Abstract
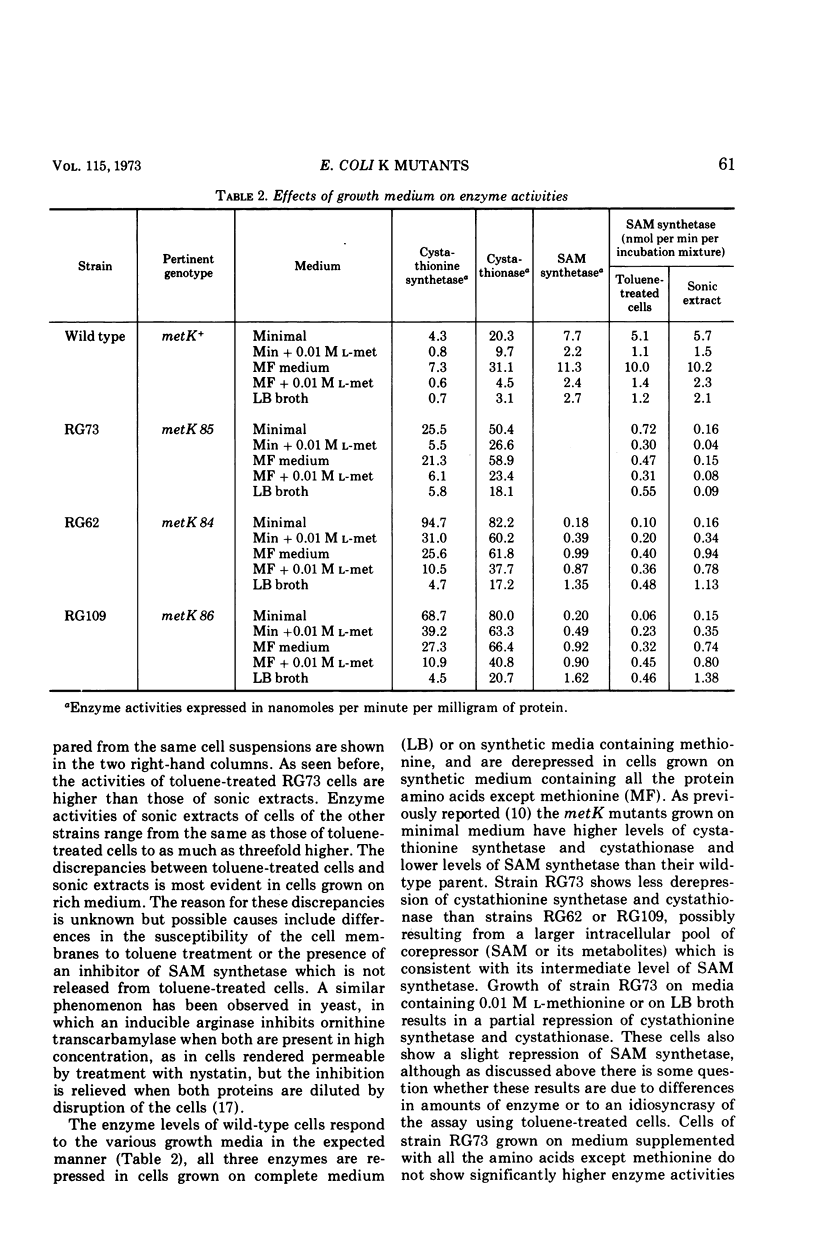
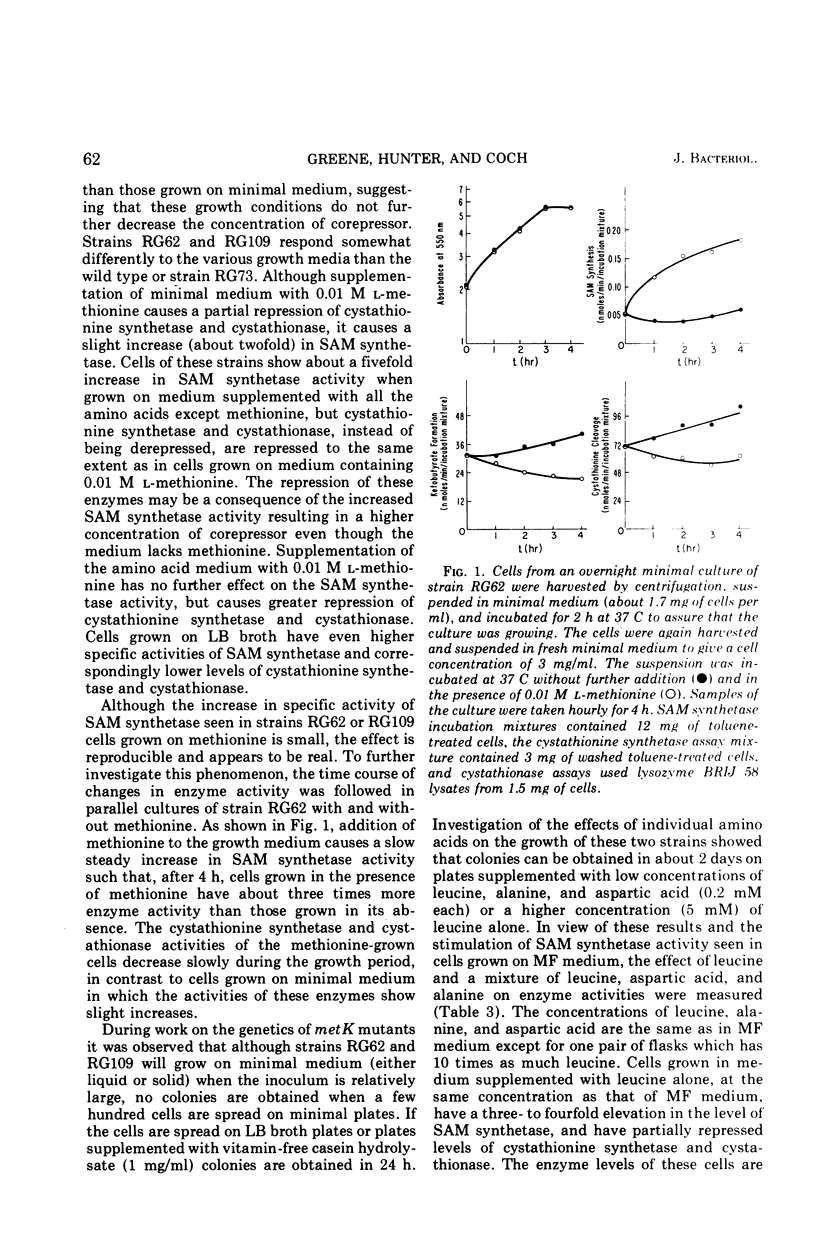
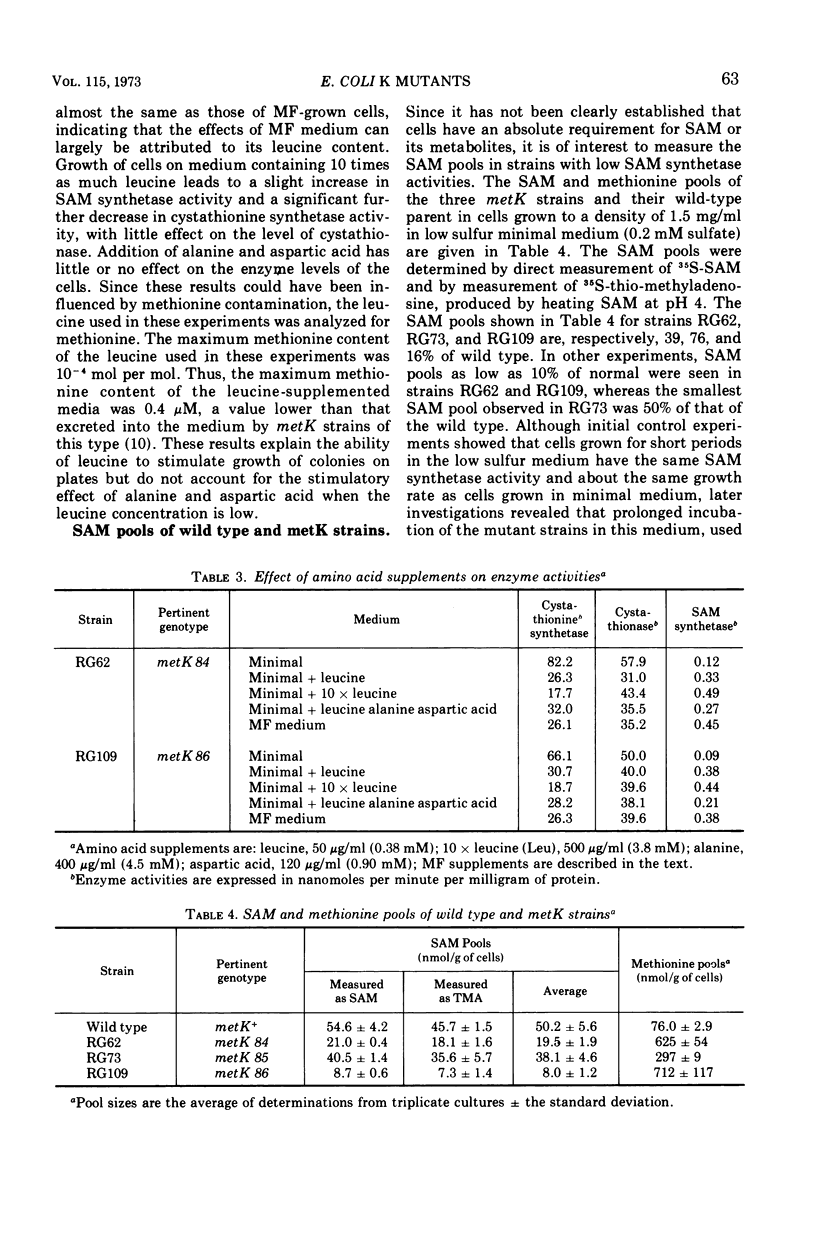
Some of the properties of three metK mutants of Escherichia coli K-12 have been examined. All three strains have lower than normal levels of SAM (S-adenosyl-l-methionine) synthetase and elevated levels of cystathionine synthetase and cystathionase. One strain (RG73) appears to have an unstable SAM synthetase, suggesting that it carries a structural gene mutation. The two strains (RG62 and RG109) which have the lowest levels of SAM synthetase when grown on minimal medium have appreciably higher levels of enzyme when grown on complete medium. Growth on defined media supplemented with leucine or methionine causes a several-fold increase in the specific activity of SAM synthetase with associated decreases in cystathionine synthetase and cystathionase, but the changes are not as large as those seen in cells grown on LB broth. The SAM pools of strains RG62 and RG109 are markedly lower than normal while that of strain RG73 is slightly below normal. The methionine pools of all three strains are elevated several-fold. The metK strains are able to synthesize cyclopropane fatty acids, but the rate of their formation is slowed. Modification and restriction of phage 21 appears to be normal, suggesting that these strains are able to methylate DNA.
Full text
PDF







Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BERTANI G., WEIGLE J. J. Host controlled variation in bacterial viruses. J Bacteriol. 1953 Feb;65(2):113–121. doi: 10.1128/jb.65.2.113-121.1953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chater K. F. Dominance of the wild-type alleles of methionine regulatory genes in Salmonella typhimurium. J Gen Microbiol. 1970 Sep;63(1):95–109. doi: 10.1099/00221287-63-1-95. [DOI] [PubMed] [Google Scholar]
