Abstract
Epidermal growth factor (EGF) and its receptors (EGFR) play important roles in tumorigenesis. In various experimental cancers, treatment with antagonists of bombesin/gastrin-releasing peptide (BN/GRP) produces a reduction in EGFRs, concomitant to inhibition of tumor growth. To investigate the mechanisms involved, we monitored concentrations of BN/GRP antagonist RC-3095 in serum of mice, rats, and hamsters given a single subcutaneous or intravenous injection of this analog. In parallel studies, we measured levels and mRNA expression of EGFRs in estrogen-dependent and independent MXT mouse mammary cancers, following a single subcutaneous administration of RC-3095 to tumor-bearing mice. Peak values of RC-3095 in serum were detected 2 min after intravenous or 15 min after subcutaneous injection. The levels of RC-3095 declined rapidly and became undetectable after 3–5 hr. In the estrogen-dependent MXT tumors, the concentration of EGF receptors was reduced by about 60% 6 hr following injection and returned to original level after 24 hr. Levels of mRNA for EGFR fell parallel with the receptor number and were nearly normal after 24 hr. In the hormone-independent MXT cancers, the number of EGFRs decreased progressively, becoming undetectable 6 hr after injection of RC-3095, and returned to normal values at 24 hr, but EGFR mRNA levels remained lower for 48 hr. Thus, in spite of rapid elimination from serum, BN/GRP antagonist RC-3095 can induce a prolonged decrease in levels and mRNA expression of EGFRs. These findings may explain how single daily injections of BN/GRP antagonists can maintain tumor growth inhibition.
Keywords: gastrin releasing peptide antagonist, receptor down-regulation, blood levels of bombesin antagonist
Bombesin (BN) and its mammalian counterpart, gastrin releasing peptide (GRP) have a wide range of neuroendocrine activities (1–3). BN and GRP influence the secretion of gastrointestinal hormones, and stimulate release of gastrin and somatostatin as well as pancreatic exocrine secretion (1–3). BN-like peptides also induce smooth muscle contraction in the stomach, gall-bladder, urinary bladder, and uterus (2). Receptors for BN/GRP have been detected in various cells of the gastrointestinal system, the pancreas, smooth muscles of the digestive tract, bladder and uterus, the neuronal elements of the myenteric plexus, and in the nervous system (1–3).
BN and GRP stimulate the proliferation of several cell types. The mitogenic effects of BN and GRP have been extensively investigated in vitro with Swiss 3T3 fibroblasts (3, 4). In vivo, GRP has an important role in fetal lung growth and differentiation (3). GRP is secreted in human and cow milk and also acts as a growth factor for the digestive tract of newborn (3). Chronic administration of BN/GRP increases the DNA content of gastric and colonic mucosa and induces hyperplasia of mucosal cells. Long-term treatment with BN or GRP causes hyperplasia of the pancreas, as shown by an increase in the weight and DNA content of the organ (2, 3).
BN-like peptides may also play a role in development and progression of some cancers. GRP, GRP receptors, and a related peptide neuromedin B and its receptors have been detected in many small cell lung carcinoma (SCLC) lines showing that BN-like peptides may act as autocrine growth factors in this tumor (5, 6). An involvement of BN/GRP in the promotion of other tumors has also been demonstrated, including gastric, pancreatic, colon, breast, and prostate cancers (3, 6). The effect of autocrine BN-like peptides on these tumors is thought to be mediated through specific membrane receptors on target cells (6).
In view of a possible role of BN-like peptides in the growth of various cancers, several series of BN/GRP antagonists have been developed and tested in experimental tumor models (4, 6–18). Pseudononapeptide BN/GRP receptor antagonist [d-Tpi6,Leu13Ψ-(CH2NH)Leu14]BN (6–14) (RC-3095) and other antagonists were synthesized in our laboratory for therapy of various cancers (6, 7). RC-3095 was shown to be a potent tumor-growth inhibitor in various experimental cancer models, including nitrosamine-induced pancreatic cancers in hamsters (8, 9), MXT mouse mammary cancers (10), Dunning rat prostate cancers (11), as well as human tumors such as SW-1990 pancreatic cancers (12), MCF-7MIII breast cancers (13), MKN-45 gastric tumors (14), HT-29 colon cancers (15), various SCLC (16), and PC-82 and PC-3 prostate cancers (6, 17) xenografted into nude mice. In some of these studies, the antagonist RC-3095 was administered by osmotic minipumps that released 20–60 μg per day (8–11), and in others injected subcutaneously (s.c.) in a dose of 20 μg once per day (14, 16, 17) or 10 μg peptide twice daily (12, 13, 15). The tumor growth inhibition caused by RC-3095 and other BN/GRP antagonists was invariably linked to a major down-regulation of epidermal growth factor receptors (EGFRs) in pancreatic, mammary, gastric, colonic, and prostatic cancers, as well as SCLC (6, 8–11, 13–18). Thus, the inhibitory effect of BN/GRP antagonists appears to be mediated by interference with EGFR mechanisms.
Little is known about the pharmacokinetics of BN antagonists (19). Several reports show that the half-life of BN-like peptides in circulation is very short. Patients with SCLC do not have high levels of GRP, possibly because of the fast metabolism of this peptide and autocrine pathways of circulation (3). The half-life of various BN/GRP antagonists was found to be between 154 and 1,388 min in an in vitro study (19). To investigate the pharmacokinetics of RC-3095, we generated antibodies against RC-3095 and developed a rapid, sensitive, and specific radioimmunoassay suitable for determination of RC-3095 levels in unextracted serum (20). In preliminary in vivo experiments, we found that blood levels of the antagonist decreased rapidly after intravenous (i.v.) or s.c. injection of RC-3095 to rats. One to 3 hr after the injection of RC-3095, the serum levels of this analog became undetectable (20).
