Abstract
The herpes simplex virus (HSV) type 1 strict late (gamma) UL38 promoter contains three cis-acting transcriptional elements: a TATA box, a specific initiator element, and the downstream activation sequence (DAS). DAS is located between positions +20 and +33 within the 5' untranslated leader region and strongly influences transcript levels during productive infection. In this communication, we further characterize DAS and investigate its mechanism of action. DAS function has a strict spacing requirement, and DAS contains an essential 6-bp core element. A similarly positioned element from the gamma gC gene (UL44) has partial DAS function within the UL38 promoter context, and the promoter controlling expression of the gamma US11 transcript contains an identically located element with functional and sequence similarity to UL38 DAS. These data suggest that downstream elements are a common feature of many HSV gamma promoters. Results with recombinant viruses containing modifications of the TATA box or initiator element of the UL38 promoter suggest that DAS functions to increase transcription initiation and not the efficiency of transcription elongation. In vitro transcription assays using uninfected HeLa nuclear extracts show that, as in productive infection with recombinant viruses, the deletion of DAS from the UL38 promoter dramatically decreases RNA expression. Finally, electrophoretic mobility shift assays and UV cross-linking experiments show that DAS DNA forms a specific, stable complex with a cellular protein (the DAS-binding factor) of approximately 35 kDa. These data strongly suggest that the interaction of cellular DAS-binding factor with DAS is required for efficient expression of UL38 and other HSV late genes.
Full text
PDF







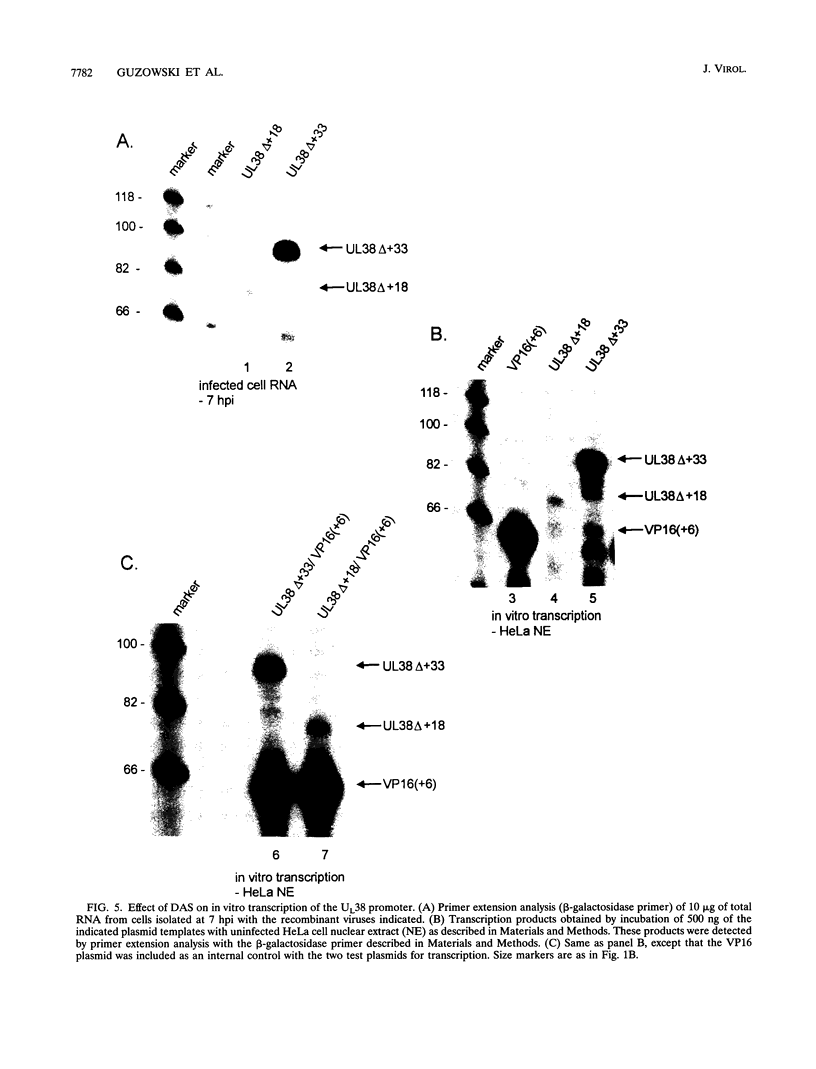







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Blair E. D., Blair C. C., Wagner E. K. Herpes simplex virus virion stimulatory protein mRNA leader contains sequence elements which increase both virus-induced transcription and mRNA stability. J Virol. 1987 Aug;61(8):2499–2508. doi: 10.1128/jvi.61.8.2499-2508.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bohenzky R. A., Papavassiliou A. G., Gelman I. H., Silverstein S. Identification of a promoter mapping within the reiterated sequences that flank the herpes simplex virus type 1 UL region. J Virol. 1993 Feb;67(2):632–642. doi: 10.1128/jvi.67.2.632-642.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cullen B. R. Regulation of HIV-1 gene expression. FASEB J. 1991 Jul;5(10):2361–2368. doi: 10.1096/fasebj.5.10.1712325. [DOI] [PubMed] [Google Scholar]
- Cullen B. R. The HIV-1 Tat protein: an RNA sequence-specific processivity factor? Cell. 1990 Nov 16;63(4):655–657. doi: 10.1016/0092-8674(90)90129-3. [DOI] [PubMed] [Google Scholar]
- Dynlacht B. D., Hoey T., Tjian R. Isolation of coactivators associated with the TATA-binding protein that mediate transcriptional activation. Cell. 1991 Aug 9;66(3):563–576. doi: 10.1016/0092-8674(81)90019-2. [DOI] [PubMed] [Google Scholar]
- Farnham P. J., Means A. L. Sequences downstream of the transcription initiation site modulate the activity of the murine dihydrofolate reductase promoter. Mol Cell Biol. 1990 Apr;10(4):1390–1398. doi: 10.1128/mcb.10.4.1390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flanagan W. M., Papavassiliou A. G., Rice M., Hecht L. B., Silverstein S., Wagner E. K. Analysis of the herpes simplex virus type 1 promoter controlling the expression of UL38, a true late gene involved in capsid assembly. J Virol. 1991 Feb;65(2):769–786. doi: 10.1128/jvi.65.2.769-786.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fridell Y. W., Searles L. L. In vivo transcriptional analysis of the TATA-less promoter of the Drosophila melanogaster vermilion gene. Mol Cell Biol. 1992 Oct;12(10):4571–4577. doi: 10.1128/mcb.12.10.4571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodart S. A., Guzowski J. F., Rice M. K., Wagner E. K. Effect of genomic location on expression of beta-galactosidase mRNA controlled by the herpes simplex virus type 1 UL38 promoter. J Virol. 