Abstract
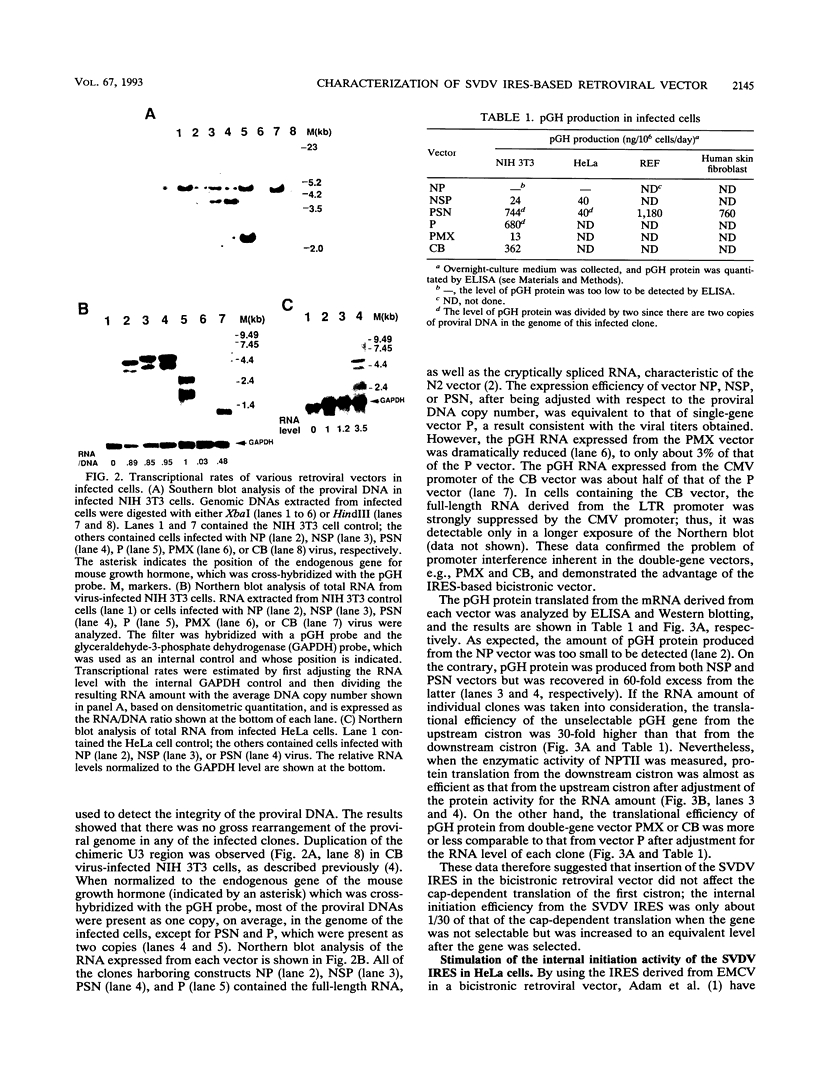
We cloned the 5' nontranslated region (NTR) from the genome of swine vesicular disease virus (SVDV), a member of the family Picornaviridae, and used it to construct a bicistronic retroviral vector. The vector is characterized by coexpression of two genes from a single transcript. We found that inclusion of the 5' NTR of SVDV did not negate the viral vector titer. Protein analysis indicated that the 5' NTR could efficiently direct internal initiation, thus allowing the downstream gene to be translated. Translation of the internally initiated porcine growth hormone gene was about 30-fold less than that when the porcine growth hormone gene was at the upstream position in NIH 3T3 cells but was about equivalent to that in HeLa cells, implying that some cellular factors that stimulated internal initiation of the SVDV 5' NTR are present in HeLa cells. However, in G418-selected clones, the Neor-encoding gene was expressed with equivalent efficiency either at a downstream position or at an upstream position in either NIH 3T3 or HeLa cells. Compared with the conventional double-gene vector or the U3-based vector, the bicistronic vector coexpressed two genes much more efficiently, owing to elimination of promoter interference. Furthermore, this type of vector infected and expressed the target genes efficiently in two primary cell lines, rat embryo and human skin fibroblast cells, which we tested. These experimental data suggest a better design for the retroviral vector and provide evidence that internal initiation of the SVDV 5' NTR was stimulated cell specifically.