The aim of this study was to elucidate how BN/GRP antagonists with a short half-life in blood can maintain a steady tumor growth inhibition. We investigated blood levels of antagonist RC-3095 in various experimental animals after i.v. or s.c. administration of this analog, and the effect of a single s.c. injection of RC-3095 on EGFR levels and the expression of mRNA for EGFR in estrogen-dependent and independent MXT mouse mammary cancers.
MATERIALS AND METHODS
Materials.
BN receptor antagonist RC-3095 [d-Tpi-Gln-Trp-Ala-Val-Gly-His-Leuψ(CH2NH)Leu-NH2], originally synthesized in our laboratory by solid phase methods (7), was made by Asta Medica (Frankfurt/Main, Germany) in acetate form (batch D-22213). The production and characteristics of JH-631b antibody against RC-3095 were reported previously (20).
RC-3095 Radioimmunoassay Procedure.
The radioiodination and purification of RC-3095, the technical details, and the validation of the radioimmunoassay were as described (20).
Animals.
Adult male Sprague–Dawley rats weighing approximately 350 g, adult female B6D2F1 mice, and male athymic (nu/nu) nude mice weighing about 22 g, as well as female Syrian golden hamsters with an average weight of 153 g were obtained from the National Cancer Institute (Frederick, MD). The animals were maintained in our animal facility under standard conditions as described (8, 10, 13, 17).
Tumors.
The source of the estrogen-dependent MXT(3.2) and estrogen-independent MXT(3.2)ovex mouse mammary cancers and the methods of tumor transplantation were as described (10).
Blood Levels of RC-3095 After a Single Injection.
Five rats were given a single injection of 100 μg of RC-3095 i.v. or s.c. Five hamsters were injected i.v. with 40 μg of RC-3095. The same dose of 40 μg was given i.v. to five nude mice and s.c. to five BDF mice. Blood samples were taken from the jugular vein before and 2, 5, 15, 60, 180, and in some cases 300 min after the injection. RC-3095 levels were determined as described (20).
EGFR Study.
Estrogen-dependent and independent MXT cancers were transplanted s.c. into 25 and 18 female BDF mice, respectively. When the tumors were well developed, the mice received one injection of 100 μg RC-3095 s.c. The mice were sacrificed 0.5, 1, 2, 4, 6, 24, and 48 hr after injection. The tumors were removed, cleaned, weighed, and processed for receptor assays and molecular biology studies.
Radioreceptor Assay.
Tumor membrane fraction was prepared as described (16, 18). 125I-EGF (specific activity 750–1,200 Ci/mmol; 1 Ci = 37 GBq) was purchased from Amersham. Binding assays and the evaluation of data were performed as reported (16, 18).
RNA Extraction.
Total RNA was extracted from mouse MXT mammary tumor samples with the RNAZol isolation Kit (Cinna/Biotecx Laboratories, Houston) and quantified spectrometrically at 260 nm.
Reverse Transcription (RT).
One microgram of total RNA was added to a tube containing 10 mM Tris⋅HCl (pH 8.3), 50 mM KCl, 5 mM MgCl2, 1 mM of each deoxynucleotide triphosphate (dNTP), 1 unit RNasin, 2.5 μM random hexamer primers, and RNase-free deionized distilled water in a final volume of 19 μl. This mixture was heated for 10 min at 65°C and quenched on ice, and 2.5 units of Moloney murine leukemia virus reverse transcriptase in 1 μl were added for a total reaction volume of 20 μl, according to the manufacturer’s instructions (Perkin–Elmer). This mixture was incubated at room temperature for 10 min and then at 42°C for 1 hr. The reaction was terminated by heating at 95°C for 5 min and quenching on ice.
PCR Amplification.
The 20 μl of the RT reaction from each sample were diluted to a final volume of 100 μl with 10 mM Tris⋅HCl (pH 8.3), 50 mM KCl, 2 mM MgCl2, and ampliTaq DNA polymerase according to the manufacturer’s instructions (Perkin–Elmer). The used primers for mouse EGFR were synthesized based on the cDNA sequence (21): 5′-GGA GGA AAA GAA AGT CTG CC-3′ (sense) and 5′-CCC ATA GTT GGA TAG GAT GG-3′ (antisense). The primers for mouse β actin were: 5′-GTG GGC CGC TCT AGG CAC CAA-3′ (sense) and 5′-CTC TTT GAT GTC ACG CAC GAT TTC-3′ (antisense).
Thirty cycles of PCR for mouse EGFR and mouse β actin were carried out with a thermal cycler (Stratagene) according to the step program of 94°C for 1 min, 54°C for 1 min, and 72°C for 1 min, followed by 10 min final extension at 72°C. The number of cycles was determined previously as in the exponential range of PCR product amplification necessary for quantitative densitometry. Negative controls were run parallel to exclude cross contamination of samples and the presence of contaminating genomic DNA in the RNA from the tumors.
After amplification, 5 μl of the PCR products were electrophoretically separated on 1.8% agarose gel. The gel was treated in denaturation buffer with 50 mM NaOH/1.5 M NaCl, followed by neutralization buffer containing Tris⋅HCl (pH 8.0)/1.5 M NaCl. The gel was then blotted onto a nylon membrane (Hybond N+, Amersham) by capillary transfer, and the DNA was linked onto it by heating for 2 hr at 80°C.
Southern Blot Analysis.
