Abstract
This study investigated the basis for intrinsic amphotericin B (AMB) resistance in Aspergillus terreus. The ergosterol content, cell wall composition, and lipid peroxidation level had no influence on AMB resistance. The level of catalase production in A. terreus was significantly higher than that in A. fumigatus (P < 0.05). This higher-level production may contribute to AMB resistance in A. terreus since oxidative damage has been implicated in AMB action.
Invasive aspergillosis has emerged as a leading cause of morbidity and mortality in immunocompromised patients (7). Aspergillus terreus in particular is an amphotericin B (AMB)-resistant mold that has been recognized as a cause of frequently lethal infections (25). The exact mechanism of AMB action is still incompletely understood. Previous work has shown that AMB binds to the ergosterol in the fungal cell membrane; this step introduces aqueous pores into the lipid bilayers, and thus small ions leak out, causing a disruption of the proton gradient (24). Others have demonstrated AMB to cause cell death by oxidative damage (21). AMB is active against a wide range of fungi, yet certain species, such as A. terreus and Pseudallescheria boydii, are intrinsically resistant to AMB (7, 17, 22, 23). Manavathu et al. (14) generated AMB-resistant mutants of Aspergillus fumigatus by UV radiation. Alternatively, resistant isolates have been collected by repeated subculturing on agar containing increasing amounts of polyene compounds (24). It has been suggested previously that for Aspergillus flavus and Candida albicans, the ergosterol content, the composition of the fungal cell wall, and the ability to produce catalase may play a role in AMB resistance (15, 20, 21). So far, the reason for the in vitro and in vivo AMB resistance in A. terreus is not known. This study examined the basis for AMB resistance in A. terreus.
Clinical isolates of AMB-sensitive A. fumigatus (n = 10) and AMB-resistant A. terreus (n = 10) were used in this study. AMB susceptibility testing was performed according to the CLSI (formerly NCCLS) M38-A technique (16). In parallel, we prepared cell wall-free Aspergillus protoplasts that were AMB sensitive (MIC < 1 μg/ml) and resistant (MIC > 2 μg/ml); protoplast preparation, including the determination of the regeneration rate, was done according to a protocol of Hearn et al. (10). MIC testing with protoplasts was performed in RPMI 1640 containing 0.55 M sorbitol as a stabilizer. Our results provide evidence that AMB resistance in A. terreus is not associated with the cell wall, as MICs for conidia and cell wall-free fungi did not differ. The mean AMB MICs for conidia and protoplasts of A. fumigatus were 0.21 ± 0.06 and 0.19 ± 0.05 μg/mg, and those for conidia and protoplasts of A. terreus were 1.70 ± 0.58 and 1.41 ± 0.38 μg/mg, respectively.
Fungal ergosterol was analyzed by a previously published method with slight modifications (2, 4). Fifty milliliters of Sabouraud medium (Merck, Vienna, Austria) was inoculated with 106 conidia and incubated for 24 h with shaking (200 rpm) at 35°C. Subsequently, fungi were harvested by vacuum filtration, and the wet weight was determined; the pellet was minced with a blade, 6 ml of 25% alcoholic potassium hydroxide solution was added and mixed for 2 min, and the mixture was incubated at 85°C for 3 h. Sterols were extracted by adding 2 ml of sterile water and 6 ml of n-heptane, mixing for 4 min, and allowing the layers to separate for 2 h. The n-heptane layer was used for spectrophotometric analysis between 240 and 300 nm with a Beckmann spectrophotometer, and the ergosterol content was calculated as previously described (2). Overall, the data on fungal ergosterol contents and AMB resistance are contradictory: Sterling and Merz (22) and Dick et al. (9) showed that AMB resistance in yeasts is based on the absence or the reduction of fungal ergosterol; changes in the sterols and phospholipids affect membrane fluidity and, thus, decrease AMB impact (19). By contrast, Joseph-Horne et al. proposed non-sterol-related mechanisms of AMB resistance in Ustilago maydis (11); the ergosterol contents do not vary between sensitive and resistant strains. Based on in vivo and biochemical in vitro studies, Dannaoui et al. (7) and Walsh et al. (25) suggested that the AMB resistance of A. terreus is not related to a lack of fungal ergosterol. Our data also suggested that the ergosterol content does not explain AMB resistance. The mean ergosterol contents for A. fumigatus and A. terreus were 4.4 ± 1.4 and 5.1 ± 2.4 μg/mg, respectively.