- Cronan J. E., Jr Phospholipid alterations during growth of Escherichia coli. J Bacteriol. 1968 Jun;95(6):2054–2061. doi: 10.1128/jb.95.6.2054-2061.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DAVIS B. D., MINGIOLI E. S. Mutants of Escherichia coli requiring methionine or vitamin B12. J Bacteriol. 1950 Jul;60(1):17–28. doi: 10.1128/jb.60.1.17-28.1950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flavin M., Slaughter C. Synthesis of the succinic ester of homoserine, a new intermediate in the bacterial biosynthesis of methionine. Biochemistry. 1965 Jul;4(7):1370–1375. doi: 10.1021/bi00883a022. [DOI] [PubMed] [Google Scholar]
- GOLDFINE H., BLOCH K. On the origin of unsaturated fatty acids in clostridia. J Biol Chem. 1961 Oct;236:2596–2601. [PubMed] [Google Scholar]
- Godson G. N., Sinsheimer R. L. Lysis of Escherichia coli with a neutral detergent. Biochim Biophys Acta. 1967 Dec 19;149(2):476–488. doi: 10.1016/0005-2787(67)90175-x. [DOI] [PubMed] [Google Scholar]
- Greene R. C., Su C. H., Holloway C. T. S-Adenosylmethionine synthetase deficient mutants of Escherichia coli K-12 with impaired control of methionine biosynthesis. Biochem Biophys Res Commun. 1970 Mar 27;38(6):1120–1126. doi: 10.1016/0006-291x(70)90355-4. [DOI] [PubMed] [Google Scholar]
- Haberman A., Heywood J., Meselson M. DNA modification methylase activity of Escherichia coli restriction endonucleases K and P. Proc Natl Acad Sci U S A. 1972 Nov;69(11):3138–3141. doi: 10.1073/pnas.69.11.3138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holloway C. T., Greene R. C., Su C. H. Regulation of S-adenosylmethionine synthetase in Escherichia coli. J Bacteriol. 1970 Nov;104(2):734–747. doi: 10.1128/jb.104.2.734-747.