Abstract
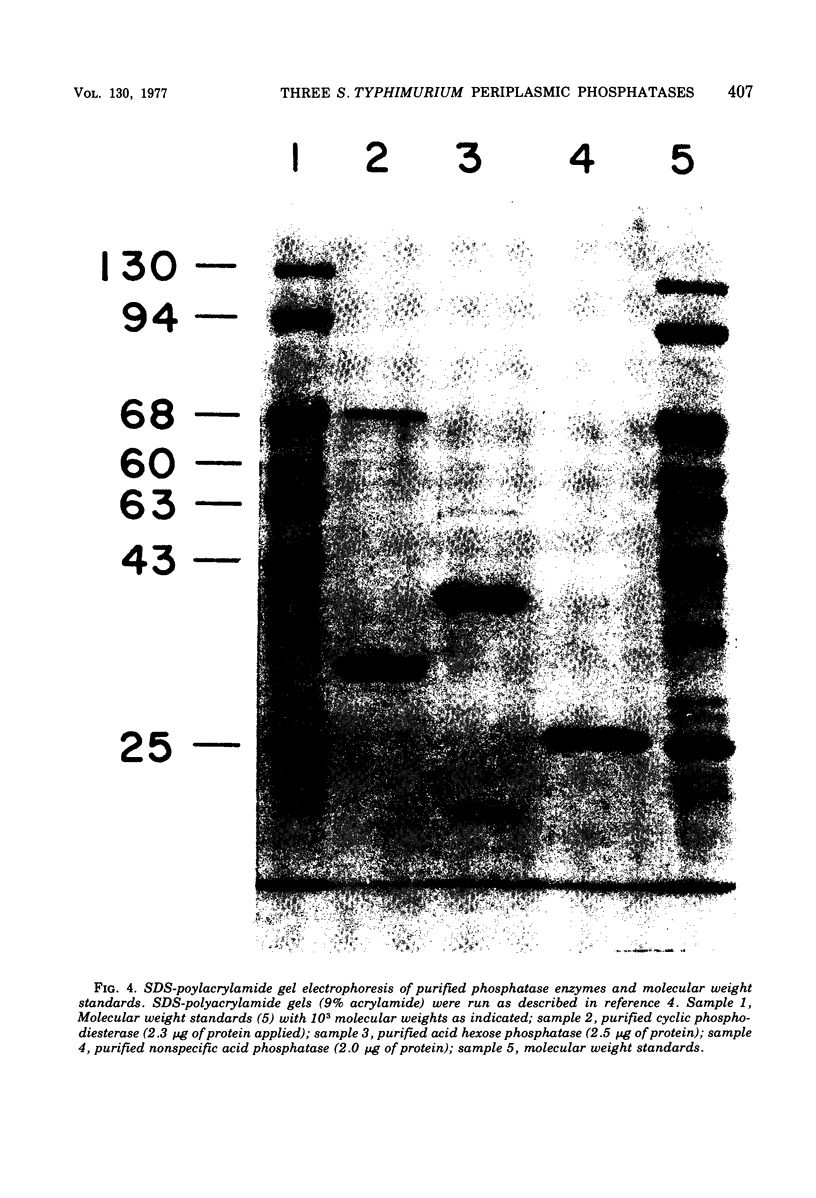
A survey of Salmonella typhimurium enzymes possessing phosphatase or phosphodiesterase activity was made using several different growth conditions. These studies revealed the presence of three major enzymes, all of which were subsequently purified: a cyclic 2' ,3'-nucleotide phosphodiesterase (EC 3.1.4.d), an acid hexose phosphatase (EC 3.1.3.2), and a nonspecific acid phosphatase (EC 3.1.3.2). A fourth enzyme hydrolyzed bis-(p-nitrophenyl)phosphate but none of the other substrates tested. No evidence was found for the existence of an alkaline phosphatase (EC 3.1.3.1) or a specific 5'-nucleotidase (EC 3.1.3.5) in S. typhimurium LT2. All three phosphatases could be measured efficiently in intact cells, which suggested a periplasmic location; however, they were not readily released by osmotic shock procedures. The nonspecific acid phosphatase, which was purified to apparent homogeneity, yielded a single polypeptide band on both sodium dodecyl sulfate and acidic urea gel electrophoretic systems.
