene rearrangement. Am J Hematol. 2004;76:44–51. doi: 10.1002/ajh.20055. [DOI] [PubMed] [Google Scholar]
- 8.Johnston DS, Wright WW, Shaper JH, Hokke CH, Van den Eijnden DH, Joziasse DH. Murine sperm-zona binding, a fucosyl residue is required for a high affinity sperm-binding ligand. A second site on sperm binds a nonfucosylated, beta-galactosyl-capped oligosaccharide. J Biol Chem. 1998;273:1888–1895. doi: 10.1074/jbc.273.4.1888. [DOI] [PubMed] [Google Scholar]
- 9.D'Costa S, Petitte J-N. Characterization of stage-specific embryonic antigen-1 (SSEA-1) expression during early development of the turkey embryo. Int J Dev Biol. 1999;43:349–356. [PubMed] [Google Scholar]
- 10.Allendoerfer KL, Durairaj A, Matthews GA, Patterson PH. Morphological domains of Lewis X-FORSE 1 immunolabeling in the embryonic neural tube are due to developmental regulation of cell surface carbohydrate expression. Dev Biol. 1999;211:208–219. doi: 10.1006/dbio.1999.9308. [DOI] [PubMed] [Google Scholar]
- 11.Geijtenbeek TB, Van Vliet SJ, Engering A, Ba TH, Van Kooyk Y. Self- and non-self-recognition by C-type lectins on dendritic cells. Annu Rev Immunol. 2004;22:33–54. doi: 10.1146/annurev.immunol.22.012703.104558. [DOI] [PubMed] [Google Scholar]
- 12.van Gisbergen KP, Aarnoudse CA, Meijer GA, Geijtenbeek TB, van Kooyk Y. Dendritic Cells Recognize tumor-specific glycosylation of carcinoembryonic antigen on colorectal cancer cells through dendritic cell–specific intercellular adhesion molecule-3–grabbing nonintegrin. Cancer Res. 2005;65:5935–5944. doi: 10.1158/0008-5472.CAN-04-4140. [DOI] [PubMed] [Google Scholar]
- 13.Kannagi R, Isawa M, Koike T, Miyazaki K, Kimura N. Carbohydrate-mediated cell adhesion in cancer metastasis and angiogenesis. Cancer Sci. 2004;95:377–384. doi: 10.1111/j.1349-7006.2004.tb03219.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Vestweber D, Blanks JE. Mechanisms that regulate the function of the selectins and their ligands. Physiol Rev. 1999;79:181–213. doi: 10.1152/physrev.1999.79.1.181. [DOI] [PubMed] [Google Scholar]
- 15.Giavazzi R, Foppolo M, Dossi R, Remuzzi A. Rolling and adhesion of human tumor cells on vascular endothelium under physiological flow conditions. J Clin Invest. 1993;92:3038–3044. doi: 10.1172/JCI116928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kitayama J, Tsuno N, Sunami E, Osada T, Muto T, Nagawa H. E-selectin can mediate the arrest type of adhesion of colon cancer cells under physiological shear flow. Eur J Cancer. 2000;36:121–127. doi: 10.1016/s0959-8049(99)00228-2. [DOI] [PubMed] [Google Scholar]
- 17.Burdick MM, McCaffery JM, Kim YS, Bochner BS, Konstantopoulos K. Colon carcinoma cell glycolipids, integrins, and other glycoproteins mediate adhesion to HUVECs under flow. Am J Physiol Cell Physiol. 2003;284:C977–C987. doi: 10.1152/ajpcell.00423.2002. [DOI] [PubMed] [Google Scholar]
- 18.Mitoma J, Petryniak B, Hiraoka N, Yeh J-C, Lowe JB, Fukuda M. Extended core 1 and core 2 branched O-glycans differentially modulate sialyl Lewis x-type L-selectin ligand activity. J Biol Chem. 2003;278:9953–9961. doi: 10.1074/jbc.M212756200. [DOI] [PubMed] [Google Scholar]
- 19.Skubitz KM, Snook RW. Monoclonal antibodies that recognize lacto-N-fucopentaose III (CD15) react with the adhesion-promoting glycoprotein family (LFA-1/Hmac-1/gp150,95) and CR1 on human neutrophils. J Immunol. 1987;139:1631–1639. [PubMed] [Google Scholar]
- 20.Skubitz KM, Mendiola JR, Collett MS. CD15 monoclonal antibodies with a phosphotyrosine-containing protein on the surface of human neutrophils. J Immunol. 1988;141:4318–4323. [PubMed] [Google Scholar]