Sample blots were prehybridized at 60°C for 16 hr in buffer containing 4× SSC solution, 2× Denhardt’s solution, 0.1% SDS, 5 mM EDTA, and 100 μg/ml denaturated salmon sperm DNA. After prehybridization, the sample blots were hybridized at 60°C for 20 hr in hybridization buffer containing 5× SSC, 0.5× Denhardt’s solution, 0.02 M Tris×HCl, 100 μg/ml sonicated salmon DNA, and 150 ng of [32P]-5′-end labeled oligonucleotide probe (mEGFR) or random-primed cDNA (β-actin cDNA). The oligonucleotide probe used for mouse EGFR was: 5′-CTC TTT GAT GTC ACG CAC GAT TTC-3. The β-actin cDNA probe (1.1 kb) (American Type Culture Collection) was used to hybridize the β-actin PCR product. Mouse EGFR oligonucleotide probe was labeled in reaction containing [32P]ATP, 3,000 Ci/mmol; 10 mCi/ml, 150 ng oligonucleotide probe; and 10 units T4 polynucleotide kinase. The β-actin cDNA was labeled using the random priming method (Amersham). After incubation at 37°C for 45 min, the reactions were stopped by adding 1 μl of 0.5 M EDTA and unincorporated radionucleotides were removed using Nuctrap columns (Stratagene). The blots were washed under stringent conditions and the signals from samples were scanned and quantified with an imaging densitometer (Model GS-700, Bio-Rad).
Statistical Methods.
All data were expressed as the mean ± SEM, and statistical analyses of the tumor data were performed using Duncan’s new multiple range test or Student’s t test. All P values are based on two-sided hypothesis testing.
RESULTS
Serum RC-3095 Levels After a Single Injection of the Antagonist.
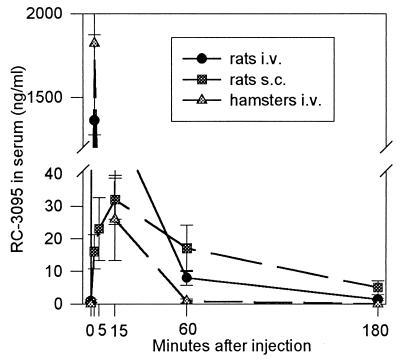
In rats, serum RC-3095 levels were in the 1.3 μg/ml range 2 min after i.v. injection of 100 μg peptide (Fig. 1). The levels decreased by about 60% at 5 min, and 3 hr after the injection RC-3095 became undetectable in blood. Following the s.c. administration, levels of the antagonist RC-3095 in serum of rats were much lower than when given i.v., with a peak (32 ng/ml) occurring at 15 min. After 3 hr, the serum RC-3095 levels fell to about 5 ng/ml (Fig. 1). The levels of RC-3095 in the blood of hamsters after i.v. injection of 40 μg peptide were similar to those found in rats (Fig. 1).
Figure 1.
Serum RC-3095 levels in rats after i.v. or s.c. injection of 100 μg peptide and in hamsters after i.v. injection of 40 μg RC-3095.
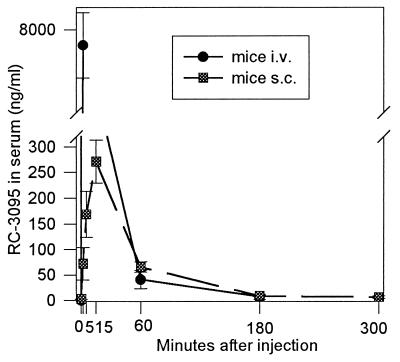
In mice, the highest serum RC-3095 levels (7.7 μg/ml) were found 2 min after i.v. injection of 40 μg peptide (Fig. 2). The values were greater than those observed in rats because the doses of RC-3095 per gram of body weight were higher in mice. At 60 min, the levels fell to 41 ng/ml and continued to decline. Following s.c. injection into mice, the levels of RC-3095 were lower than after i.v. administration; the peak (271 ng/ml) being observed at 15 min. At 180 min, the levels of RC-3095 in blood fell to 10 ng/ml, and the peptide became undetectable 5 hr after s.c. injection (Fig. 2).
Figure 2.
RC-3095 levels in sera of mice injected i.v. or s.c. with 40 μg of the antagonist.
Analysis of EGFRs.
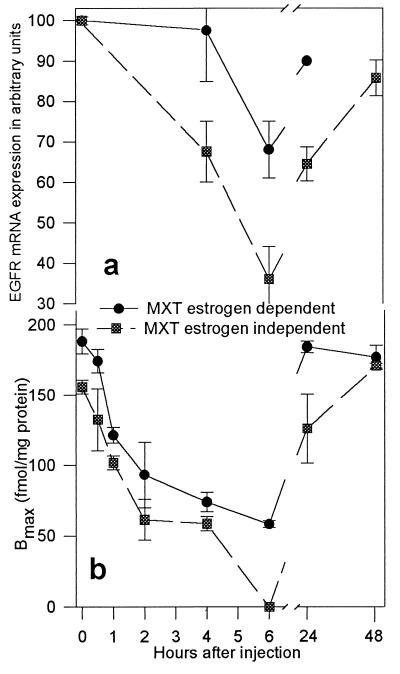
Changes in mRNA levels for EGFR and binding capacity of EGFR before and after treatment with BN antagonist RC-3095 are shown in Fig. 3 a and b. In the control estrogen-independent MXT cancers, labeled EGF was bound to one class of binding sites with high affinity (Kd = 0.77 ± 0.08 × 10−9 M). The binding capacity (Bmax) of the receptors was 155.8 ± 4.95 × 10−15 mol/mg of membrane protein. After a single s.c. injection of 100 μg RC-3095, the binding capacity of EGFRs decreased progressively becoming significantly lower than the original levels at 1 hr (Table 1). This decline continued and receptors became undetectable at 6 hr after injection of RC-3095. The recovery in receptor number occurred during the following 18 hr and EGFR levels returned to initial values between 24 and 48 hr after injection of RC-3095 (Fig. 3b, Table 1).
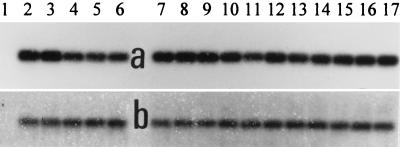
Figure 3.