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawrence D. A., Smith D. A., Rowbury R. J. Regulation of methionine synthesis in Salmonella typhimurium: mutants resistant to inhibition by analogues of methionine. Genetics. 1968 Apr;58(4):473–492. doi: 10.1093/genetics/58.4.473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lowry O. H., Carter J., Ward J. B., Glaser L. The effect of carbon and nitrogen sources on the level of metabolic intermediates in Escherichia coli. J Biol Chem. 1971 Nov;246(21):6511–6521. [PubMed] [Google Scholar]
- Maas W. K. Mapping of genes involved in the synthesis of spermidine in Escherichia coli. Mol Gen Genet. 1972;119(1):1–9. doi: 10.1007/BF00270439. [DOI] [PubMed] [Google Scholar]
- Meselson M., Yuan R. DNA restriction enzyme from E. coli. Nature. 1968 Mar 23;217(5134):1110–1114. doi: 10.1038/2171110a0. [DOI] [PubMed] [Google Scholar]
- Messenguy F., Wiame J. -M. The control of ornithinetranscarbamylase activity by arginase in Saccharomyces cerevisiae. FEBS Lett. 1969 Apr;3(1):47–49. doi: 10.1016/0014-5793(69)80093-1. [DOI] [PubMed] [Google Scholar]
- Minson A. C., Smith D. A. Methionine regulatory defects in Salmonella typhimurium arising from amber-suppressible mutations. J Gen Microbiol. 1972 May;70(3):471–476. doi: 10.1099/00221287-70-3-471. [DOI] [PubMed] [Google Scholar]
- NOVICK R. P., MAAS W. K. Control by endogenously synthesized arginine of the formation of ornithine transcarbamylase in Escherichia coli. J Bacteriol. 1961 Feb;81:236–240. doi: 10.1128/jb.81.2.236-240.1961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- PATTERSON M. S., GREENE R. C. MEASUREMENT OF LOW ENERGY BETA-EMITTERS IN AQUEOUS SOLUTION BY LIQUID SCINTILLATION COUNTING OF EMULSIONS. Anal Chem. 1965 Jun;37:854–857. doi: 10.1021/ac60226a017. [DOI] [PubMed] [Google Scholar]
