Abstract
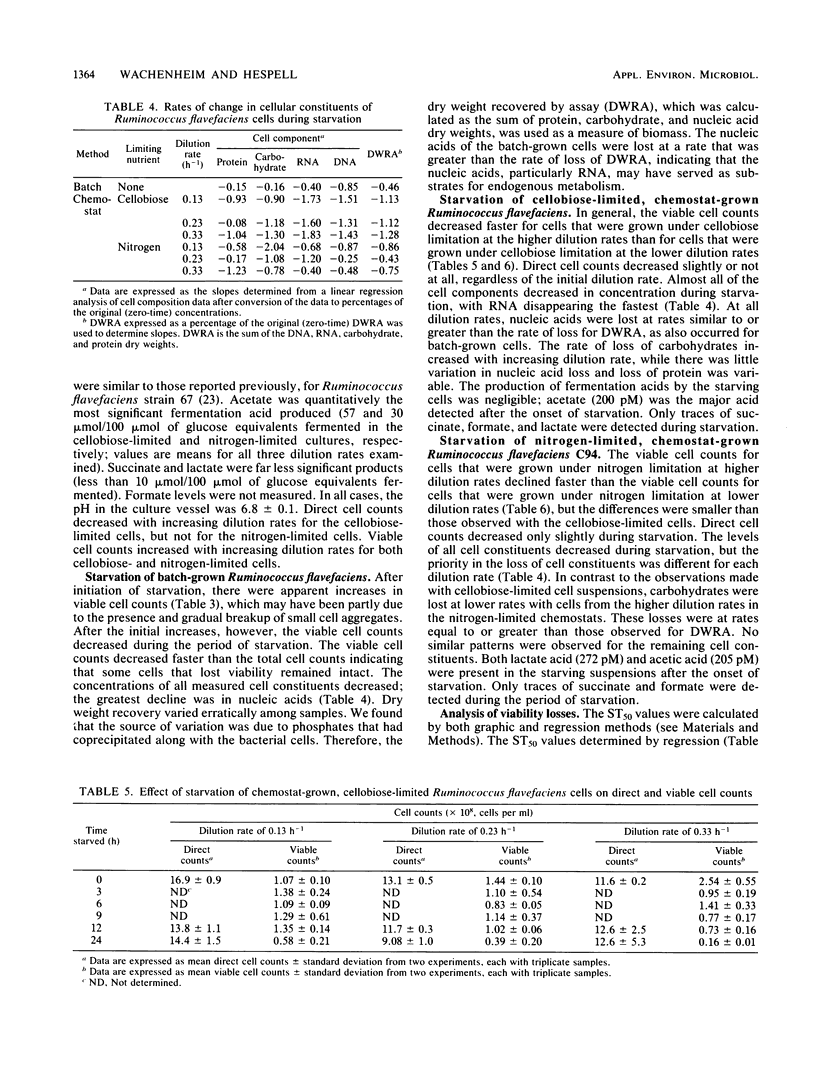
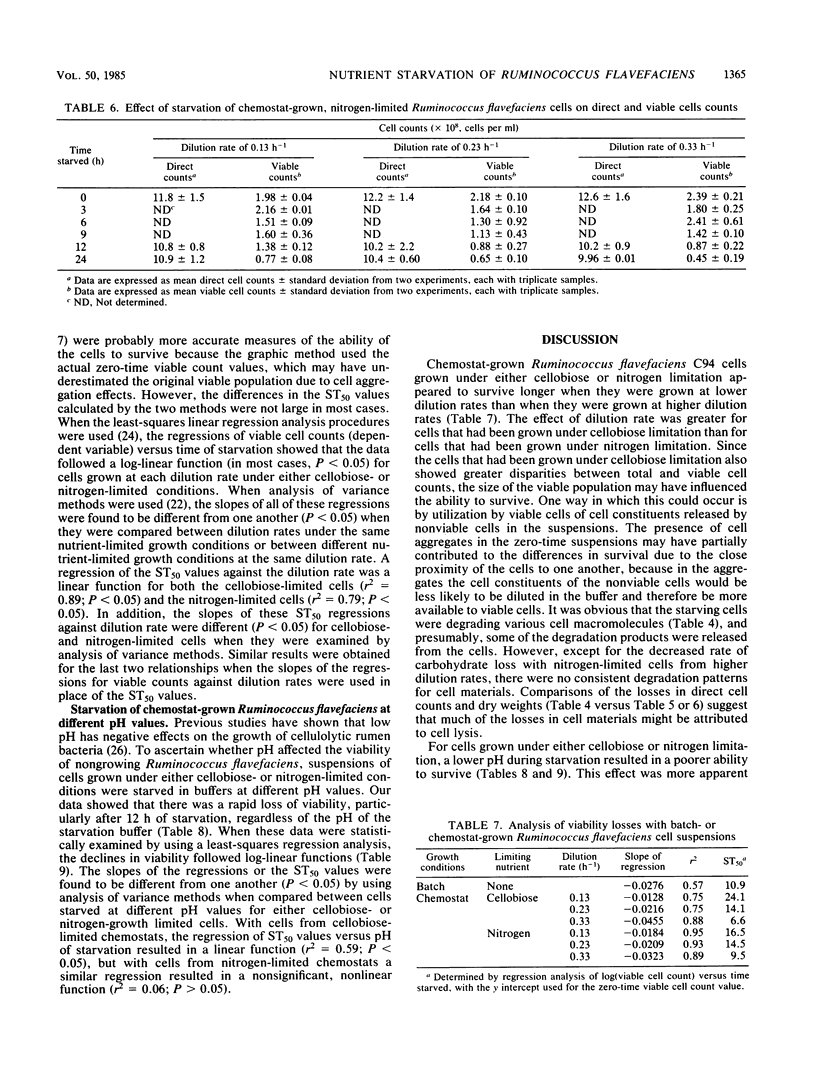
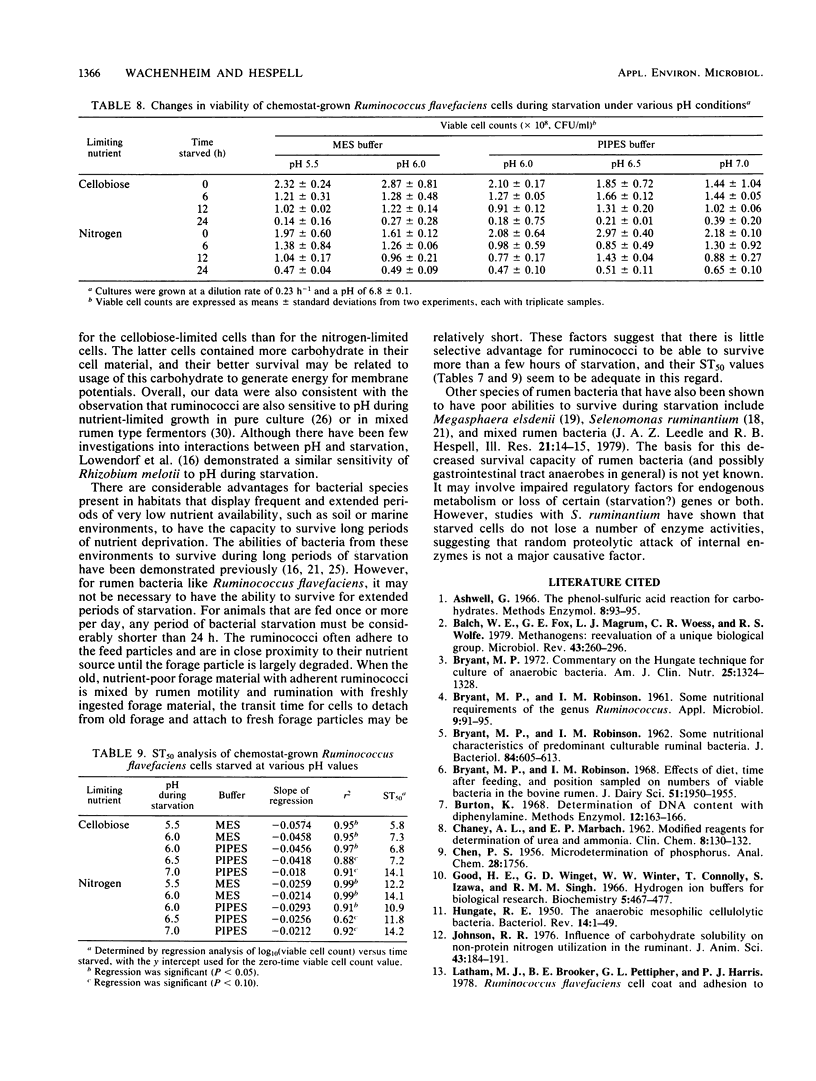
Ruminococcus flavefaciens strain C94, a strictly anaerobic, cellulolytic ruminal bacterial species, was grown either in batch or continuous cultures (cellobiose limited or nitrogen limited) at various dilution rates. Washed cell suspensions were incubated anaerobically at 39°C without nutrients for various times up to 24 h. The effects of starvation on direct and viable cell counts, cell composition (DNA, RNA, protein, and carbohydrate), and endogenous production of volatile fatty acids by the cell suspensions were determined. In addition, the effect of the pH of the starvation buffer on direct and viable cell counts was determined. Survival of batch-grown cells during starvation was variable, with an average time for one-half the cells to lose viability (ST50) of 10.9 h. We found with continuous cultures that viable cell counts declined faster when the initial cell suspensions had been grown at faster dilution rates; this effect was more pronounced for suspensions that had been limited by cellobiose (ST50 = 6.6 h at a dilution rate of 0.33 h−1) than for suspensions that had been limited by nitrogen (ST50 = 9.5 h at a dilution rate of 0.33 h−1). With continuous cultures, viable cell counts in all cases declined faster than direct cell counts did. The rates of disappearance of specific cell components during starvation varied with the initial growth conditions, but could not be correlated with the loss of viability. Volatile fatty acid production by starving cells was very low, and acetate was the main product. Starved cells survived longer at pH 7.0 than they did at pH 5.5, and this effect of pH was greater for