1992 May;66(5):2973–2981. doi: 10.1128/jvi.66.5.2973-2981.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guzowski J. F., Wagner E. K. Mutational analysis of the herpes simplex virus type 1 strict late UL38 promoter/leader reveals two regions critical in transcriptional regulation. J Virol. 1993 Sep;67(9):5098–5108. doi: 10.1128/jvi.67.9.5098-5108.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halpern M. E., Smiley J. R. Effects of deletions on expression of the herpes simplex virus thymidine kinase gene from the intact viral genome: the amino terminus of the enzyme is dispensable for catalytic activity. J Virol. 1984 Jun;50(3):733–738. doi: 10.1128/jvi.50.3.733-738.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holland L. E., Anderson K. P., Shipman C., Jr, Wagner E. K. Viral DNA synthesis is required for the efficient expression of specific herpes simplex virus type 1 mRNA species. Virology. 1980 Feb;101(1):10–24. doi: 10.1016/0042-6822(80)90479-1. [DOI] [PubMed] [Google Scholar]
- Huang C. J., Goodart S. A., Rice M. K., Guzowski J. F., Wagner E. K. Mutational analysis of sequences downstream of the TATA box of the herpes simplex virus type 1 major capsid protein (VP5/UL19) promoter. J Virol. 1993 Sep;67(9):5109–5116. doi: 10.1128/jvi.67.9.5109-5116.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang C. J., Rice M. K., Devi-Rao G. B., Wagner E. K. The activity of the pseudorabies virus latency-associated transcript promoter is dependent on its genomic location in herpes simplex virus recombinants as well as on the type of cell infected. J Virol. 1994 Mar;68(3):1972–1976. doi: 10.1128/jvi.68.3.1972-1976.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang C. J., Wagner E. K. The herpes simplex virus type 1 major capsid protein (VP5-UL19) promoter contains two cis-acting elements influencing late expression. J Virol. 1994 Sep;68(9):5738–5747. doi: 10.1128/jvi.68.9.5738-5747.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Imbalzano A. N., DeLuca N. A. Substitution of a TATA box from a herpes simplex virus late gene in the viral thymidine kinase promoter alters ICP4 inducibility but not temporal expression. J Virol. 1992 Sep;66(9):5453–5463. doi: 10.1128/jvi.66.9.5453-5463.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Javahery R., Khachi A., Lo K., Zenzie-Gregory B., Smale S. T. DNA sequence requirements for transcriptional initiator activity in mammalian cells. Mol Cell Biol. 1994 Jan;14(1):116–127. doi: 10.1128/mcb.14.1.116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson P. A., Everett R. D. The control of herpes simplex virus type-1 late gene transcription: a 'TATA-box'/cap site region is sufficient for fully efficient regulated activity. Nucleic Acids Res. 1986 Nov 11;14(21):8247–8264. doi: 10.1093/nar/14.21.8247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kibler P. K., Duncan J., Keith B. D., Hupel T., Smiley J. R. Regulation of herpes simplex virus true late gene expression: sequences downstream from the US11 TATA box inhibit expression from an unreplicated template. J Virol. 1991 Dec;65(12):6749–6760. doi: 10.1128/jvi.65.12.6749-6760.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee K. A., Green M. R. Small-scale preparation of extracts from radiolabeled cells efficient in pre-mRNA splicing. Methods Enzymol. 1990;181:20–30. doi: 10.1016/0076-6879(90)81108-7. [DOI] [PubMed] [Google Scholar]
- Mavromara-Nazos P., Roizman B. Activation of herpes simplex virus 1 gamma 2 genes by viral DNA replication. Virology. 1987 Dec;161(2):593–598. doi: 10.1016/0042-6822(87)90156-5. [DOI] [PubMed] [Google Scholar]
