Abstract
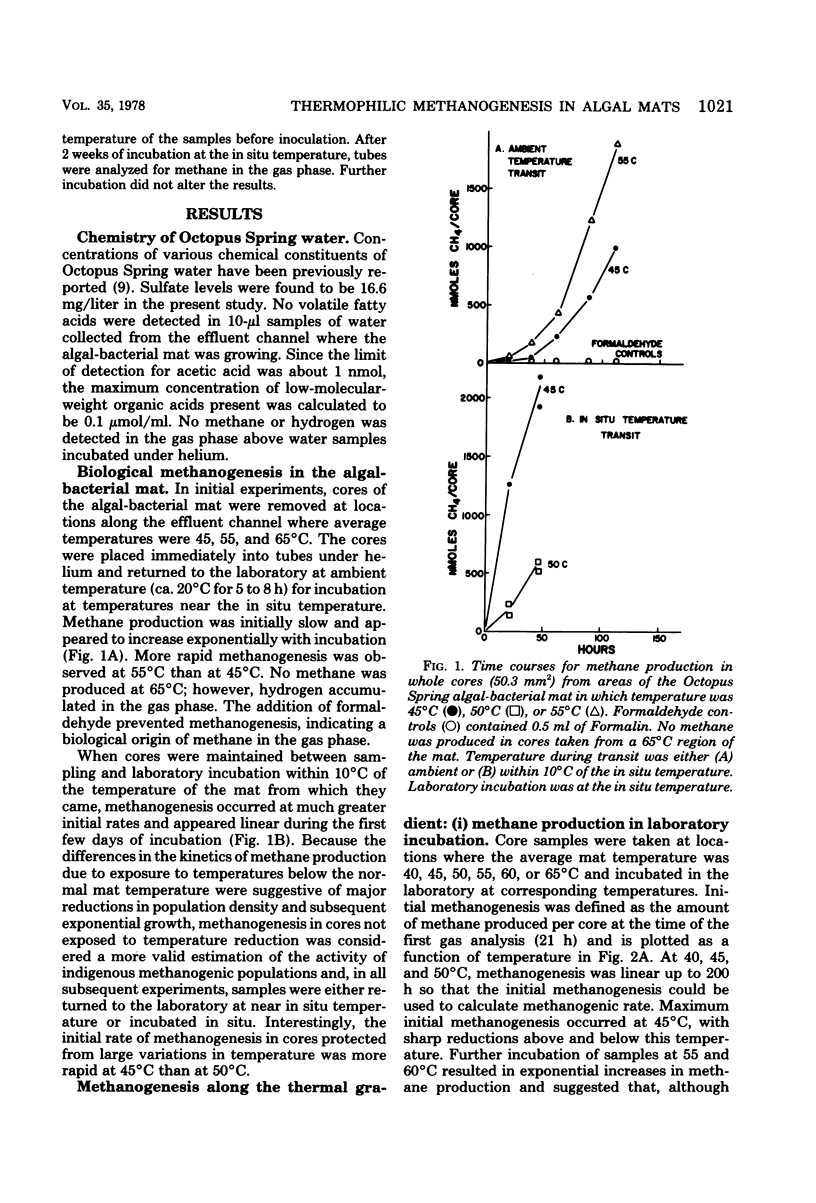
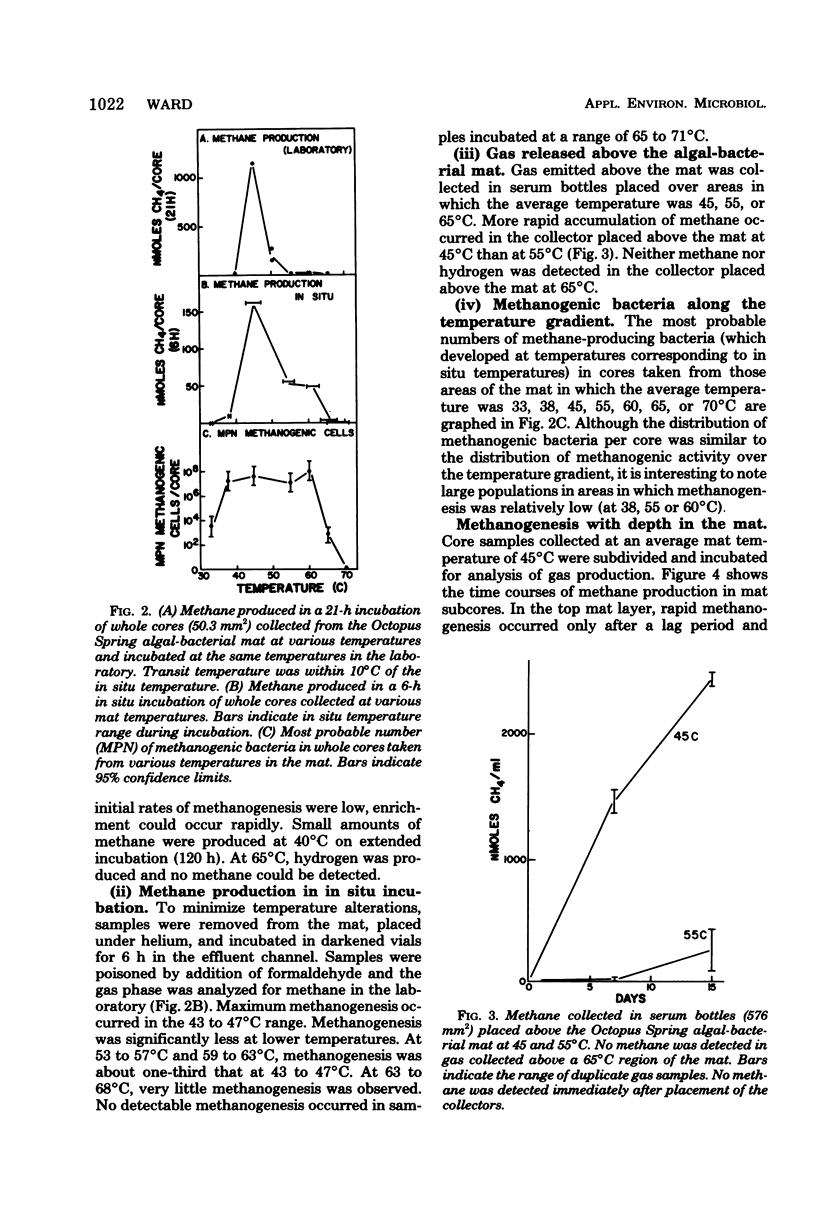
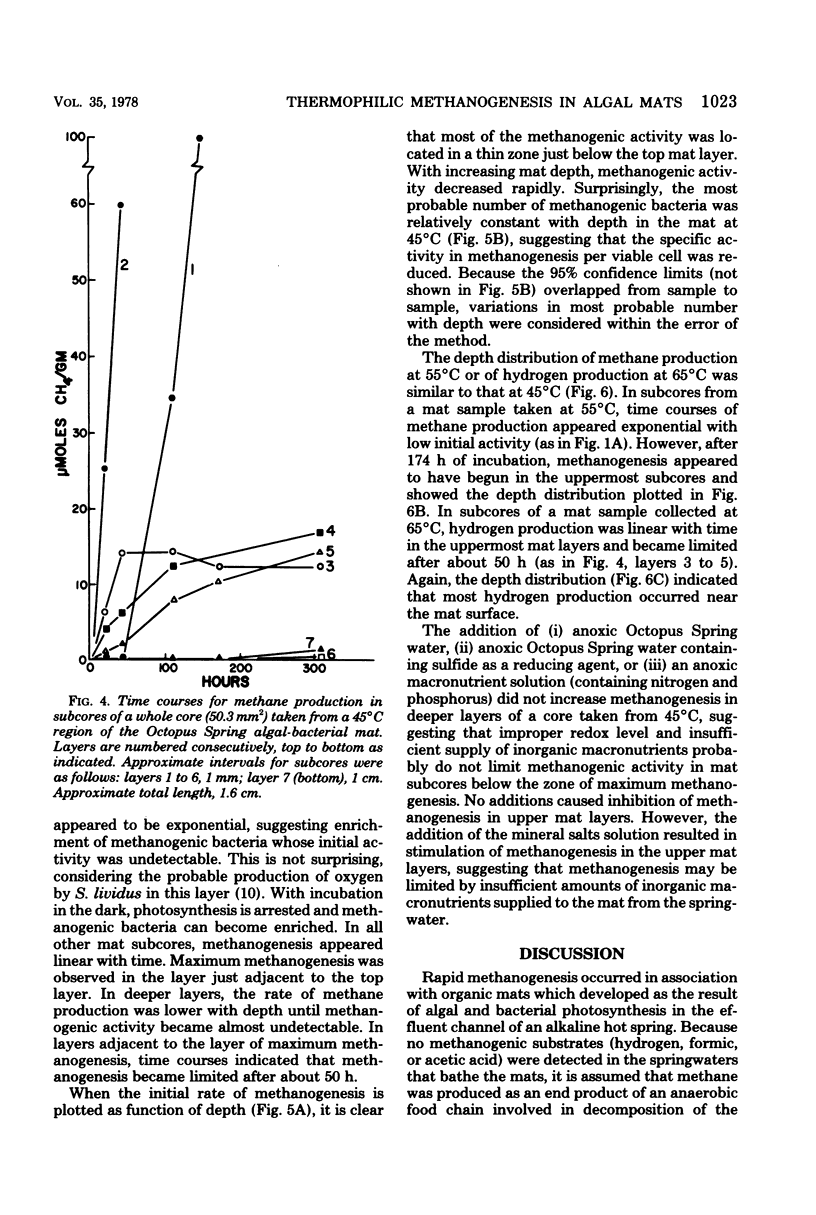
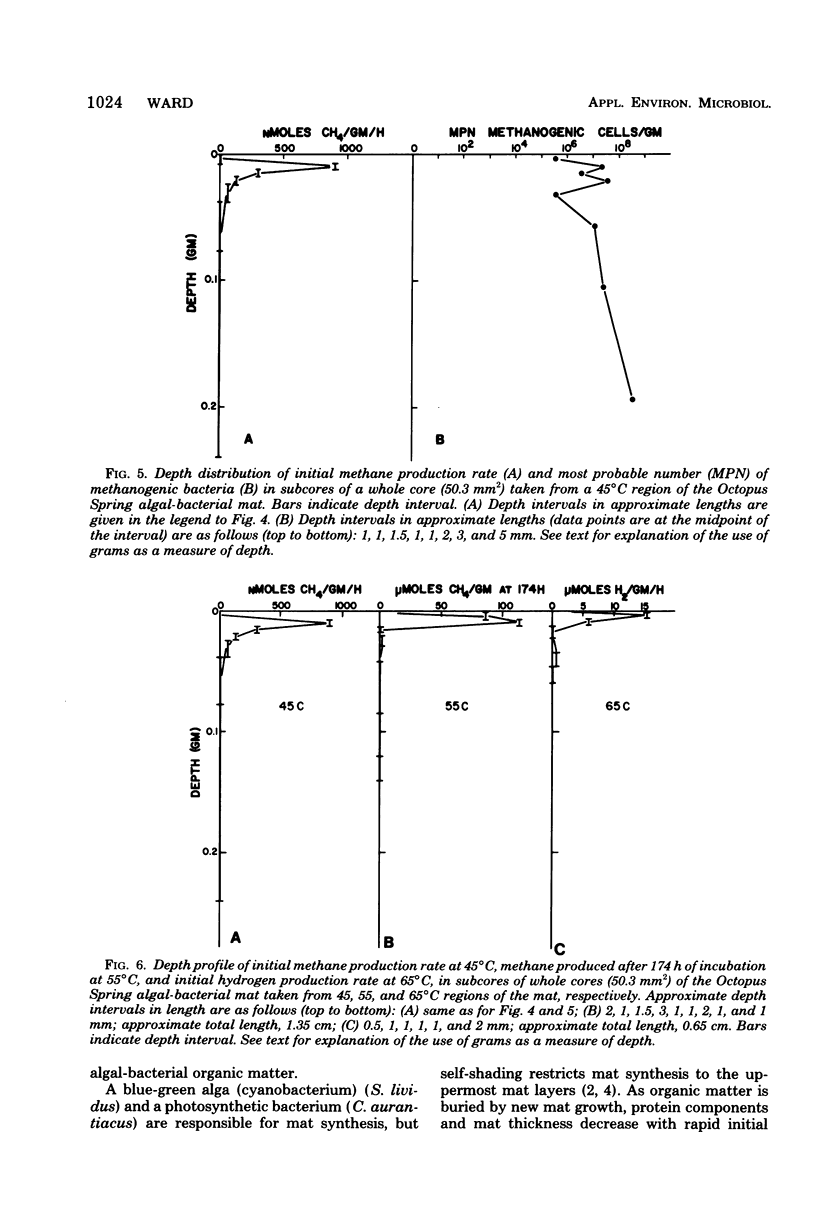
Algal-bacterial mats which grow in the effluent channels of alkaline hot springs provided an environment suitable for studying natural thermophilic methane producing bacteria. Methane was rapidly produced in cores taken from the meat and appeared to be an end product of decomposition of the algal-bacterial organic matter. Formaldehyde prevented production of methane. Initial methanogenic rate was lower and methanogenesis became exponential when samples were permitted to cool before laboratory incubation. Methanogenesis occurred and methanogenic bacteria were present over a range of 68 to 30 degrees C, with optimum methanogenesis near 45 degrees C. The temperature distribution of methanogenesis in the mat is discussed