Abstract
Seven hundred fifty-one Escherichia coli clinical isolates collected from 140 Japanese hospitals between 2002 and 2006 were screened for the qepA and qnr genes. Two E. coli isolates (0.3%) harbored qepA, but no qnr was identified. The results suggested a low prevalence of E. coli harboring qepA or qnr in Japan.
The most common chromosomal mechanism of resistance to fluoroquinolones (FQs) in pathogenic bacteria is amino acid substitutions in the quinolone resistance-determining regions of DNA gyrase (GyrA) and/or topoisomerase IV (ParC), which are the main target molecules of FQs (7, 8). Efflux pumps and alteration in the outer membrane proteins also contribute to chromosomal FQ resistance (6). Plasmid-mediated mechanisms of resistance to FQs such as Qnr and AAC(6′)-Ib-cr have also been described (13). We recently identified qepA, a new plasmid-mediated gene responsible for reduced FQ susceptibility from Escherichia coli C316, which was isolated in 2002 from the urine of an inpatient in Japan (21), and qepA was also reported from E. coli 1450, which was isolated in a Belgian hospital (12). qepA encodes an efflux pump belonging to the major facilitator subfamily (MSF). The MICs of norfloxacin, enrofloxacin, and ciprofloxacin were 32- to 64-fold higher for the experimental strains expressing QepA compared with the host strain (21). The MICs of ampicillin, erythromycin, kanamycin, tetracycline, and chemical substances such as carbonyl cyanide m-chlorophenylhydrazone, acriflavine, rhodamine 6G, crystal violet, and sodium dodecyl sulfate were not affected, however, indicating that FQs are the specific substrates of QepA. Moreover, a norfloxacin accumulation assay with or without carbonyl cyanide m-chlorophenylhydrazone, an efflux pump inhibitor, showed that QepA is an FQ-specific MSF-type efflux pump (21).
qnrA was the first plasmid-mediated gene that conferred resistance to quinolones such as nalidixic acid and increased MICs of FQs, originally reported in Klebsiella pneumoniae clinical isolates from the United States (11, 17). Subsequently, two other groups of qnr genes, qnrB (9) and qnrS (5), as well as their variants, have been reported. Qnrs belong to the pentapeptide repeat family and mimic DNA fragments bound to the DNA gyrase (17). The qnr genes have been identified in various bacterial species belonging to the family Enterobacteriaceae in many countries (13). In Japan, qnrS was first identified in Shigella flexneri (5) and qnrA was also identified recently (15, 16). Clinically, E. coli is the most frequent cause of urinary tract infections and FQs are some of the preferred antimicrobial agents for treatment (19). In this study, we investigated the prevalence of qepA, as well as qnrA, qnrB, and qnrS, among E. coli clinical isolates collected from Japanese medical facilities.
A total of 751 nonduplicate E. coli isolates isolated from patients admitted to 140 medical facilities in Japan between 2002 and 2006 were submitted to our reference laboratory for characterization of the genetic determinants responsible for antimicrobial resistance, as well as their genetic relatedness. All of the isolates were suspended in Luria-Bertani (LB) broth supplemented with 25% glycerol and stored in a −80°C deep freezer until analysis. The isolates were initially screened by growth on LB agar plates containing 0.025 μg/ml norfloxacin. PCR analyses for qepA and the three qnr genes were performed for all of the isolates that grew on the norfloxacin-containing plates. DNA templates for the PCR were prepared by the standard boiling method. The primer sets used for detection of qnrA, qnrB, and qnrS have been described by Cattoir et al. (1) and Robicsek et al. (14). The pairs of primers designed by Cattoir et al. (1) were able to amplify internal fragments with qnrA1 to qnrA6, qnrB1 to qnrB8, and qnrS1 to qnrS2, respectively. A 199-bp fragment of qepA was amplified by PCR with primers QEPA-F (5′-GCA GGT CCA GCA GCG GGT AG-3′) and QEPA-R (5′-CTT CCT GCC CGA GTA TCG TG-3′). The pair of primers used for detection of rmtB have been described by Doi and Arakawa (4). rmtB is a 16S rRNA methylase gene that confers resistance to aminoglycosides and was located in close proximity to qepA on a transferable plasmid in E. coli C316 (21). Positive control strains for qnrA, qnrB, and qnrS were E. coli J53(pMG252) (11), E. coli J53(pMG298) (9), and E. coli DH10B(pBC-H2.6) (5), respectively, and that for qepA was E. coli KAM32(pSTVqepA) (21). The PCR conditions used for qepA were as follows: initial denaturation at 96°C for 1 min, followed by 30 cycles of amplification at 96°C for 1 min, annealing at 60°C for 1 min, and extension at 72°C for 1 min. The final extension step was at 72°C for 5 min. The multiplex PCR condition for the qnr genes has been described previously (1, 14).
