Abstract
Transformation of rodent cells by human adenoviruses is a well-established model system for studying the expression, regulation, and function of class I antigens. In this report, we demonstrate that the highly oncogenic adenovirus type 12 operates at the transcriptional and posttranscriptional levels in regulating the activity of major histocompatibility complex class I genes and products in transformed cells. Adenovirus type 12 suppresses the cell surface expression of class I antigens in most cell lines. Nevertheless, in a number of cell lines suppression is the result of reduction in the amount of stable specific mRNA, while in another group of cell lines suppression involves interference with processing of a posttranscriptional product. The two mechanisms operate both for the endogenous H-2 genes and for a miniature swine class I transgene that is expressed in the cells.
Full text
PDF
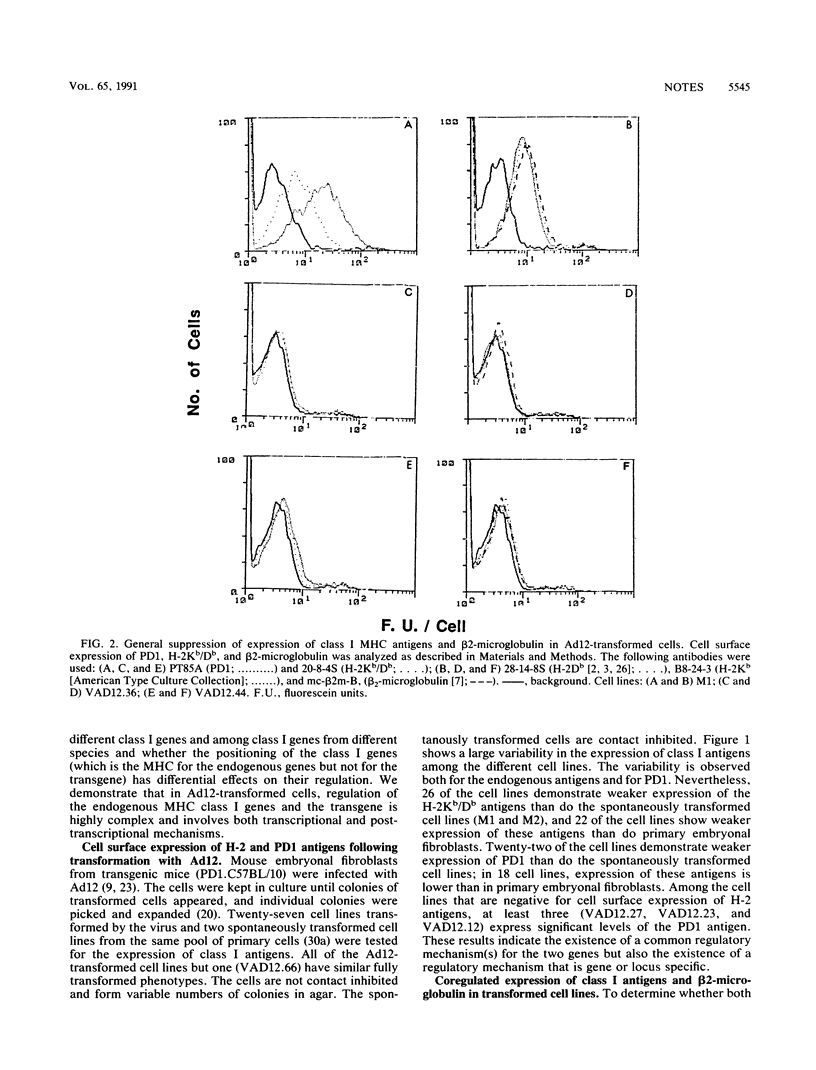
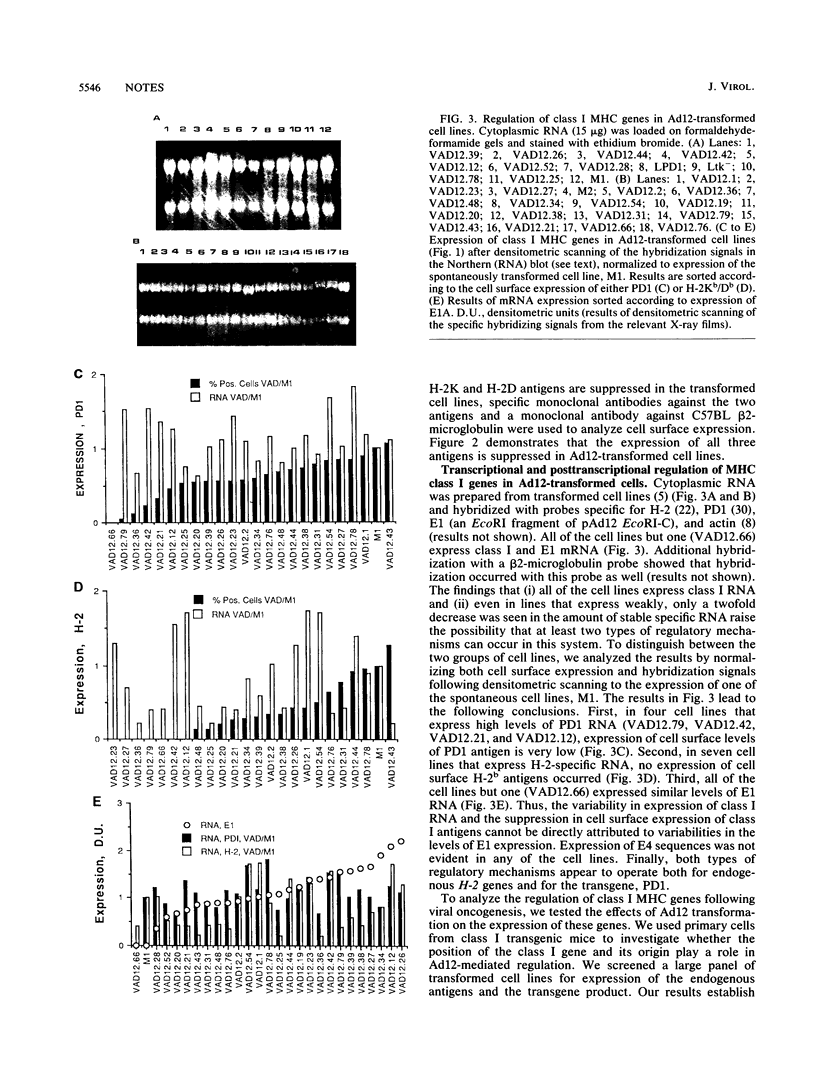


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ackrill A. M., Blair G. E. Regulation of major histocompatibility class I gene expression at the level of transcription in highly oncogenic adenovirus transformed rat cells. Oncogene. 1988 Oct;3(4):483–487. [PubMed] [Google Scholar]
- Allen H., Fraser J., Flyer D., Calvin S., Flavell R. Beta 2-microglobulin is not required for cell surface expression of the murine class I histocompatibility antigen H-2Db or of a truncated H-2Db. Proc Natl Acad Sci U S A. 1986 Oct;83(19):7447–7451. doi: 10.1073/pnas.83.19.7447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allen H., Wraith D., Pala P., Askonas B., Flavell R. A. Domain interactions of H-2 class I antigens alter cytotoxic T-cell recognition sites. Nature. 1984 May 17;309(5965):279–281. doi: 10.1038/309279a0. [DOI] [PubMed] [Google Scholar]
- Andersson M., Päbo S., Nilsson T., Peterson P. A. Impaired intracellular transport of class I MHC antigens as a possible means for adenoviruses to evade immune surveillance. Cell. 1985 Nov;43(1):215–222. doi: 10.1016/0092-8674(85)90026-1. [DOI] [PubMed] [Google Scholar]
