Abstract
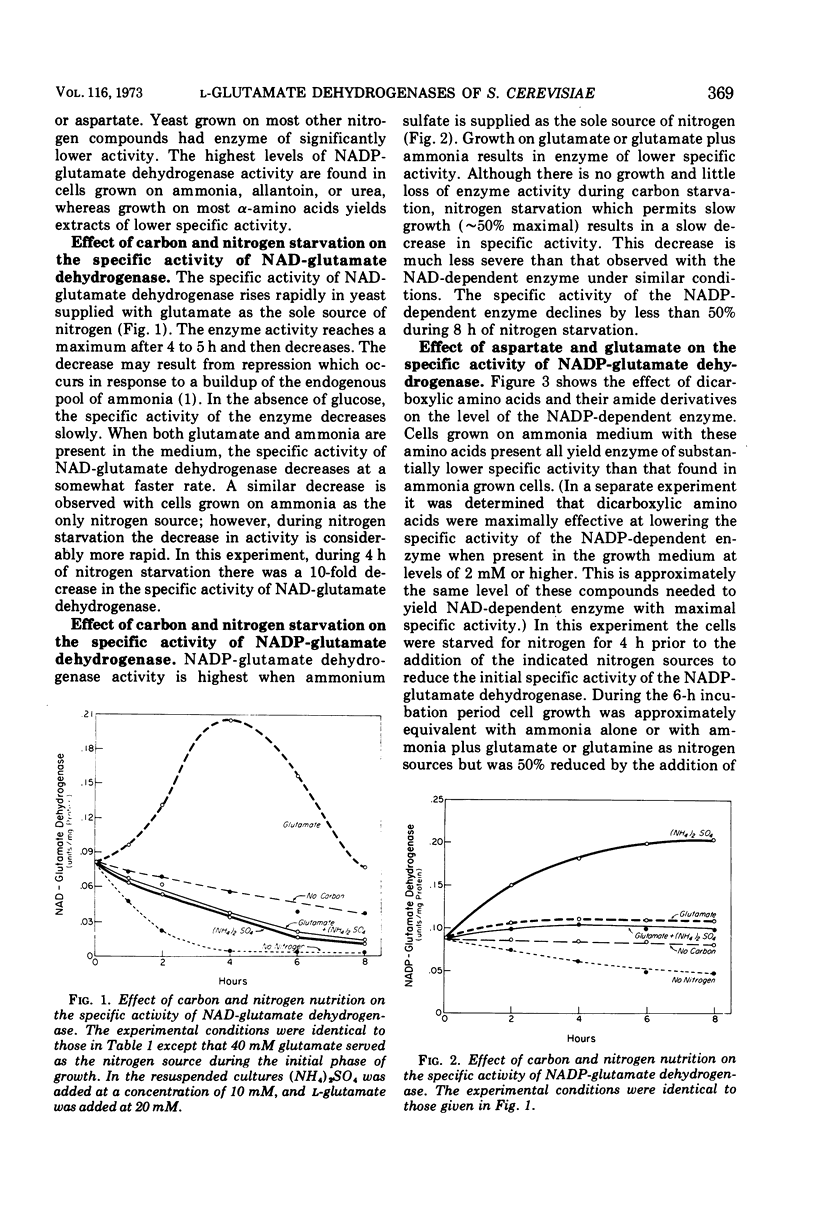
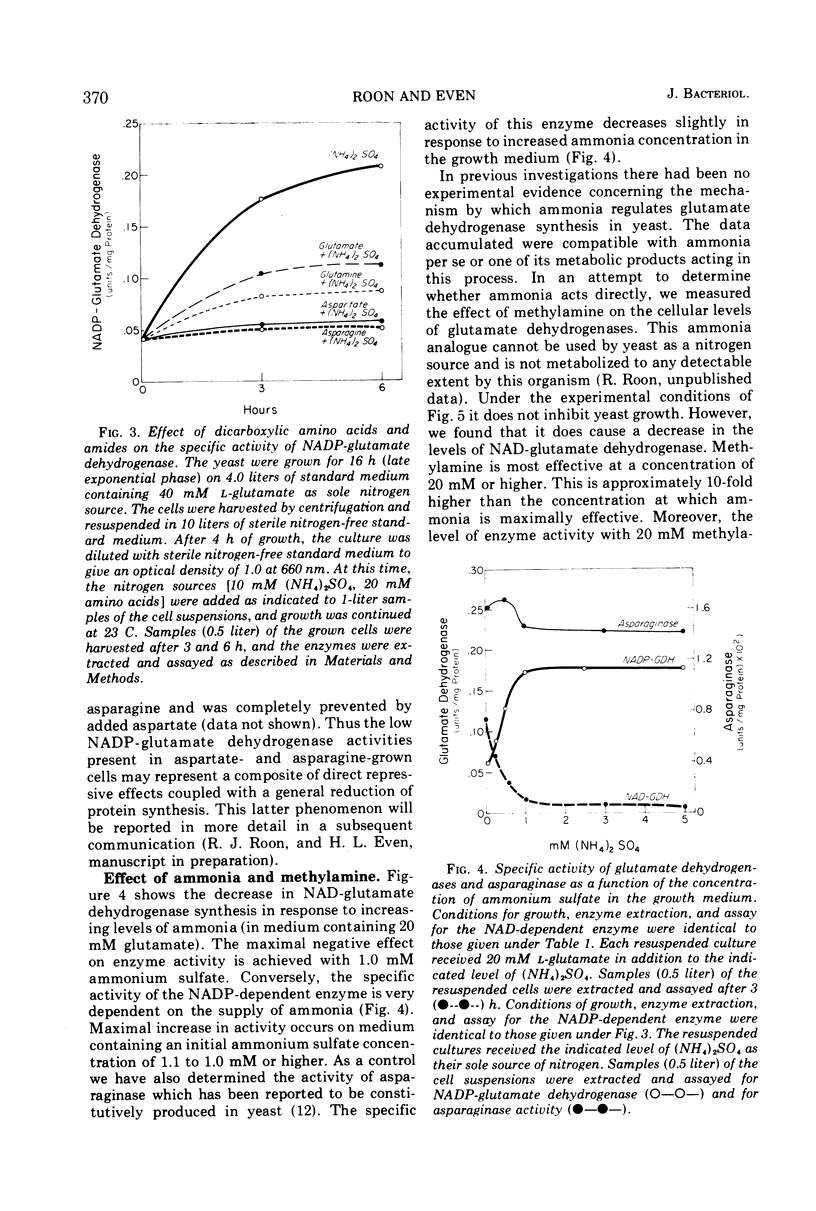
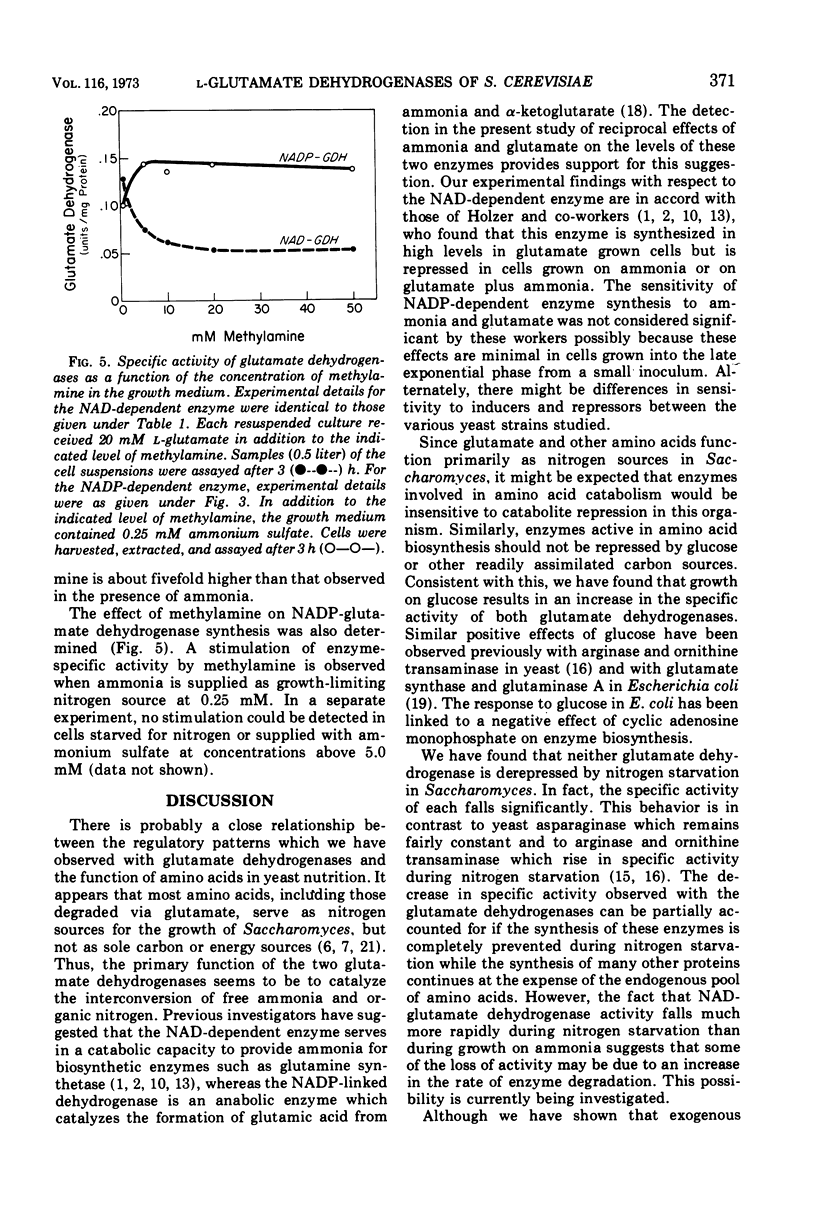
Saccharomyces cerevisiae contains two distinct l-glutamate dehydrogenases. These enzymes are affected in a reciprocal fashion by growth on ammonia or dicarboxylic amino acids as the nitrogen source. The specific activity of the nicotinamide adenine dinucleotide phosphate (NADP) (anabolic) enzyme is highest in ammonia-grown cells and is reduced in cells grown on glutamate or aspartate. Conversely, the specific activity of the nicotinamide adenine dinucleotide (NAD) (catabolic) glutamate dehydrogenase is highest in cells grown on glutamate or aspartate and is much lower in cells grown on ammonia. The specific activity of both enzymes is very low in nitrogen-starved yeast. Addition of the ammonia analogue methylamine to the growth medium reduces the specific activity of the NAD-dependent enzyme and increases the specific activity of the NADP-dependent enzyme.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bernhardt W., Panten K., Holzer H. Gedämpftes Oscillieren der Synthesegeschwindigkeit von DPN-Abhängiger Glutamatdehydrogenase in Hefezellen. Biochim Biophys Acta. 1965 Jun 22;99(3):531–539. [PubMed] [Google Scholar]
- Bernhardt W., Zink M., Holzer H. NAD-abhängige Glutamatdehydrogenase aus reprimierter und dereprimierter Bäckerhefe. Biochim Biophys Acta. 1966 Jun 15;118(3):549–555. [PubMed] [Google Scholar]