cellobiose-limited cells (mean ST50 = 7.1 h) than for nitrogen-limited cells (mean ST50 = 12 h). Although it has relatively low ST50 values, R. flavefaciens has sufficient survival abilities to maintain reasonable numbers in domestic animals having maintenance or greater feed intake.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BRYANT M. P., ROBINSON I. M. Some nutritional characteristics of predominant culturable ruminal bacteria. J Bacteriol. 1962 Oct;84:605–614. doi: 10.1128/jb.84.4.605-614.1962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balch W. E., Fox G. E., Magrum L. J., Woese C. R., Wolfe R. S. Methanogens: reevaluation of a unique biological group. Microbiol Rev. 1979 Jun;43(2):260–296. doi: 10.1128/mr.43.2.260-296.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bryant M. P. Commentary on the Hungate technique for culture of anaerobic bacteria. Am J Clin Nutr. 1972 Dec;25(12):1324–1328. doi: 10.1093/ajcn/25.12.1324. [DOI] [PubMed] [Google Scholar]
- Bryant M. P., Robinson I. M. Effects of diet, time after feeding, and position sampled on numbers of viable bacteria in the bovine rumen. J Dairy Sci. 1968 Dec;51(12):1950–1955. doi: 10.3168/jds.S0022-0302(68)87320-5. [DOI] [PubMed] [Google Scholar]
- Bryant M. P., Robinson I. M. Some Nutritional Requirements of the Genus Ruminococcus. Appl Microbiol. 1961 Mar;9(2):91–95. doi: 10.1128/am.9.2.91-95.1961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CHANEY A. L., MARBACH E. P. Modified reagents for determination of urea and ammonia. Clin Chem. 1962 Apr;8:130–132. [PubMed] [Google Scholar]
- Good N. E., Winget G. D., Winter W., Connolly T. N., Izawa S., Singh R. M. Hydrogen ion buffers for biological research. Biochemistry. 1966 Feb;5(2):467–477. doi: 10.1021/bi00866a011. [DOI] [PubMed] [Google Scholar]