- Piperno J. R., Oxender D. L. Amino acid transport systems in Escherichia coli K-12. J Biol Chem. 1968 Nov 25;243(22):5914–5920. [PubMed] [Google Scholar]
- SCHLENK F., DAINKO J. L., STANFORD S. M. Improved procedure for the isolation of S-adenosylmethionine and S-adenosylethionine. Arch Biochem Biophys. 1959 Jul;83(1):28–34. doi: 10.1016/0003-9861(59)90006-2. [DOI] [PubMed] [Google Scholar]
- Savin M. A., Flavin M., Slaughter C. Regulation of homocysteine biosynthesis in Salmonella typhimurium. J Bacteriol. 1972 Aug;111(2):547–556. doi: 10.1128/jb.111.2.547-556.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlenk F., Zydek-Cwick C. R. Stabilit of the glycosidic bond of S-adenosylsulfonium compounds toward acid. Arch Biochem Biophys. 1969 Nov;134(2):414–422. doi: 10.1016/0003-9861(69)90301-4. [DOI] [PubMed] [Google Scholar]
- Scott J. R. Genetic studies on bacteriophage P1. Virology. 1968 Dec;36(4):564–574. doi: 10.1016/0042-6822(68)90188-8. [DOI] [PubMed] [Google Scholar]
- Shapiro S. K., Ehninger D. J. Methods for the analysis and preparation of adenosylmethionine and adenosylhomocysteine. Anal Biochem. 1966 May;15(2):323–333. doi: 10.1016/0003-2697(66)90038-8. [DOI] [PubMed] [Google Scholar]
- Silbert D. F., Ruch F., Vagelos P. R. Fatty acid replacements in a fatty acid auxotroph of Escherichia coli. J Bacteriol. 1968 May;95(5):1658–1665. doi: 10.1128/jb.95.5.1658-1665.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Su C. H., Greene R. C. Regulation of methionine biosynthesis in Escherichia coli: mapping of the metJ locus and properties of a metJ plus-metJ minus diploid. Proc Natl Acad Sci U S A. 1971 Feb;68(2):367–371. doi: 10.1073/pnas.68.2.367. [DOI] [PMC free article] [PubMed] [Google Scholar]