- 21.Albrechtsen M, Kerr MA. Characterization of human neutrophil glycoproteins expressing the CD15 differentiation antigen (3-fucosyl-N-acetyllactosamine) British J Haematol. 1989;72:312–320. doi: 10.1111/j.1365-2141.1989.tb07710.x. [DOI] [PubMed] [Google Scholar]
- 22.Lucka L, Fernando M, Grunow D, Kannicht C, Horst AK, Nollau P, Wagener C. Identification of Lewis x structures of the cell adhesion molecule CEACAM1 from human granulocytes. Glycobiology. 2005;15:87–100. doi: 10.1093/glycob/cwh139. [DOI] [PubMed] [Google Scholar]
- 23.Coombs PJ, Graham SA, Drickamer K, Taylor ME. Selective binding of the scavenger receptor C-type lectin to Lewisx trisaccharide and related glycan ligands. J Biol Chem. 2005;280:22993–22999. doi: 10.1074/jbc.M504197200. [DOI] [PubMed] [Google Scholar]
- 24.Ballaré C, Barrio M, Portela P, Mordoh J. Functional properties of FC-2.15, a monoclonal antibody that mediates human complement cytotoxicity against breast cancer cells. Cancer Immunol Immunother. 1995;41:15–22. doi: 10.1007/BF01788955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Capurro M, Bover L, Portela P, Livingston P, Mordoh J. FC-2.15, a monoclonal antibody active against human breast cancer, specifically recognizes Lewis X hapten. Cancer Immunol Immunother. 1998;45:334–339. doi: 10.1007/s002620050451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Mordoh J, Silva C, Albarellos M, Bravo AI, Kairiyama C. Phase I clinical trial in cancer patients of a new monoclonal antibody FC-2.15 reacting with tumor proliferating cells. J Immunother. 1995;17:151–160. doi: 10.1097/00002371-199504000-00004. [DOI] [PubMed] [Google Scholar]
- 27.Capurro M, Ballaré C, Bover L, Portela P, Mordoh J. Differential lytic and agglutinating activity of the anti-LewisX monoclonal antibody FC-2.15 on human polymorphonuclear neutrophils and MCF-7 breast tumor cells. In vitro and in vivo study. Cancer Immunol Immunother. 1999;48:100–108. doi: 10.1007/s002620050553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Robinson MK, Andrew D, Rosen H, Brown D, Stephens P, Butcher EC. Antibody against the Leu-CAM beta-chain (CD18) promotes both LFA-1- and CR3-dependent adhesion events. J Immunol. 1992;148:1080–1085. [PubMed] [Google Scholar]
- 29.Sánchez-Madrid F, Krensky AM, Ware CF, Robbins E, Strominger JL, Burakoff SJ, Springer TA. Three distinct antigens associated with human T-lymphocyte-mediated cytolysis: LFA-1, LFA-2 and LFA-3. Proc Natl Acad Sci USA. 1982;79:7489–7493. doi: 10.1073/pnas.79.23.7489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Drbal K, Angelisová P, Cerný J, Pavlistova D, Cebecauer M, Novak P, Horejsi V. Human leukocytes contain a large pool of free forms of CD18. Biochem Biophys Res Commun. 2000;275:295–299. doi: 10.1006/bbrc.2000.3299. [DOI] [PubMed] [Google Scholar]
- 31.Jaffé EA, Nachman RL, Becker CG, Minick CR. Culture of human endothelial cells derived from umbilical veins: identification by morphologic and immunologic criteria. J Clin Invest. 1973;52:2745–2756. doi: 10.1172/JCI107470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Soule HD, Vazguez J, Long A, Albert S, Brennan M. A human cell line from a pleural effusion derived from a breast carcinoma. J Natl Cancer Inst. 1973;51:1409–1416. doi: 10.1093/jnci/51.5.1409. [DOI] [PubMed] [Google Scholar]
- 33.Kawamura N, Imanishi N, Koike H, Nakahara H, Phillips L, Morooka S. Lipoteichoic acid-induced neutrophil adhesion via E-selectin to human umbilical vein endothelial cells (HUVECs) Biochem Biophys Res Commun. 1995;217:1208–1215. doi: 10.1006/bbrc.1995.2897. [DOI] [PubMed] [Google Scholar]