Changes in (a) mRNA expression and (b) binding capacity of EGFRs of estrogen-dependent and estrogen-independent MXT mouse mammary cancers after a single s.c. injection of RC-3095.
Table 1.
Effect of a single treatment s.c. with BN/GRP antagonist RC-3095 on binding characteristics and mRNA expression of EGFRs in estrogen-dependent and independent MXT mouse mammary cancers
| Hours after injection | Kd, nM | Bmax, fmol/mg protein | mRNA expression, % of control levels |
|---|---|---|---|
| Estrogen-independent MXT | |||
| Control | 0.77 ± 0.08 | 155.8 ± 4.95 | 100 ± 1.0 |
| 0.5 | 1.43 ± 0.35 | 132.4 ± 22.1 | |
| 1 | 1.36 ± 0.29 | 101.6 ± 4.8* | |
| 2 | 0.89 ± 0.13 | 61.5 ± 14.4† | |
| 4 | 0.48 ± 0.14 | 58.6 ± 5.05† | 67.5 ± 7.5* |
| 6 | ND | ND | 36.0 ± 8.0† |
| 24 | 0.86 ± 0.06 | 125.9 ± 24.6 | 64.5 ± 4.2* |
| 48 | 1.06 ± 0.11 | 170.9 ± 1.7 | 85.8 ± 4.4* |
| Estrogen-dependent MXT | |||
| Control | 0.99 ± 0.1 | 188.3 ± 8.8 | 100 ± 0 |
| 0.5 | 0.65 ± 0.02 | 174.3 ± 8.4 | |
| 1 | 0.94 ± 0.07 | 121.2 ± 5.5† | |
| 2 | 0.84 ± 0.33 | 93.1 ± 23.3† | |
| 4 | 0.67 ± 0.04 | 73.9 ± 6.95† | 97.5 ± 12.5 |
| 6 | 0.70 ± 0.11 | 58.3 ± 2.4† | 68.0 ± 7.0* |
| 24 | 0.99 ± 0.03 | 184.2 ± 4.1 | 90.0 ± 0 |
| 48 | 1.13 ± 0.02 | 176.9 ± 8.55 | |
ND, not detectable. Values are means ± SEM.
*P < 0.05; †P < 0.01.
The changes in the levels of EGFRs in estrogen-dependent MXT cancers followed a pattern similar to that in estrogen-independent tumors. After s.c. administration of RC-3095, the receptor levels fell significantly at 1 hr and continued to decline, the lowest EGFR concentration being found at 6 hr (Fig. 3b, Table 1). The recovery in receptor number occurred within 24 hr. The binding affinities of the receptors remained virtually unchanged in both experiments.
mRNA Expression for EGFR.
The results of RT-PCR analysis of mouse EGFR mRNA expression performed with specific primers and with 1 μg of total RNA from MXT cancers are shown in Fig. 4a. The expected length of the PCR product was 304 bp. Mouse β-actin gene expression was used as a control (Fig. 4b). EGFR and β-actin cDNA obtained after RT-PCR was analyzed by Southern blotting (Fig. 5 a and b). The blots were scanned and quantified with a densitometer. The results are shown in Table 1 and Fig. 3a. In estrogen-independent breast cancers, mRNA for EGFR decreased gradually after the injection of the BN/GRP antagonist. A nadir was observed at 6 hr. The mRNA expression for EGFR began to rise within ≈18 hr, but it was still lower at 24 and 48 hr than the original level. In estrogen-dependent MXT cancers, mRNA expression for EGFR showed a significant decrease 6 hr after the injection of RC-3095. However, the mRNA EGFR levels returned to nearly original values after 24 hr.
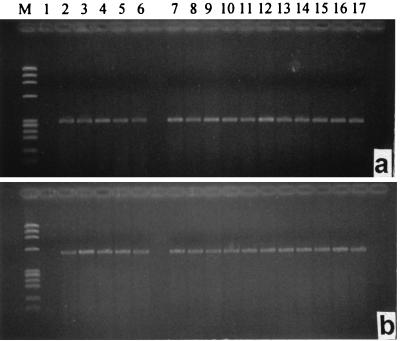
Figure 4.
RT-PCR analysis of mouse EGFR (a) and β-actin (b) gene expression. PCR products were separated by 1.8% agarose gel electrophoresis and stained with ethidium bromide. The expected length was 304 bp (a) and 540 bp (b). Lanes: M, (molecular weight marker) ΦX 174 HaeIII digest; 1, negative control; 2–6, tumor samples from estrogen-dependent MXT cancer; 2 and 3, untreated animals; 4–6, animals treated with RC-3095 4, 6, and 24 hr, respectively, after treatment; 7–17, tumor samples from estrogen-independent MXT tumor; 7–9, untreated animals; 10 and 11, animals treated with RC-3095 4 and 6 hr, respectively, after treatment; 12–14, animals treated with RC-3095 24 hr after treatment; 15–17, animals treated with RC-3095 48 hr after treatment.
Figure 5.
Southern blot analysis of the mouse EGFR cDNA and β-actin cDNA obtained after RT-PCR. Hybridizations were performed with the oligonucleotide specific for mouse EGFR (a) and cDNA probe specific for β actin (b). Numbering is the same as in Fig. 4.