Of the 751 E. coli isolates tested, 325 grew on LB agar plates supplemented with 0.025 μg/ml norfloxacin. Two isolates (0.3%) were positive for qepA and rmtB (MRY04-1030 and MRY05-3283). The two isolates came from geographically distant hospitals. However, no qnr gene was detected among the E. coli isolates tested in this study.
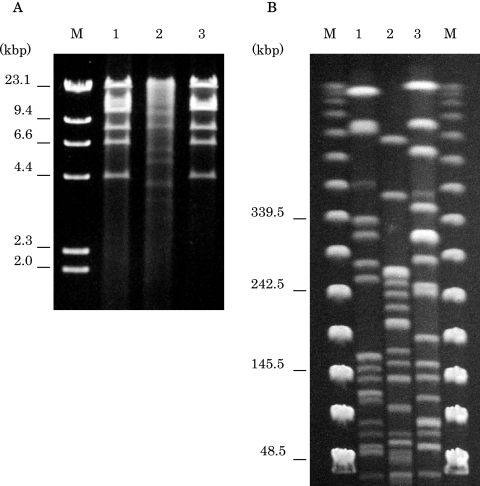
Transconjugation analysis was performed by the filter mating method with E. coli DH10B as the recipient (3). Transconjugants were selected on LB agar plates supplemented with streptomycin (50 μg/ml) and amikacin (50 μg/ml) because the plasmid carried rmtB, which confers resistance to amikacin. Plasmids were digested with EcoRI (New England BioLabs, Beverly, MA) and electrophoresed through a 1.0% agarose gel. FQ resistance was successfully transferred from the two qepA-positive E. coli isolates to E. coli DH10B at a frequency of 10−5 to 10−6 cells per recipient cell by conjugation. EcoRI restriction patterns for qepA carrying plasmids are shown in Fig. 1. The restriction patterns of pHPA from E. coli C316 and p05283 from E. coli MRY05-3283 were very similar. However, those of p05283 and p041060 from E. coli MRY04-1060 were completely different from the other two. Neither of the qepA-positive plasmids conferred resistance to ceftazidime and cefotaxime.
FIG. 1.
(A) EcoRI restriction profiles of qepA-carrying plasmids from the transconjugants. Lane M, lambda HindIII marker; lane 1, pHPA from E. coli C316; lane 2, p041060 from E. coli MRY04-1060; lane 3, p05283 from E. coli MRY05-3283. (B) PFGE fingerprinting patterns of XbaI-digested total DNA preparations from three E. coli isolates. Lanes M, lambda ladder PFGE marker used as a molecular size marker. Lanes 1 to 3, E. coli C316, MRY04-1030, and MRY05-3283, respectively.
Pulsed-field gel electrophoresis (PFGE) was performed with the CHEF-Mapper system (Bio-Rad Laboratories, Hercules, CA). Genomic DNA preparations from E. coli C316, MRY04-1060, and MRY05-3283 were digested with XbaI (New England BioLabs) (Fig. 1). The PFGE fingerprinting patterns of the three qepA-positive strains were apparently different from each other.
Antimicrobial susceptibility testing of the qepA-positive isolates and their transconjugants was performed by the agar dilution method according to the guidelines recommended by the Clinical and Laboratory Standards Institute (2) (Table 1). The MICs of norfloxacin for the transconjugants of each qepA-positive isolate were four- to fivefold higher than that for the recipient strain. The two qepA-positive isolates were also highly resistant to all of the aminoglycosides tested, including amikacin, tobramycin, and gentamicin, but susceptible to the expanded-spectrum cephalosporins and imipenem.
TABLE 1.