- Mavromara-Nazos P., Roizman B. Delineation of regulatory domains of early (beta) and late (gamma 2) genes by construction of chimeric genes expressed in herpes simplex virus 1 genomes. Proc Natl Acad Sci U S A. 1989 Jun;86(11):4071–4075. doi: 10.1073/pnas.86.11.4071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moura-Neto R., Dudov K. P., Perry R. P. An element downstream of the cap site is required for transcription of the gene encoding mouse ribosomal protein L32. Proc Natl Acad Sci U S A. 1989 Jun;86(11):3997–4001. doi: 10.1073/pnas.86.11.3997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakatani Y., Brenner M., Freese E. An RNA polymerase II promoter containing sequences upstream and downstream from the RNA startpoint that direct initiation of transcription from the same site. Proc Natl Acad Sci U S A. 1990 Jun;87(11):4289–4293. doi: 10.1073/pnas.87.11.4289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakatani Y., Horikoshi M., Brenner M., Yamamoto T., Besnard F., Roeder R. G., Freese E. A downstream initiation element required for efficient TATA box binding and in vitro function of TFIID. Nature. 1990 Nov 1;348(6296):86–88. doi: 10.1038/348086a0. [DOI] [PubMed] [Google Scholar]
- Pal S. K., Zinkel S. S., Kiessling A. A., Cooper G. M. c-mos expression in mouse oocytes is controlled by initiator-related sequences immediately downstream of the transcription initiation site. Mol Cell Biol. 1991 Oct;11(10):5190–5196. doi: 10.1128/mcb.11.10.5190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papavassiliou A. G., Wilcox K. W., Silverstein S. J. The interaction of ICP4 with cell/infected-cell factors and its state of phosphorylation modulate differential recognition of leader sequences in herpes simplex virus DNA. EMBO J. 1991 Feb;10(2):397–406. doi: 10.1002/j.1460-2075.1991.tb07961.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pederson N. E., Person S., Homa F. L. Analysis of the gB promoter of herpes simplex virus type 1: high-level expression requires both an 89-base-pair promoter fragment and a nontranslated leader sequence. J Virol. 1992 Oct;66(10):6226–6232. doi: 10.1128/jvi.66.10.6226-6232.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Purnell B. A., Gilmour D. S. Contribution of sequences downstream of the TATA element to a protein-DNA complex containing the TATA-binding protein. Mol Cell Biol. 1993 Apr;13(4):2593–2603. doi: 10.1128/mcb.13.4.2593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Romanelli M. G., Mavromara-Nazos P., Spector D., Roizman B. Mutational analysis of the ICP4 binding sites in the 5' transcribed noncoding domains of the herpes simplex virus 1 UL 49.5 gamma 2 gene. J Virol. 1992 Aug;66(8):4855–4863. doi: 10.1128/jvi.66.8.4855-4863.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sethna M., Weir J. P. Mutational analysis of the herpes simplex virus type 1 glycoprotein E promoter. Virology. 1993 Oct;196(2):532–540. doi: 10.1006/viro.1993.1508. [DOI] [PubMed] [Google Scholar]
- Smale S. T., Baltimore D. The "initiator" as a transcription control element. Cell. 1989 Apr 7;57(1):103–113. doi: 10.1016/0092-8674(89)90176-1. [DOI] [PubMed] [Google Scholar]
- Snowden B. W., Blair E. D., Wagner E. K. Transcriptional activation with concurrent or nonconcurrent template replication has differential effects on transient expression from herpes simplex virus promoters. Virus Genes. 1989 Mar;2(2):129–145. doi: 10.1007/BF00315257. [DOI] [PubMed] [Google Scholar]
- Steffy K. R., Weir J. P. Mutational analysis of two herpes simplex virus type 1 late promoters. J Virol. 1991 Dec;65(12):6454–6460. doi: 10.1128/jvi.65.12.6454-6460.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steffy K. R., Weir J. P. Upstream promoter elements of the herpes simplex virus type 1 glycoprotein H gene. J Virol. 1991 Feb;65(2):972–975. doi: 10.1128/jvi.65.2.972-975.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weir J. P., Narayanan P. R. Expression of the herpes simplex virus type 1 glycoprotein C gene requires sequences in the 5' noncoding region of the gene. J Virol. 1990 Jan;64(1):445–449. doi: 10.1128/jvi.64.1.445-449.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yeh L., Schaffer P. A. A novel class of transcripts expressed with late kinetics in the absence of ICP4 spans the junction between the long and short segments of the herpes simplex virus type 1 genome. J Virol. 1993 Dec;67(12):7373–7382. doi: 10.1128/jvi.67.12.7373-7382.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]


