DISCUSSION
BN/GRP antagonists synthesized by us and by other investigators have been tested mainly in various in vitro models for their ability to inhibit pharmacological effects of BN or GRP (4, 6, 7, 22, 23). In vivo tests have also been used for investigating the effects of antagonists, but most in vivo systems are inherently more complicated and more difficult to interpret. Numerous complex pharmacological effects of BN/GRP are due to both direct and indirect mechanisms of action and depend on the species studied and the route of administration (3). Various studies show that the metabolic effects of BN/GRP and their antagonists appear very rapidly, but are relatively short lasting (24–29). An i.v. injection of GRP produces a prompt increase in gastric secretion that peaks within 10 min. A BN/GRP antagonist given 2 min before GRP abolishes the effect of GRP (24). Infusion (i.v.) of neuromedin C to rats increases plasma cholecystokinin levels within 15–30 min (25). The perfusion of isolated stomach by way of the celiac artery increases somatostatin release with a peak at 4 min from the start of infusion (26). BN-like peptides cause a delay of gastric emptying 5 min after i.v. administration, and BN/GRP antagonists given 5 min before the agonists inhibit this effect (27). In our previous studies, bolus i.v. injection of GRP (14–27) elevated serum gastrin level with a peak at 3 min, and the effects lasted for 6 min (28, 29). The gastrin response to GRP could be significantly decreased by i.v. injection of different BN/GRP antagonists 2 min before the GRP administration (28, 29). Thus, RC-3095 given i.v. or by inhalation 2 min prior to GRP inhibited the effect of GRP on gastrin release. However, i.v. injection of RC-3095 30 min before GRP could not suppress GRP-induced gastrin release. These findings indicate that after i.v. administration, BN-like peptides act within a few minutes and reach maximal effects between 2–6 min, but their activity does not last long. However, when these peptides are given s.c., the maximal effects are obtained at 15–30 min after injection and last for a few hours (28, 30, 31). Thus, plasma cholecystokinin levels were significantly increased after a single s.c. injection of BN, with a peak at 15 min (30). BN stimulated pancreatic secretion with a peak at 20–30 min after s.c. injection, and this effect could be inhibited by an antagonist given 2.5 hr before the agonist (31). RC-3095 administered s.c. inhibited plasma glucose elevation caused by GRP and had the greatest antagonistic activity 30 min after injection (28). RC-3095 given subcutaneously 1 hr before GRP (14–27) was also able to significantly inhibit gastrin release.
As we report in this investigation, RC-3095 levels in blood reach a peak 15 min after a single s.c. injection, and the administered peptide virtually disappears from circulation 3–5 hr later. The key question to be answered by our study was how BN antagonist RC-3095, with a short half-life in blood, could exert tumor suppression (6, 8–18). The therapeutic results (6, 8–18) seemed to be independent of whether the administration was made s.c. once a day, twice daily, or by a continuous release from osmotic minipumps.
The fall in the levels of EGFRs in various cancers, with a duration of up to 24 hr after a single injection of BN/GRP antagonists, might explain the mechanism of tumor inhibitory action of this class of analogs (8–11, 13–18). EGF and EGFR have well-established roles in the development and progression of various cancers (6, 10, 11, 13–16). Overexpression of EGFR is a specific marker for malignant transformation (32). EGFR contributes to androgen-independent prostatic cancer growth, and EGFR-blocking monoclonal antibodies inhibit growth of prostate cancer (33). EGFR was detected by ligand binding studies in 49% of human breast cancers (interstudy range 16–91%) (34). Hormone-independent breast cancers have higher levels of EGFR and erbB-2 and an increase in the number of EGFR may indicate poor prognosis (35, 36). EGFR levels correlate inversely with those for sex steroid hormone receptors and most agents have the opposite effect on these two types of receptors (37). Based on these and related findings, it appears that the reduction in mitogenic effects generated through the EGFR system might be a promising approach to tumor therapy.
Our results show that in MXT breast cancers RC-3095 can induce heterologous down-regulation of EGFRs in a very short time, and these receptors remain down-regulated for several hours. In the MXT model, the concentration of the EGFRs returned to normal levels 24 hr after injection. Expression of mRNA for EGFR correlated well with receptor levels in the estrogen-dependent MXT tumors. However, in hormone-independent MXT cancers, EGFR mRNA levels remained lower than the original values for 24–48 hr, whereas receptor concentration returned to normal. The fact that an incomplete resynthesis of receptors was found following the down-regulation of EGFRs by RC-3095 indicates that possibly EGFR recirculation after internalization might contribute to the recovery in receptor number.
The mechanisms for how BN/GRP antagonists induce a heterologous down-regulation of EGFRs are not clear. There are several possibilities for interactions between signaling pathways of receptor tyrosine kinases (TKs) and G protein-coupled receptors. After ligand binding, EGFRs undergo dimerization, the intracellular protein TK domain of the receptor becomes activated and the receptor is autophosphorylated. Consequently, EGFR is internalized and undergoes a destruction in endosomes or may recycle to the cell surface.
Receptor functions are controlled at various levels that may affect the receptor itself or specific steps of the signaling process. Ligand binding initiates regulatory mechanisms at the receptor level. EGFR kinase activity can be controlled by the state of threonine phosphorylation of the receptor and the dimeric or oligomeric state of the receptor (37). Binding of EGF to the receptor results in internalization and degradation, which are followed by elevation of EGFR mRNA levels (38).
In addition to these auto-controlling mechanisms, several other possibilities for heterologous regulation of signaling pathways exist. Various hormones and growth factors can modulate EGFR by phosphorylation of the receptors, by causing down-regulation through internalization, or by changing the production of the receptor at transcriptional level. Estrogens and progestins cause induction of EGFR in hormone-dependent breast cancer cells (37) and treatment with anti-estrogens produces a decrease in EGFR level. Platelet-derived growth factor generates an acute decrease in the high-affinity binding of EGF to cell surface receptors and an increase in the phosphorylation of EGFR at Thr-654 (39). Insulin-like growth factor I transmodulates EGFR, increasing binding capacity of EGFR without altering EGF binding affinity (40). Retinoic acid and interferon α up-regulate EGFR in some systems and down-regulate it in others (32). Several mitogens, including peptides of the BN family, as well as vasopressin and phorbol esters, can transmodulate EGFR (41). These tumor promoters block tyrosine-specific phosphorylation of the EGFR. Binding of EGF to its receptor results in internalization and lysosomal degradation of the ligand-receptor complex, whereas phorbol esters induce EGFR internalization without degradation (38), in a fashion that may be similar to that caused by our BN antagonists.