Full text
PDF










Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- AMES B. N., HARTMAN P. E., JACOB F. Chromosomal alterations affecting the regulation of histidine biosynthetic enzymes in Salmonella. J Mol Biol. 1963 Jul;7:23–42. doi: 10.1016/s0022-2836(63)80016-9. [DOI] [PubMed] [Google Scholar]
- ANRAKU Y. A NEW CYCLIC PHOSPHODIESTERASE HAVING A 3'-NUCLEOTIDASE ACTIVITY FROM ESCHERICHIA COLI B. I. PURIFICATION AND SOME PROPERTIES OF THE ENZYME. J Biol Chem. 1964 Oct;239:3412–3419. [PubMed] [Google Scholar]
- ANRAKU Y. A NEW CYCLIC PHOSPHODIESTERASE HAVING A 3'-NUCLEOTIDASE ACTIVITY FROM ESCHERICHIA COLI B. II. FURTHER STUDIES ON SUBSTRATE SPECIFICITY AND MODE OF ACTION OF THE ENZYME. J Biol Chem. 1964 Oct;239:3420–3424. [PubMed] [Google Scholar]
- Abrell J. W. Ribonuclease I released from Escherichia coli by osmotic shock. Arch Biochem Biophys. 1971 Feb;142(2):693–700. doi: 10.1016/0003-9861(71)90535-2. [DOI] [PubMed] [Google Scholar]
- Ames G. F., Lever J. Components of histidine transport: histidine-binding proteins and hisP protein. Proc Natl Acad Sci U S A. 1970 Aug;66(4):1096–1103. doi: 10.1073/pnas.66.4.1096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brockman R. W., Heppel L. A. On the localization of alkaline phosphatase and cyclic phosphodiesterase in Escherichia coli. Biochemistry. 1968 Jul;7(7):2554–2562. doi: 10.1021/bi00847a016. [DOI] [PubMed] [Google Scholar]
- DAVIS B. J. DISC ELECTROPHORESIS. II. METHOD AND APPLICATION TO HUMAN SERUM PROTEINS. Ann N Y Acad Sci. 1964 Dec 28;121:404–427. doi: 10.1111/j.1749-6632.1964.tb14213.x. [DOI] [PubMed] [Google Scholar]
- Datta A. K., Burma D. P. Association of ribonuclease I with ribosomes and their subunits. J Biol Chem. 1972 Nov 10;247(21):6795–6801. [PubMed] [Google Scholar]
- Dvorak H. F., Brockman R. W., Heppel L. A. Purification and properties of two acid phosphatase fractions isolated from osmotic shock fluid of Escherichia coli. Biochemistry. 1967 Jun;6(6):1743–1751. doi: 10.1021/bi00858a024. [DOI] [PubMed] [Google Scholar]
- Fairbanks G., Steck T. L., Wallach D. F. Electrophoretic analysis of the major polypeptides of the human erythrocyte membrane. Biochemistry. 1971 Jun 22;10(13):2606–2617. doi: 10.1021/bi00789a030. [DOI] [PubMed] [Google Scholar]
- GAREN A., LEVINTHAL C. A fine-structure genetic and chemical study of the enzyme alkaline phosphatase of E. coli. I. Purification and characterization of alkaline phosphatase. Biochim Biophys Acta. 1960 Mar 11;38:470–483. doi: 10.1016/0006-3002(60)91282-8. [DOI] [PubMed] [Google Scholar]
- Glaser L., Melo A., Paul R. Uridine diphosphate sugar hydrolase. Purification of enzyme and protein inhibitor. J Biol Chem. 1967 Apr 25;242(8):1944–1954. [PubMed] [Google Scholar]
- Gutnick D., Calvo J. M., Klopotowski T., Ames B. N. Compounds which serve as the sole source of carbon or nitrogen for Salmonella typhimurium LT-2. J Bacteriol. 1969 Oct;100(1):215–219. doi: 10.1128/jb.100.1.215-219.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HEPPEL L. A., HARKNESS D. R., HILMOE R. J. A study of the substrate specificity and other properties of the alkaline phosphatase of Escherichia coli. J Biol Chem. 1962 Mar;237:841–846. [PubMed] [Google Scholar]