- HUNGATE R. E. The anaerobic mesophilic cellulolytic bacteria. Bacteriol Rev. 1950 Mar;14(1):1–49. doi: 10.1128/br.14.1.1-49.1950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson R. R. Influence of carbohydrate solubility on non-protein nitrogen utilization in the ruminant. J Anim Sci. 1976 Jul;43(1):184–191. doi: 10.2527/jas1976.431184x. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Latham M. J., Brooker B. E., Pettipher G. L., Harris P. J. Ruminococcus flavefaciens Cell Coat and Adhesion to Cotton Cellulose and to Cell Walls in Leaves of Perennial Ryegrass (Lolium perenne). Appl Environ Microbiol. 1978 Jan;35(1):156–165. doi: 10.1128/aem.35.1.156-165.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leedle J. A., Bryant M. P., Hespell R. B. Diurnal variations in bacterial numbers and fluid parameters in ruminal contents of animals fed low- or high-forage diets. Appl Environ Microbiol. 1982 Aug;44(2):402–412. doi: 10.1128/aem.44.2.402-412.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leedle J. A., Hespell R. B. Differential carbohydrate media and anaerobic replica plating techniques in delineating carbohydrate-utilizing subgroups in rumen bacterial populations. Appl Environ Microbiol. 1980 Apr;39(4):709–719. doi: 10.1128/aem.39.4.709-719.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lowendorf H. S., Baya A. M., Alexander M. Survival of Rhizobium in Acid soils. Appl Environ Microbiol. 1981 Dec;42(6):951–957. doi: 10.1128/aem.42.6.951-957.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mink R. W., Hespell R. B. Long-term nutrient starvation of continuously cultured (glucose-limited) Selenomonas ruminantium. J Bacteriol. 1981 Nov;148(2):541–550. doi: 10.1128/jb.148.2.541-550.