- Dubois E., Grenson M., Wiame J. M. Release of the "ammonia effect" on three catabolic enzymes by NADP-specific glutamate dehydrogenaseless mutations in Saccharomyces cerevisiae. Biochem Biophys Res Commun. 1973 Feb 20;50(4):967–972. doi: 10.1016/0006-291x(73)91500-3. [DOI] [PubMed] [Google Scholar]
- Grenson M., Hou C. Ammonia inhibition of the general amino acid permease and its suppression in NADPH-specific glutamate dehydrogenaseless mutants of saccharomyces cerevisiae. Biochem Biophys Res Commun. 1972 Aug 21;48(4):749–756. doi: 10.1016/0006-291x(72)90670-5. [DOI] [PubMed] [Google Scholar]
- HIERHOLZER G., HOLZER H. REPRESSION DER SYNTHESE VON DPN-ABHAENGIGER GLUTAMINSAEUREDEHYDROGENASE IN SACCHAROMYCES CEREVISIAE DURCH AMMONIUMIONEN. Biochem Z. 1963 Dec 3;339:175–185. [PubMed] [Google Scholar]
- HOLZER H., SCHNEIDER S. Anreicherung und Trennung einer DPN-spezifischen und einer TPN-spezifischen Glutaminsäure-dehydrogenase aus Hefe. Biochem Z. 1957;329(5):361–369. [PubMed] [Google Scholar]
- Jones G. E., Mortimer R. K. L-asparaginase-deficient mutants of yeast. Science. 1970 Jan 9;167(3915):181–182. doi: 10.1126/science.167.3915.181. [DOI] [PubMed] [Google Scholar]
- KOHLHAW G., DRAEGERT W., HOLZER H. PARALLEL-REPRESSION DER SYNTHESE VON GLUTAMIN-SYNTHETASE UND DPN-ABHAENGIGER GLUTAMAT-DEHYDROGENASE IN HEFE. Biochem Z. 1965 Feb 8;341:224–238. [PubMed] [Google Scholar]
- MCGILVERY R. W., MOKRASCH L. C. Purification and properties of fructose-1, 6-diphosphatase. J Biol Chem. 1956 Aug;221(2):909–917. [PubMed] [Google Scholar]