Full text
PDF


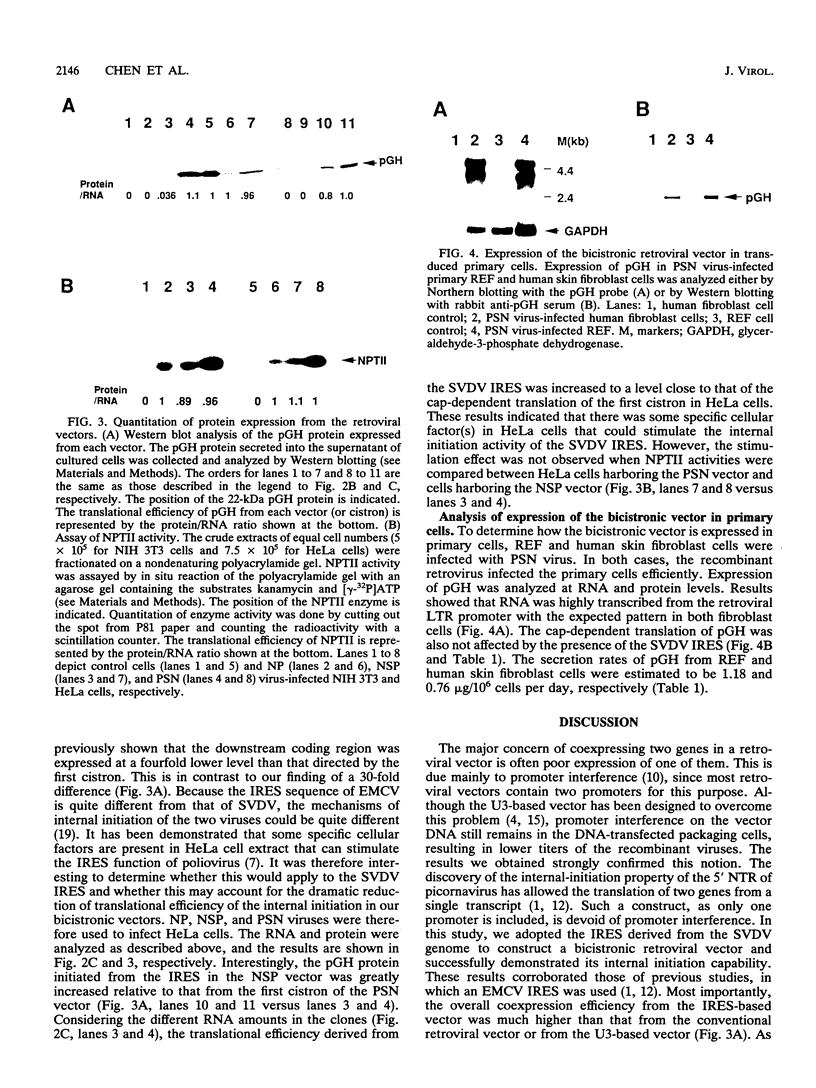


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adam M. A., Ramesh N., Miller A. D., Osborne W. R. Internal initiation of translation in retroviral vectors carrying picornavirus 5' nontranslated regions. J Virol. 1991 Sep;65(9):4985–4990. doi: 10.1128/jvi.65.9.4985-4990.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Armentano D., Yu S. F., Kantoff P. W., von Ruden T., Anderson W. F., Gilboa E. Effect of internal viral sequences on the utility of retroviral vectors. J Virol. 1987 May;61(5):1647–1650. doi: 10.1128/jvi.61.5.1647-1650.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burnette W. N. "Western blotting": electrophoretic transfer of proteins from sodium dodecyl sulfate--polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem. 1981 Apr;112(2):195–203. doi: 10.1016/0003-2697(81)90281-5. [DOI] [PubMed] [Google Scholar]
- Chen B. F., Hsieh C. L., Chen D. S., Hwang L. H. Improved gene expression by a U3-based retroviral vector. Biochem Biophys Res Commun. 1992 Apr 15;184(1):330–337. doi: 10.1016/0006-291x(92)91197-x. [DOI] [PubMed] [Google Scholar]
- Chomczynski P., Sacchi N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem. 1987 Apr;162(1):156–159. doi: 10.1006/abio.1987.9999. [DOI] [PubMed] [Google Scholar]
- Dawe P. S., Forman A. J., Smale C. J. A preliminary investigation of the swine vesicular disease epidemic in Britain. Nature. 1973 Feb 23;241(5391):540–542. doi: 10.1038/241540a0. [DOI] [PubMed] [Google Scholar]
- Dorner A. J., Semler B. L., Jackson R. J., Hanecak R., Duprey E., Wimmer E. In vitro translation of poliovirus RNA: utilization of internal initiation sites in reticulocyte lysate. J Virol. 1984 May;50(2):507–514. doi: 10.1128/jvi.50.2.507-514.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eglitis M. A., Anderson W. F. Retroviral vectors for introduction of genes into mammalian cells. Biotechniques. 1988 Jul-Aug;6(7):608–614. [PubMed] [Google Scholar]
- Emerman M., Temin H. M. Genes with promoters in retrovirus vectors can be independently suppressed by an epigenetic mechanism. Cell. 1984 Dec;39(3 Pt 2):449–467. [PubMed] [Google Scholar]
- Emerman M., Temin H. M. Quantitative analysis of gene suppression in integrated retrovirus vectors. Mol Cell Biol. 1986 Mar;6(3):792–800. doi: 10.1128/mcb.6.3.792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghattas I. R., Sanes J. R., Majors J. E. The encephalomyocarditis virus internal ribosome entry site allows efficient coexpression of two genes from a recombinant provirus in cultured cells and in embryos. Mol Cell Biol. 1991 Dec;11(12):5848–5859. doi: 10.1128/mcb.11.12.5848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graham F. L., van der Eb A. J. A new technique for the assay of infectivity of human adenovirus 5 DNA. Virology. 1973 Apr;52(2):456–467. doi: 10.1016/0042-6822(73)90341-3. [DOI] [PubMed] [Google Scholar]
- Hantzopoulos P. A., Sullenger B. A., Ungers G., Gilboa E. Improved gene expression upon transfer of the adenosine deaminase minigene outside the transcriptional unit of a retroviral vector. Proc Natl Acad Sci U S A. 1989 May;86(10):3519–3523. doi: 10.1073/pnas.86.10.3519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hong T. H., Chen S. T., Tang T. K., Wang S. C., Chang T. H. The production of polyclonal and monoclonal antibodies in mice using novel immunization methods. J Immunol Methods. 1989 Jun 21;120(2):151–157. doi: 10.1016/0022-1759(89)90236-6. [DOI] [PubMed] [Google Scholar]