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mink R. W., Patterson J. A., Hespell R. B. Changes in Viability, Cell Composition, and Enzyme Levels During Starvation of Continuously Cultured (Ammonia-Limited) Selenomonas ruminantium. Appl Environ Microbiol. 1982 Oct;44(4):913–922. doi: 10.1128/aem.44.4.913-922.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Novitsky J. A., Morita R. Y. Survival of a psychrophilic marine Vibrio under long-term nutrient starvation. Appl Environ Microbiol. 1977 Mar;33(3):635–641. doi: 10.1128/aem.33.3.635-641.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russell J. B., Dombrowski D. B. Effect of pH on the efficiency of growth by pure cultures of rumen bacteria in continuous culture. Appl Environ Microbiol. 1980 Mar;39(3):604–610. doi: 10.1128/aem.39.3.604-610.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salanitro J. P., Muirhead P. A. Quantitative method for the gas chromatographic analysis of short-chain monocarboxylic and dicarboxylic acids in fermentation media. Appl Microbiol. 1975 Mar;29(3):374–381. doi: 10.1128/am.29.3.374-381.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schaefer D. M., Davis C. L., Bryant M. P. Ammonia saturation constants for predominant species of rumen bacteria. J Dairy Sci. 1980 Aug;63(8):1248–1263. doi: 10.3168/jds.S0022-0302(80)83076-1. [DOI] [PubMed] [Google Scholar]
- Slyter L. L., Bryant M. P., Wolin M. J. Effect of pH on population and fermentation in a continuously cultured rumen ecosystem. Appl Microbiol. 1966 Jul;14(4):573–578. doi: 10.1128/am.14.4.573-578.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WARNER A. C. Enumeration of rumen micro-organisms. J Gen Microbiol. 1962 Apr;28:119–128. doi: 10.1099/00221287-28-1-119. [DOI] [PubMed] [Google Scholar]