- 34.Liao J-W, Kang J-J, Jeng C-R, Chang S-K, Kuo M-J, Wang S-C, Liu MRS, Pang VF. Cartap-induced cytotoxicity in mouse C2C12 myoblast cell line and the roles of calcium ion and oxidative stress on the toxic effects. Toxicology. 2006;219:73–84. doi: 10.1016/j.tox.2005.11.002. [DOI] [PubMed] [Google Scholar]
- 35.Majuri M-L, Pinola M, Niemelä R, Tiisala S, Natunen J, Renkonen O, Renkonen R. α2,3-Sialyl and α1,3-fucosyltransferase-dependent synthesis of sialyl Lewis x, an essential oligosaccharide present on L-selectin counterreceptors, in culture endothelial cells. Eur J Immunol. 1994;24:3205–3210. doi: 10.1002/eji.1830241244. [DOI] [PubMed] [Google Scholar]
- 36.Phillips ML, Nudelman E, Gaeta FCA, Perez M, Singhal AK, Hakomori S-I, Paulson JC. ELAM-1 mediates cell adhesion by recognition of a carbohydrate ligand, sialyl-Lex. Science. 1990;250:1130–1132. doi: 10.1126/science.1701274. [DOI] [PubMed] [Google Scholar]
- 37.Buyon JP, Abramson SB, Philips MR, Slade SG, Ross GD, Weissman G, Winchester RJ. Dissociation between increased surface expression of gp165/95 and homotypic neutrophil aggregation. J Immunol. 1988;140:3156–3160. [PubMed] [Google Scholar]
- 38.Bild T, Jose J, Hartmann RW. Discovery of inhibitors of MCF-7 tumor cell adhesion to endothelial cells and investigation on their mode of action. Archiv der Pharmazie. 2004;337:687–694. doi: 10.1002/ardp.200400622. [DOI] [PubMed] [Google Scholar]
- 39.Harris JF, Chin J, Jewett MA, Kennedy M, Gorczynski RM. Monoclonal antibodies against SSEA-1 antigen: binding properties and inhibition of human natural killer cell activity against target cells bearing SSEA-1 antigen. J Immunol. 1984;132:2502–2509. [PubMed] [Google Scholar]
- 40.Magnani JL, Ball ED, Fanger MW, Hakomori S-I, Ginsburg V. Monoclonal antibodies PMN6, PMN 29, and PM-81 bind differently to glycolipids containing a sugar sequence ocurring in lacto-N-fucopentaose III. Arch Biochem Biophys. 1984;233:501–506. doi: 10.1016/0003-9861(84)90473-9. [DOI] [PubMed] [Google Scholar]
- 41.van Roon AM, Pannu NS, de Vrind JP, van der Marel GA, van Boom JH, Hokke CH, Deelder AM, Abrahams JP. Structure of an anti-Lewis X Fab fragment in complex with its Lewis X antigen. Structure. 2004;7:1227–1236. doi: 10.1016/j.str.2004.05.008. [DOI] [PubMed] [Google Scholar]
- 42.Ball ED, Selvaggi K, Hurd D, Herzig R, Clark L, Malley V, Persichetti J, deMagelhaus-Silverman M. Phase I clinical trial of serotherapy in patients with acute myeloid leukemia with an immunoglobulin M monoclonal antibody to CD15. Clin Cancer Res. 1995;1:965–972. [PubMed] [Google Scholar]
- 43.Stocks SC, Kerr MA. Stimulation of neutrophil adhesion by antibodies recognizing CD15 (Lex) and CD15-expressing carcinoembryonic antigen-related glycoprotein NCA-160. Biochem J. 1992;288:23–27. doi: 10.1042/bj2880023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wong IHN, Lau WY, Leung T, Yeo W, Johnson PJ. Hematogenous dissemination of hepatocytes and tumor cells after surgical resection of hepatocellular carcinoma: a quantitative analysis. Clin Can Res. 1999;5:4021–4027. [PubMed] [Google Scholar]
- 45.Yamashita JI, Kurusu Y, Fujino N, Saisyoji T, Ogawa M. Detection of circulating tumor cells in patients with non-small cell lung cancer undergoing lobectomy by video-assisted thoracic surgery: a potential hazard for intraoperative hematogenous tumor cell dissemination. J Thorac Cardiovasc Surg. 2000;119:899–905. doi: 10.1016/S0022-5223(00)70084-5. [DOI] [PubMed] [Google Scholar]