Antimicrobial susceptibilities of the qepA donor, transconjugant, and recipient strains used in this study
| Antimicrobial agent | MIC (μg/ml) for E. coli strain:
|
||||
|---|---|---|---|---|---|
| MRY04-1060 | MRY05-3283 | DH10B(p041060)a | DH10B(p05283)b | DH10B | |
| Norfloxacin | >128 | >128 | 0.25 | 0.25 | ≤0.008 |
| Levofloxacin | 64 | 128 | 0.008 | 0.015 | ≤0.008 |
| Ciprofloxacin | >128 | >128 | 0.015 | 0.015 | ≤0.008 |
| Ceftazidime | 0.5 | 0.5 | 0.5 | 0.5 | 0.25 |
| Cefotaxime | 0.13 | 0.13 | 0.06 | 0.06 | 0.06 |
| Imipenem | 0.13 | 0.13 | 0.25 | 0.25 | 0.25 |
| Gentamicin | >128 | >128 | >128 | >128 | 0.5 |
| Amikacin | >128 | >128 | >128 | >128 | 2 |
| Tobramycin | >128 | >128 | >128 | >128 | 0.5 |
Transconjugant of E. coli MRY04-1060.
Transconjugant of E. coli MRY05-3283.
In our previous study, qepA and rmtB were found to be encoded on the same transferable plasmid, and the analysis of the genetic environment of qepA in E. coli showed that qepA and rmtB were likely mediated by a composite transposon flanked by two copies of IS26 (21). Interestingly, an E. coli strain positive for both qepA and rmtB has also been isolated in Belgium (12). The genetic organization of the region containing qepA and rmtB was very similar to that of E. coli C316, suggesting the qepA-harboring isolates demonstrating panresistance to aminoglycosides by production of RmtB may well have already spread worldwide.
Although qnr genes have been identified in E. coli and other members of the family Enterobacteriaceae isolated from other East Asian countries, such as China, Korea, and Taiwan (10, 18, 20), the results of our study indicate that qnr-harboring E. coli is still very rare in Japanese medical facilities.
Acknowledgments
We are grateful to George A. Jacoby for provision of the qnrA- and qnrB-positive control strains.
pBC-H2.6, deposited by Mami Hata, was provided by the DNA Bank, RIKEN BioResource Center (Tsukuba, Japan), with the support of the National Bio-Resources Project of the Ministry of Education, Culture, Sports, Science, and Technology, Japan (MEXT). Part of this work was funded by a grant from the Kurozumi Medical Foundation and Grant-in-Aid for Young Scientists (B) 19790400 from the Ministry of Education, Culture, Sports, Science, and Technology, Japan.
Footnotes
Published ahead of print on 19 February 2008.
REFERENCES
- 1.Cattoir, V., L. Poirel, V. Rotimi, C. J. Soussy, and P. Nordmann. 2007. Multiplex PCR for detection of plasmid-mediated quinolone resistance qnr genes in ESBL-producing enterobacterial isolates. J. Antimicrob. Chemother. 60:394-397. [DOI] [PubMed] [Google Scholar]
- 2.Clinical and Laboratory Standards Institute. 2005. Performance standards for antimicrobial susceptibility testing; 15th informational supplement. CLSI M100-S15. Clinical and Laboratory Standards Institute, Wayne, PA.