- Hwang L. H., Chen B. F., Lee P. J., Ho S. Y., Liu J. J. Use of helper-free retroviral vector to direct a high expression of porcine growth hormone in mouse fibroblast cells. Biotechnol Appl Biochem. 1992 Oct;16(2):171–181. [PubMed] [Google Scholar]
- Inoue T., Suzuki T., Sekiguchi K. The complete nucleotide sequence of swine vesicular disease virus. J Gen Virol. 1989 Apr;70(Pt 4):919–934. doi: 10.1099/0022-1317-70-4-919. [DOI] [PubMed] [Google Scholar]
- Jackson R. J., Howell M. T., Kaminski A. The novel mechanism of initiation of picornavirus RNA translation. Trends Biochem Sci. 1990 Dec;15(12):477–483. doi: 10.1016/0968-0004(90)90302-r. [DOI] [PubMed] [Google Scholar]
- Kozak M. The scanning model for translation: an update. J Cell Biol. 1989 Feb;108(2):229–241. doi: 10.1083/jcb.108.2.229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Markowitz D., Goff S., Bank A. Construction and use of a safe and efficient amphotropic packaging cell line. Virology. 1988 Dec;167(2):400–406. [PubMed] [Google Scholar]
- Miller A. D., Rosman G. J. Improved retroviral vectors for gene transfer and expression. Biotechniques. 1989 Oct;7(9):980-2, 984-6, 989-90. [PMC free article] [PubMed] [Google Scholar]
- Mowat G. N., Darbyshire J. H., Huntley J. F. Differentiation of a vesicular disease of pigs in Hong Kong from foot-and-mouth disease. Vet Rec. 1972 May 27;90(22):618–621. doi: 10.1136/vr.90.22.618. [DOI] [PubMed] [Google Scholar]
- Nardelli L., Lodetti E., Gualandi G. L., Burrows R., Goodridge D., Brown F., Cartwright B. A foot and mouth disease syndrome in pigs caused by an enterovirus. Nature. 1968 Sep 21;219(5160):1275–1276. doi: 10.1038/2191275a0. [DOI] [PubMed] [Google Scholar]
- Pelletier J., Kaplan G., Racaniello V. R., Sonenberg N. Translational efficiency of poliovirus mRNA: mapping inhibitory cis-acting elements within the 5' noncoding region. J Virol. 1988 Jul;62(7):2219–2227. doi: 10.1128/jvi.62.7.2219-2227.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pestova T. V., Hellen C. U., Wimmer E. Translation of poliovirus RNA: role of an essential cis-acting oligopyrimidine element within the 5' nontranslated region and involvement of a cellular 57-kilodalton protein. J Virol. 1991 Nov;65(11):6194–6204. doi: 10.1128/jvi.65.11.6194-6204.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reiss B., Sprengel R., Will H., Schaller H. A new sensitive method for qualitative and quantitative assay of neomycin phosphotransferase in crude cell extracts. Gene. 1984 Oct;30(1-3):211–217. doi: 10.1016/0378-1119(84)90122-7. [DOI] [PubMed] [Google Scholar]
- Temin H. M. Retrovirus vectors: promise and reality. Science. 1989 Nov 24;246(4933):983–983. doi: 10.1126/science.2686028. [DOI] [PubMed] [Google Scholar]
- Tracy S., Liu H. L., Chapman N. M. Coxsackievirus B3: primary structure of the 5' non-coding and capsid protein-coding regions of the genome. Virus Res. 1985 Oct;3(3):263–270. doi: 10.1016/0168-1702(85)90050-4. [DOI] [PubMed] [Google Scholar]