Several agents that transmodulate EGFR act via G protein-coupled receptors. Some second messengers of G protein-coupled receptor signaling pathways may affect EGF-caused mitogenesis. The effects of Ca2+ on receptor TKs may be stimulatory or inhibitory depending on the presence or absence of the specific ligand for the receptor (42). Another regulatory mechanism involved in the TK signal transduction cascade is provided by the inhibitory effect of cAMP on activation of Raf by Ras (37). EGFR function is attenuated via protein kinase C-mediated phosphorylation of a submembrane residue of the receptor (37). This phosphorylation results in a suppression of tyrosine kinase of EGFR and decreases high-affinity EGF binding, thus providing a mechanism for heterologous regulation of the EGFR by tumor promoters (41). It was discovered, however, that phosphorylation of the juxtamembrane Thr-686 by protein kinase C does not directly abolish EGFR or c-erbB-2 activity. Thus, other still unidentified mechanisms may be involved in interactions of heterologous growth factor receptors with EGFR in tumor cells (43).
EGFR activity can also be influenced by altering production of the receptor protein that can be monitored by measuring EGFR mRNA expression. In MXT mouse mammary cancers, BN antagonist RC-3095 caused down-regulation of EGFRs and also significantly decreased EGFR synthesis. Production of EGFR may be influenced in different ways (38, 44–48). Thus, exposure of human KB carcinoma cells to EGF results in elevated levels of EGFR mRNA, indicating that the amount of EGFR is regulated by its own ligand (38). Binding of EGF by some human breast cancer cells can be regulated by both progestins and glucocorticoids, acting through their respective receptors and inducing increases in EGFR mRNA levels (44). Up-regulation of EGFR mRNA is most likely due to a direct effect of estrogens on the EGFR gene (45). Exposure to retinoic acid resulted in a 58.5% reduction in EGFR mRNA in head and neck cancer cell lines, as a result of decreased gene transcription (46). Overexpression of EGFR occurs in estrogen receptor-positive breast cancer cells in the absence of estrogen (47). Hormones and growth factors increase the affinity of the high-affinity receptors, while reducing the receptor number and EGFR mRNA levels in sheep pituitary cells. Both EGFR concentration and mRNA levels for EGFR in normal pituitary can be modulated by hormones and growth factors (48). These events are similar to our present findings on EGFR changes in MXT cancers after treatment with the BN/GRP antagonist.
These mechanisms may explain some aspects of the interactions between BN/GRP and the EGF–EGFR system. In contrast, little is known about the mechanisms for how BN antagonists affect EGF signaling pathways. BN antagonists can prevent the effects of BN on EGF binding (41) by blocking elevation of intracellular Ca2+ caused by BN. However, the inhibitory effect of different BN antagonists on Ca2+ mobilization does not correlate with that on cell proliferation (49, 50). Liebow et al. (51) showed that in various carcinomas and cancer cell lines BN enhanced phosphorylating activity of EGFRs, and BN/GRP antagonist RC-3095 inhibited the phosphorylation responses. Thus, BN-like peptides may function by up-regulating EGFRs and the BN antagonists block this action and cause down-regulation of EGFRs (51). Broad spectrum substance P antagonists inhibit BN-stimulated phospholipase C and growth of 3T3 and SCLC cells, but do not affect the Raf1-MEK-mitogen-activated protein kinase pathway (52). In SCLC cells, BN induces an increase in TK activity and tyrosine phosphorylation of polypeptides. This can be inhibited by BN antagonists or TK inhibitors that prevent BN-stimulated proliferation of SCLC cells. It is established that most GRP antagonists have partial agonistic activity. Cells with 3T3 type of BN receptors may respond similarly to BN and BN antagonists (9). The findings that low doses of BN antagonists prevent the growth-promoting effect of BN on SCLC cells, but that higher doses are needed for the suppression of DNA synthesis, indicate that BN antagonists may use mechanisms other than BN antagonism in tumor growth suppression (53).
It is apparent that the effects of BN/GRP antagonists are not limited to their ability to occupy receptor binding sites and to inhibit the binding of the natural ligand. Binding of a BN/GRP antagonist to the BN receptor initiates intracellular changes that may be similar to or different from those induced by the BN agonists. This might indicate that BN/GRP antagonists, in the process of blocking metabolic and mitogenic effects of BN and GRP, appear to induce some intracellular signaling mechanisms that interfere with EGFR pathways and result in growth inhibition of various cancers that depend on the EGF–EGFR system.
Acknowledgments
We thank Amy Giroir, Dora Rigo, and Lisa Karczewski for excellent technical assistance. We are grateful to Prof. J. Engel, Dr. B. Kutscher, and Dr. M. Bernd (Asta Medica, Frankfurt/Main, Germany) for the supply of BN/GRP antagonist RC-3095, and Dr. C. Liebow (State University of New York, Buffalo) for useful suggestions in the preparation of this manuscript. Some work described in this paper was supported by the Medical Research Service of the Department of Veterans Affairs (to A.V.S.).
ABBREVIATIONS
- BN
bombesin
- EGF
epidermal growth factor
- EGFR
epidermal growth factor receptor
- GRP
gastrin releasing peptide
- SCLC
small cell lung cancer
- TK
tyrosine kinase
- i.v.
intravenous
- s.c.
subcutaneous
- RT
reverse transcription
References
- 1.Tache, Y., Melchiorri, P. & Negri, L., eds. (1988) Ann. N.Y. Acad. Sci. 547, 1–538.