- Hafkenscheid J. C. Properties of an acid phosphatase in Escherichia coli. Biochim Biophys Acta. 1968 Nov 19;167(3):582–589. doi: 10.1016/0005-2744(68)90048-x. [DOI] [PubMed] [Google Scholar]
- LICHTENSTEIN J., BARNER H. D., COHEN S. S. The metabolism of exogenously supplied nucleotides by Escherichia coli. J Biol Chem. 1960 Feb;235:457–465. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lever J. E. Purification and properties of a component of histidine transport in Salmonella typhimurium. The histidine-binding protein J. J Biol Chem. 1972 Jul 10;247(13):4317–4326. [PubMed] [Google Scholar]
- Melo A., Glaser L. Nucleotide diphosphate hexose pyrophosphatases. Biochem Biophys Res Commun. 1966 Mar 8;22(5):524–531. doi: 10.1016/0006-291x(66)90306-8. [DOI] [PubMed] [Google Scholar]
- NEU H. C., HEPPEL L. A. SOME OBSERVATIONS ON THE "LATENT" RIBONUCLEASE OF ESCHERICHIA COLI. Proc Natl Acad Sci U S A. 1964 Jun;51:1267–1274. doi: 10.1073/pnas.51.6.1267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neu H. C., Heppel L. A. The release of enzymes from Escherichia coli by osmotic shock and during the formation of spheroplasts. J Biol Chem. 1965 Sep;240(9):3685–3692. [PubMed] [Google Scholar]
- Neu H. C. The 5'-nucleotidase of Escherichia coli. I. Purification and properties. J Biol Chem. 1967 Sep 10;242(17):3896–3904. [PubMed] [Google Scholar]
- Neu H. C. The 5'-nucleotidases and cyclic phosphodiesterases (3'-nucleotidases) of the Enterobacteriaceae. J Bacteriol. 1968 May;95(5):1732–1737. doi: 10.1128/jb.95.5.1732-1737.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nossal N. G., Heppel L. A. The release of enzymes by osmotic shock from Escherichia coli in exponential phase. J Biol Chem. 1966 Jul 10;241(13):3055–3062. [PubMed] [Google Scholar]
- ROGERS D., REITHEL F. J. Acid phosphatases of Escherichia coli. Arch Biochem Biophys. 1960 Jul;89:97–104. doi: 10.1016/0003-9861(60)90018-7. [DOI] [PubMed] [Google Scholar]
- Schlesinger M. J., Olsen R. Expression and localization of Escherichia coli alkaline phosphatase synthesized in Salmonella typhimurium cytoplasm. J Bacteriol. 1968 Nov;96(5):1601–1605. doi: 10.1128/jb.96.5.1601-1605.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takayama K., MacLennan D. H., Tzagoloff A., Stoner C. D. Studies on the electron transfer system. LXVII. Polyacrylamide gel electrophoresis of the mitochondrial electron transfer complexes. Arch Biochem Biophys. 1966 Apr;114(1):223–230. doi: 10.1016/0003-9861(66)90324-9. [DOI] [PubMed] [Google Scholar]
- Uerkvitz W., Karlström O., Munch-Petersen A. A deoxyuridine monophosphate phosphatase detected in mutants of Escherichia coli lacking alkaline phosphatase and 5'-nucleotidase. Mol Gen Genet. 1973 Mar 19;121(4):337–346. doi: 10.1007/BF00433232. [DOI] [PubMed] [Google Scholar]
- VOGEL H. J., BONNER D. M. Acetylornithinase of Escherichia coli: partial purification and some properties. J Biol Chem. 1956 Jan;218(1):97–106. [PubMed] [Google Scholar]
- von HOFSTEN, PORATH J. Purification and some properties of an acid phosphatase from Escherichia coli. Biochim Biophys Acta. 1962 Oct 8;64:1–12. doi: 10.1016/0006-3002(62)90754-0. [DOI] [PubMed] [Google Scholar]