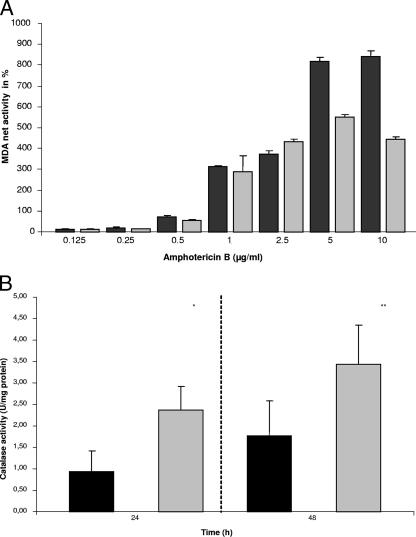
It is speculated that AMB leads to cell death by acting as an oxidizing agent (21, 24). Hydrogen peroxide (H2O2), which permeates freely through biological membranes, potentially causes cell damage by lipid peroxidation (12). Therefore, hyphal lipid peroxidation and endogenous catalase levels were measured using the thiobarbituric acid test for malonaldehyde (6, 18) and the H2O2 degradation test (1); the decrease in absorbance at a wavelength of 240 nm over 30 s was recorded (5), and the protein concentration was determined with the Bradford method (3). There were no clear differences in lipid peroxidation levels at moderate AMB concentrations between A. fumigatus and A. terreus (Fig. 1A). However, the addition of 5 and 10 μg of AMB/ml resulted in significantly higher levels of lipid peroxidation in A. fumigatus than in A. terreus. Catalases are ubiquitous metalloenzymes in aerobic organisms and protect cells from oxidative damage by converting H2O2 into 2H2O and O2. Sokol-Anderson et al. demonstrated oxidative damage to be involved in the AMB-induced killing of C. albicans (21). This effect was stopped by the addition of exogenous catalase. Our results show that A. terreus produced significantly more catalase than A. fumigatus (Fig. 1B), a finding which may play a role in the AMB resistance. A. terreus may be able to catch toxic H2O2 and transform it into less toxic substances before H2O2 can harm the fungus. Differences in oxidative-stress-buffering capacity may underlie the differential susceptibilities to AMB.
FIG. 1.
(A) Lipid peroxidation induced by increasing concentrations of AMB in A. fumigatus (black columns) and A. terreus (gray columns) hyphae (24-h culture), as measured by the quantification of malonaldehyde (MDA) after 8 h of incubation. Significant differences were seen at high AMB concentrations (5 and 10 μg/ml) (P < 0.01). (B) Hyphal catalase activity of A. fumigatus (black columns) and A. terreus (gray columns). Fungi were incubated at 37°C for 24 and 48 h and tested for catalase activity (*, P < 0.01; **, P < 0.05). Differences between means were analyzed by Student's t test. A P value of <0.05 was considered significant.
This study shows that ergosterol content and cell wall composition do not play a crucial role in the AMB resistance of A. terreus. In contrast, catalase activity in A. terreus was significantly stronger than that in A. fumigatus (P < 0.05). The level of lipid peroxidation was higher in A. fumigatus, yet only at very high AMB concentrations. Catalase may be one of the several reactive-oxidant scavengers playing a role in AMB resistance in A. terreus.
Footnotes
Published ahead of print on 11 February 2008.
REFERENCES
- 1.Aebi, H. 1984. Catalase in vitro. Methods Enzymol. 105:121-126. [DOI] [PubMed] [Google Scholar]
- 2.Arthington-Skaggs, B. A., H. Jradi, T. Desai, and C. J. Morrison. 1999. Quantitation of ergosterol content: novel method for determination of fluconazole susceptibility of Candida albicans. J. Clin. Microbiol. 37:3332-3337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bradford, M. M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72:248-254. [DOI] [PubMed] [Google Scholar]
- 4.Breivik, O. N., and J. L. Owades. 1957. Spectrophotometric semi-microdetermination of ergosterol in yeast. Agric. Food Chem. 5:360-363. [Google Scholar]
- 5.Buckova, M., J. Godocikova, A. Simonovicova, and B. Polek. 2005. Production of catalases by Aspergillus niger isolates as a response to pollutant stress by heavy metals. Curr. Microbiol. 50:175-179. [DOI] [PubMed] [Google Scholar]
- 6.Buege, J. A., and S. D. Aust. 1978. Microsomal lipid peroxidation. Methods Enzymol. 52:302-310. [DOI] [PubMed] [Google Scholar]
- 7.Dannaoui, E., E. Borel, F. Persat, M. A. Piens, and S. Picot. 1999. Amphotericin B resistance of Aspergillus terreus in a murine model of disseminated aspergillosis. J. Med. Microbiol. 49:601-606. [DOI] [PubMed] [Google Scholar]
- 8.Dannaoui, E., F. Persat, M. F. Monier, E. Borel, M. A. Piens, and S. Picot. 1999. In-vitro susceptibility of Aspergillus spp. isolates to amphotericin B and itraconazole. J. Antimicrob. Chemother. 44:553-555. [DOI] [PubMed] [Google Scholar]
- 9.Reference deleted.