- 2.Bunnett N. In: Gut Peptides: Biochemistry and Physiology. Walsh J H, Dockray G J, editors. New York: Raven; 1994. pp. 423–445. [Google Scholar]
- 3.Thomas F, Mormont C, Morgan B. Drugs Future. 1994;19:349–359. [Google Scholar]
- 4.Rozengurt E, Fabregat I, Coffer A, Gil J, Sinnett-Smith J. J Cell Sci Suppl. 1990;13:43–56. doi: 10.1242/jcs.1990.supplement_13.6. [DOI] [PubMed] [Google Scholar]
- 5.Cuttitta F, Carney D N, Mulshine J, Moody T W, Fedorko J, Fischler A, Minna J D. Nature (London) 1985;316:823–826. doi: 10.1038/316823a0. [DOI] [PubMed] [Google Scholar]
- 6.Schally A V, Comaru-Schally A M. In: Cancer Medicine. Holland J F, Frei E III, Bast R C Jr, Kufe D E, Morton D L, Weichselbaum R R, editors. Baltimore: Williams and Wilkins; 1997. pp. 1067–1085. [Google Scholar]
- 7.Radulovic S, Cai R-Z, Serfozo P, Groot K, Redding T W, Pinski J, Schally A V. Int J Peptide Protein Res. 1991;38:593–600. doi: 10.1111/j.1399-3011.1991.tb01545.x. [DOI] [PubMed] [Google Scholar]
- 8.Szepeshazi K, Schally A V, Cai R-Z, Radulovic S, Milovanovic S, Szoke B. Cancer Res. 1991;51:5980–5986. [PubMed] [Google Scholar]
- 9.Szepeshazi K, Schally A V, Groot K, Halmos G. Int J Cancer. 1993;54:282–289. doi: 10.1002/ijc.2910540220. [DOI] [PubMed] [Google Scholar]
- 10.Szepeshazi K, Schally A V, Halmos G, Groot K, Radulovic S. J Natl Cancer Inst. 1992;84:1915–1922. doi: 10.1093/jnci/84.24.1915. [DOI] [PubMed] [Google Scholar]
- 11.Pinski J, Halmos G, Szepeshazi K, Schally A V. Cancer. 1993;72:3263–3270. doi: 10.1002/1097-0142(19931201)72:11<3263::aid-cncr2820721122>3.0.co;2-v. [DOI] [PubMed] [Google Scholar]
- 12.Qin Y, Ertl T, Cai R-Z, Horvath J E, Groot K, Schally A V. Int J Cancer. 1995;63:257–262. doi: 10.1002/ijc.2910630219. [DOI] [PubMed] [Google Scholar]
- 13.Shirahige Y, Cai R-Z, Szepeshazi K, Halmos G, Pinski J, Groot K, Schally A V. Biomed Pharmacother. 1994;48:465–472. doi: 10.1016/0753-3322(94)90007-8. [DOI] [PubMed] [Google Scholar]
- 14.Pinski J, Halmos G, Yano T, Szepeshazi K, Qin Y, Ertl T, Schally A V. Int J Cancer. 1994;57:574–580. doi: 10.1002/ijc.2910570422. [DOI] [PubMed] [Google Scholar]
- 15.Radulovic S, Schally A V, Reile H, Halmos G, Szepeshazi K, Groot K, Milovanovic S, Miller G, Yano T. Acta Oncol. 1994;33:693–701. doi: 10.3109/02841869409121784. [DOI] [PubMed] [Google Scholar]
- 16.Halmos G, Schally A V. Proc Natl Acad Sci USA. 1997;94:956–960. doi: 10.1073/pnas.94.3.956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Pinski J, Schally A V, Halmos G, Szepeshazi K. Int J Cancer. 1993;55:963–967. doi: 10.1002/ijc.2910550615. [DOI] [PubMed] [Google Scholar]
- 18.Yano T, Pinski J, Szepeshazi K, Halmos G, Radulovic S, Groot K, Schally A V. Cancer. 1994;73:1229–1238. doi: 10.1002/1097-0142(19940215)73:4<1229::aid-cncr2820730417>3.0.co;2-1. [DOI] [PubMed] [Google Scholar]
- 19.Davis T P, Crowell S, Taylor J, Clark D L, Coy D, Staley J, Moody T W. Peptides. 1992;13:401–407. doi: 10.1016/0196-9781(92)90128-p. [DOI] [PubMed] [Google Scholar]
- 20.Groot K, Horvath J E, Cai R-Z, Schally A V. Int J Peptide Protein Res. 1995;45:561–566. doi: 10.1111/j.1399-3011.1995.tb01320.x. [DOI] [PubMed] [Google Scholar]
- 21.Paria B C, Das S K, Andrews G K, Dey S K. Proc Natl Acad Sci USA. 1993;90:55–59. doi: 10.1073/pnas.90.1.55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Woll P J, Rozengurt E. Growth Factors. 1988;1:75–83. doi: 10.3109/08977198809000249. [DOI] [PubMed] [Google Scholar]
- 23.Mahmoud S, Palaszynski E, Fiskum G, Coy D H, Moody T W. Life Sci. 1989;44:367–373. doi: 10.1016/0024-3205(89)90231-2. [DOI] [PubMed] [Google Scholar]
- 24.Campbell B J, Garner A, Dockray G J, Hughes J, Dimaline R. Regul Pept. 1994;49:249–255. doi: 10.1016/0167-0115(94)90147-3. [DOI] [PubMed] [Google Scholar]
- 25.Okubo K, Miyasaka K, Matsumoto M, Funakoshi A. Pancreas. 1994;9:585–590. doi: 10.1097/00006676-199409000-00008. [DOI] [PubMed] [Google Scholar]
- 26.Guo Y-S, Thompson J C, Singh P. Gastroenterology. 1990;99:1297–1302. doi: 10.1016/0016-5085(90)91153-w. [DOI] [PubMed] [Google Scholar]