relative to published results on standing crop, primary production, and decomposition in the thermal gradient. The depth distribution of methanogenesis was similar to that of freshwater sediments, with a zone of intense methanogenesis near the mat surface. Methanogenesis in deeper mat layers was very low or undetectable despite large numbers of viable methanogenic bacteria and could not be stimulated by addition of anoxic source water, sulfide, or a macronutrient solution.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Cappenberg T. E. Interrelations between sulfate-reducing and methane-producing bacteria in bottom deposits of a fresh-water lake. I. Field observations. Antonie Van Leeuwenhoek. 1974;40(2):285–295. doi: 10.1007/BF00394387. [DOI] [PubMed] [Google Scholar]
- Cooney C. L. Thermophilic anaerobic digestion of solid waste for fuel gas production. Biotechnol Bioeng. 1975 Aug;17(8):1119–1135. doi: 10.1002/bit.260170804. [DOI] [PubMed] [Google Scholar]
- Doemel W. N., Brock T. D. Structure, growth, and decomposition of laminated algal-bacterial mats in alkaline hot springs. Appl Environ Microbiol. 1977 Oct;34(4):433–452. doi: 10.1128/aem.34.4.433-452.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mah R. A., Ward D. M., Baresi L., Glass T. L. Biogenesis of methane. Annu Rev Microbiol. 1977;31:309–341. doi: 10.1146/annurev.mi.31.100177.001521. [DOI] [PubMed] [Google Scholar]
- Varel V. H., Isaacson H. R., Bryant M. P. Thermophilic methane production from cattle waste. Appl Environ Microbiol. 1977 Feb;33(2):298–307. doi: 10.1128/aem.33.2.298-307.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winfrey M. R., Zeikus J. G. Effect of sulfate on carbon and electron flow during microbial methanogenesis in freshwater sediments. Appl Environ Microbiol. 1977 Feb;33(2):275–281. doi: 10.1128/aem.33.2.275-281.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolfe R. S. Microbial formation of methane. Adv Microb Physiol. 1971;6:107–146. doi: 10.1016/s0065-2911(08)60068-5. [DOI] [PubMed] [Google Scholar]
- Zeikus J. G. The biology of methanogenic bacteria. Bacteriol Rev. 1977 Jun;41(2):514–541. doi: 10.1128/br.41.2.514-541.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeikus J. G., Wolfe R. S. Methanobacterium thermoautotrophicus sp. n., an anaerobic, autotrophic, extreme thermophile. J Bacteriol. 1972 Feb;109(2):707–715. doi: 10.1128/jb.109.2.707-713.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zinder S. H., Doemel W. N., Brock T. D. Production of volatile sulfur compounds during the decomposition of algal mats. Appl Environ Microbiol. 1977 Dec;34(6):859–860. doi: 10.1128/aem.34.6.859-860.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]