- 10.Hearn, V. M., E. V. Wilson, and D. W. Mackenzie. 1980. The preparation of protoplasts from Aspergillus fumigatus mycelium. Sabouraudia 18:75-77. [PubMed] [Google Scholar]
- 11.Joseph-Horne, T., N. Manning, D. Holoman, and S. Kelly. 1996. Nonsterol related resistance in Ustilago maydis to the polyene antifungals, amphotericin B and nystatin. Phytochemistry 42:637-639. [DOI] [PubMed] [Google Scholar]
- 12.Kagan, V. E. 1988. Lipid peroxidation in biomembranes. CRC Press Inc., Boca Raton, FL.
- 13.Kawasaki, L., D. Wysong, R. Diamond, and J. Aguirre. 1997. Two divergent catalase genes are differentially regulated during Aspergillus nidulans development and oxidative stress. J. Bacteriol. 179:3284-3292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Reference deleted.
- 15.Moore, C. B., D. Law, and D. W. Denning. 1993. In-vitro activity of the new triazole D0870 compared with amphotericin B and itraconazole against Aspergillus spp. J. Antimicrob. Chemother. 32:831-836. [DOI] [PubMed] [Google Scholar]
- 16.National Committee for Clinical Laboratory Standards. 2002. Reference methods for broth dilution antifungal susceptibility testing of filamentous fungi. Approved standard M38-A. National Committee for Clinical Laboratory Standards, Wayne, PA.
- 17.Oakley, K. L., C. B. Moore, and D. W. Denning. 1999. Comparison of in vitro activity of liposomal nystatin against Aspergillus species with those of nystatin, amphotericin B (AB) deoxycholate, AB colloidal dispersion, liposomal AB, AB lipid complex, and itraconazole. Antimicrob. Agents Chemother. 43:1264-1266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Peever, T. L., and V. J. Higgins. 1989. Electrolyte leakage, lipoxygenase, and lipid peroxidation induced in tomato leaf tissue by specific and nonspecific elicitors from Cladosporium fulvum. Plant Physiol. 90:867-875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Pierce, A. M., H. D. Pierce, A. M. Unrau, and A. C. Oehlschlager. 1978. Lipid composition and polyene antibiotic resistance of Candida albicans mutants. Can. J. Biochem. 56:135-142. [DOI] [PubMed] [Google Scholar]
- 20.Seo, K., H. Akiyoshi, and Y. Ohnishi. 1999. Alteration of cell wall composition leads to amphotericin B resistance in Aspergillus flavus. Microbiol. Immunol. 43:1017-1025. [DOI] [PubMed] [Google Scholar]
- 21.Sokol-Anderson, M. L., J. Brajtburg, and G. Medoff. 1986. Amphotericin B-induced oxidative damage and killing of Candida albicans. J. Infect. Dis. 154:76-83. [DOI] [PubMed] [Google Scholar]
- 22.Sterling, T. R., and W. G. Merz. 1998. Resistance to amphotericin B: emerging clinical and microbiological patterns. Drug Resist. Updat. 1:161-165. [DOI] [PubMed] [Google Scholar]
- 23.Sutton, D. A., S. E. Sanche, S. G. Revankar, A. W. Fothergill, and M. G. Rinaldi. 1999. In vitro amphotericin B resistance in clinical isolates of Aspergillus terreus, with a head-to-head comparison to voriconazole. J. Clin. Microbiol. 37:2343-2345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Vanden Bossche, H., D. W. Warnock, B. Dupont, D. Kerridge, S. Sen Gupta, L. Improvisi, P. Marichal, F. C. Odds, F. Provost, and O. Ronin. 1994. Mechanisms and clinical impact of antifungal drug resistance. J. Med. Vet. Mycol. 32:189-202. [DOI] [PubMed] [Google Scholar]
- 25.Walsh, T. J., V. Petraitis, R. Petraitiene, A. Field-Ridley, D. Sutton, M. Ghannoum, T. Sein, R. Schaufele, J. Peter, J. Bacher, H. Armstrong, A. Espinel-Ingroff, M. G. Rinaldi, and C. A. Lyman. 2003. Experimental pulmonary aspergillosis due to Aspergillus terreus: pathogenesis and treatment of an emerging fungal pathogen resistant to amphotericin B. J. Infect. Dis. 188:305-319. [DOI] [PubMed] [Google Scholar]