- 27.Varga G, Liehr R-M, Scarpignato C, Coy D H. Eur J Pharmacol. 1995;286:109–112. doi: 10.1016/0014-2999(95)00567-5. [DOI] [PubMed] [Google Scholar]
- 28.Pinski J, Yano T, Groot K, Cai R-Z, Radulovic S, Schally A V. Am J Physiol. 1992;263:E712–E717. doi: 10.1152/ajpendo.1992.263.4.E712. [DOI] [PubMed] [Google Scholar]
- 29.Pinski J, Yano T, Rekasi Z, Cai R-Z, Radulovic S, Schally A V. Regul Pept. 1992;41:185–193. doi: 10.1016/0167-0115(92)90112-8. [DOI] [PubMed] [Google Scholar]
- 30.Liehr R-M, Reidelberger R D, Rosewitz S, Bussjaeger L J, Solomon T E. Regul Pept. 1992;38:207–219. doi: 10.1016/0167-0115(92)90103-2. [DOI] [PubMed] [Google Scholar]
- 31.Camble R, Cotton R, Dutta A S, Garner A, Hayward C F, Moore V E, Scholes P B. Life Sci. 1989;45:1521–1527. doi: 10.1016/0024-3205(89)90417-7. [DOI] [PubMed] [Google Scholar]
- 32.Budillon A, Tagliaferi P, Caraglia M, Torrisi M R, Normanno N, Iacobelli S, Palmieri G, Stoppelli M P, Frati L, Bianco A R. Cancer Res. 1991;51:1294–1299. [PubMed] [Google Scholar]
- 33.Peng D, Fan Z, Lu Y, DeBlasio T, Scher H, Mendelsohn J. Cancer Res. 1996;56:3666–3669. [PubMed] [Google Scholar]
- 34.Robertson K W, Reeves J R, Smith G, Keith W N, Ozanne B W, Cooke T G, Stanton P D. Cancer Res. 1996;56:3823–3830. [PubMed] [Google Scholar]
- 35.Sainsbury J R C, Farndon J R, Needham G K, Malcolm A J, Harris A L. Lancet. 1987;i:1398–1402. doi: 10.1016/s0140-6736(87)90593-9. [DOI] [PubMed] [Google Scholar]
- 36.Nicholson S, Sainsbury J R C, Halcrow P, Chambers P, Farndon J R, Harris A L. Lancet. 1989;i:182–184. doi: 10.1016/s0140-6736(89)91202-6. [DOI] [PubMed] [Google Scholar]
- 37.Dickson R B, Lippman M E. Endocr Rev. 1995;16:559–589. doi: 10.1210/edrv-16-5-559. [DOI] [PubMed] [Google Scholar]
- 38.Clark A J L, Ishii S, Richert N, Merlino G T, Pastan I. Proc Natl Acad Sci USA. 1985;82:8374–8378. doi: 10.1073/pnas.82.24.8374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Countaway J L, Girones N, Davis R J. J Biol Chem. 1989;264:13642–13647. [PubMed] [Google Scholar]
- 40.Krane J F, Murphy D P, Carter D M, Krueger J G. J Invest Dermatol. 1991;96:419–424. doi: 10.1111/1523-1747.ep12469799. [DOI] [PubMed] [Google Scholar]
- 41.Zachary I, Sinnett-Smith J W, Rozengurt E. J Cell Biol. 1986;102:2211–2222. doi: 10.1083/jcb.102.6.2211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Epstein R. Cell Signalling. 1995;7:377–388. doi: 10.1016/0898-6568(95)00006-b. [DOI] [PubMed] [Google Scholar]
- 43.Ouyang X, Gulliford T, Zhang H, Huang G C, Epstein R. J Biol Chem. 1996;271:21786–21792. doi: 10.1074/jbc.271.36.21786. [DOI] [PubMed] [Google Scholar]
- 44.Ewing T M, Murphy L J, Ng M-L, Pang G Y N, Lee C S L, Watts K W, Sutherland R L. Int J Cancer. 1989;44:744–752. doi: 10.1002/ijc.2910440432. [DOI] [PubMed] [Google Scholar]
- 45.Yarden R I, Lauber A H, El-Ashry D, Chrysogelos S A. Endocrinology. 1996;137:2739–2747. doi: 10.1210/endo.137.7.8770893. [DOI] [PubMed] [Google Scholar]
- 46.Grandis J R, Zeng Q, Tweardy D J. Nat Med. 1996;2:237–240. doi: 10.1038/nm0296-237. [DOI] [PubMed] [Google Scholar]
- 47.Miller D L, el-Ashry D, Cheville A L, Liu Y, McLeskey S W, Kern F G. Cell Growth Differ. 1994;5:1263–1274. [PubMed] [Google Scholar]
- 48.Chaidarun S S, Eggo M C, Stewart P M, Sheppard M C. Endocrinology. 1994;143:489–496. doi: 10.1677/joe.0.1430489. [DOI] [PubMed] [Google Scholar]
- 49.Casanueva F F, Perez F R, Casabiell X, Camina J P, Cai R-Z, Schally A V. Proc Natl Acad Sci USA. 1996;93:1406–1411. doi: 10.1073/pnas.93.4.1406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Hirai M, Ishizuka J, Bold R J, Townsend C M, Jr, Thompson J C. Life Sci. 1993;53:1859–1865. doi: 10.1016/0024-3205(93)90493-m. [DOI] [PubMed] [Google Scholar]
- 51.Liebow C, Crean D H, Lee M T, Kamer A R, Mang T S, Schally A V. Proc Natl Acad Sci USA. 1994;91:3804–3808. doi: 10.1073/pnas.91.9.3804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Dhanasekaran N, Heasley L E, Johnson G L. Endocr Rev. 1995;16:259–270. doi: 10.1210/edrv-16-3-259. [DOI] [PubMed] [Google Scholar]
- 53.Takuwa N, Takuwa Y, Ohue Y, Mukai H, Endoh K, Yamashita K, Kumada M, Munekata E. Cancer Res. 1990;50:240–244. [PubMed] [Google Scholar]