- 3.de Lorenzo, V., and K. N. Timmis. 1994. Analysis and construction of stable phenotypes in gram-negative bacteria with Tn5- and Tn10-derived minitransposons. Methods Enzymol. 235:386-405. [DOI] [PubMed] [Google Scholar]
- 4.Doi, Y., and Y. Arakawa. 2007. 16S rRNA methylation: emerging resistance mechanism against aminoglycosides. Clin. Infect. Dis. 45:88-94. [DOI] [PubMed] [Google Scholar]
- 5.Hata, M., M. Suzuki, M. Matsumoto, M. Takahashi, K. Sato, S. Ibe, and K. Sakae. 2005. Cloning of a novel gene for quinolone resistance from a transferable plasmid in Shigella flexneri 2b. Antimicrob. Agents Chemother. 49:801-803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hooper, D. C. 1999. Mechanisms of fluoroquinolone resistance. Drug Resist. Updates 2:38-55. [DOI] [PubMed] [Google Scholar]
- 7.Hooper, D. C. 2001. Emerging mechanisms of fluoroquinolone resistance. Emerg. Infect. Dis. 7:337-341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hopkins, K. L., R. H. Davies, and E. J. Threlfall. 2005. Mechanisms of quinolone resistance in Escherichia coli and Salmonella: recent developments. Int. J. Antimicrob. Agents 25:358-373. [DOI] [PubMed] [Google Scholar]
- 9.Jacoby, G. A., K. E. Walsh, D. M. Mills, V. J. Walker, H. Oh, A. Robicsek, and D. C. Hooper. 2006. qnrB, another plasmid-mediated gene for quinolone resistance. Antimicrob. Agents Chemother. 50:1178-1182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jeong, J. Y., H. J. Yoon, E. S. Kim, Y. Lee, S. H. Choi, N. J. Kim, J. H. Woo, and Y. S. Kim. 2005. Detection of qnr in clinical isolates of Escherichia coli from Korea. Antimicrob. Agents Chemother. 49:2522-2524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Martínez-Martínez, L., A. Pascual, and G. A. Jacoby. 1998. Quinolone resistance from a transferable plasmid. Lancet 351:797-799. [DOI] [PubMed] [Google Scholar]
- 12.Périchon, B., P. Courvalin, and M. Galimand. 2007. Transferable resistance to aminoglycosides by methylation of G1405 in 16S rRNA and to hydrophilic fluoroquinolones by QepA-mediated efflux in Escherichia coli. Antimicrob. Agents Chemother. 51:2464-2469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Robicsek, A., G. A. Jacoby, and D. C. Hooper. 2006. The worldwide emergence of plasmid-mediated quinolone resistance. Lancet Infect. Dis. 6:629-640. [DOI] [PubMed] [Google Scholar]
- 14.Robicsek, A., J. Strahilevitz, D. F. Sahm, G. A. Jacoby, and D. C. Hooper. 2006. qnr prevalence in ceftazidime-resistant Enterobacteriaceae isolates from the United States. Antimicrob. Agents Chemother. 50:2872-2874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Saga, T., T. Akasaka, H. Takase, M. Tanaka, K. Sato, and M. Kaku. 2007. First detection of the plasmid-mediated quinolone resistance determinant qnrA in Enterobacteriaceae clinical isolates in Japan. Int. J. Antimicrob. Agents 29:738-739. [DOI] [PubMed] [Google Scholar]
- 16.Saito, R., W. Kumita, K. Sato, T. Chida, N. Okamura, K. Moriya, and K. Koike. 2007. Detection of plasmid-mediated quinolone resistance associated with qnrA in an Escherichia coli clinical isolate producing CTX-M-9 β-lactamase in Japan. Int. J. Antimicrob. Agents 29:600-602. [DOI] [PubMed] [Google Scholar]
- 17.Tran, J. H., and G. A. Jacoby. 2002. Mechanism of plasmid-mediated quinolone resistance. Proc. Natl. Acad. Sci. USA 99:5638-5642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wang, M., J. H. Tran, G. A. Jacoby, Y. Zhang, F. Wang, and D. C. Hooper. 2003. Plasmid-mediated quinolone resistance in clinical isolates of Escherichia coli from Shanghai, China. Antimicrob. Agents Chemother. 47:2242-2248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Warren, J. W., E. Abrutyn, J. R. Hebel, J. R. Johnsin, A. J. Schaeffer, and W. E. Stamm. 1999. Guidelines for antimicrobial treatment of uncomplicated acute bacterial cystitis and acute pyelonephritis in woman. Clin. Infect. Dis. 29:745-758. [DOI] [PubMed] [Google Scholar]
- 20.Wu, J. J., W. C. Ko, S. H. Tsai, and J. J. Yan. 2007. Prevalence of plasmid-mediated quinolone resistance determinants QnrA, QnrB, and QnrS among clinical isolates of Enterobacter cloacae in a Taiwanese hospital. Antimicrob. Agents Chemother. 51:1223-1227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yamane, K., J. Wachino, S. Suzuki, K. Kimura, N. Shibata, H. Kato, K. Shibayama, T. Konda, and Y. Arakawa. 2007. New plasmid-mediated fluoroquinolone efflux pump, QepA, found in an Escherichia coli clinical isolate. Antimicrob. Agents Chemother. 51:3354-3360. [DOI] [PMC free article] [PubMed] [Google Scholar]