- Middelhoven W. J. Induction and repression of arginase and ornithine transaminase in baker's yeast. Antonie Van Leeuwenhoek. 1970;36(1):1–19. doi: 10.1007/BF02069003. [DOI] [PubMed] [Google Scholar]
- Middelhoven W. J. The derepression of arginase and of ornithine transaminase in nitrogen-starved baker's yeast. Biochim Biophys Acta. 1968 Mar 11;156(2):440–443. doi: 10.1016/0304-4165(68)90284-5. [DOI] [PubMed] [Google Scholar]
- Nuñez de Castro I., Ugarte M., Cano A., Mayor F. Effect of glucose, galactose, and different nitrogen-sources on the activity of yeast glutamate dehydrogenase (NAD and NADP-linked) from normal strain and impaired respiration mutant. Eur J Biochem. 1970 Nov;16(3):567–570. doi: 10.1111/j.1432-1033.1970.tb01118.x. [DOI] [PubMed] [Google Scholar]
- Polakis E. S., Bartley W. Changes in the enzyme activities of Saccharomyces cerevisiae during aerobic growth on different carbon sources. Biochem J. 1965 Oct;97(1):284–297. doi: 10.1042/bj0970284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prusiner S., Miller R. E., Valentine R. C. Adenosine 3':5'-cyclic monophosphate control of the enzymes of glutamine metabolism in Escherichia coli. Proc Natl Acad Sci U S A. 1972 Oct;69(10):2922–2926. doi: 10.1073/pnas.69.10.2922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomulka K. W., Moat A. G. Inorganic nitrogen assimilation in yeasts: alteration in enzyme activities associated with changes in cultural conditions and growth phase. J Bacteriol. 1972 Jan;109(1):25–33. doi: 10.1128/jb.109.1.25-33.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]