- Berger S. L. Isolation of cytoplasmic RNA: ribonucleoside-vanadyl complexes. Methods Enzymol. 1987;152:227–234. doi: 10.1016/0076-6879(87)52024-9. [DOI] [PubMed] [Google Scholar]
- Burgert H. G., Maryanski J. L., Kvist S. "E3/19K" protein of adenovirus type 2 inhibits lysis of cytolytic T lymphocytes by blocking cell-surface expression of histocompatibility class I antigens. Proc Natl Acad Sci U S A. 1987 Mar;84(5):1356–1360. doi: 10.1073/pnas.84.5.1356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chorney M., Shen F. W., Michaelson J., Boyse E. A. Monoclonal antibody to an alloantigenic determinant on beta2-microglobulin (beta 2M) of the mouse. Immunogenetics. 1982;16(1):91–93. doi: 10.1007/BF00364446. [DOI] [PubMed] [Google Scholar]
- Cleveland D. W., Lopata M. A., MacDonald R. J., Cowan N. J., Rutter W. J., Kirschner M. W. Number and evolutionary conservation of alpha- and beta-tubulin and cytoplasmic beta- and gamma-actin genes using specific cloned cDNA probes. Cell. 1980 May;20(1):95–105. doi: 10.1016/0092-8674(80)90238-x. [DOI] [PubMed] [Google Scholar]
- Cook J. L., Lewis A. M., Jr Host response to adenovirus 2-transformed hamster embryo cells. Cancer Res. 1979 May;39(5):1455–1461. [PubMed] [Google Scholar]
- Davis W. C., Marusic S., Lewin H. A., Splitter G. A., Perryman L. E., McGuire T. C., Gorham J. R. The development and analysis of species specific and cross reactive monoclonal antibodies to leukocyte differentiation antigens and antigens of the major histocompatibility complex for use in the study of the immune system in cattle and other species. Vet Immunol Immunopathol. 1987 Jul;15(4):337–376. doi: 10.1016/0165-2427(87)90005-5. [DOI] [PubMed] [Google Scholar]
- Eager K. B., Williams J., Breiding D., Pan S., Knowles B., Appella E., Ricciardi R. P. Expression of histocompatibility antigens H-2K, -D, and -L is reduced in adenovirus-12-transformed mouse cells and is restored by interferon gamma. Proc Natl Acad Sci U S A. 1985 Aug;82(16):5525–5529. doi: 10.1073/pnas.82.16.5525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ehrlich R., Maguire J. E., Singer D. S. Identification of negative and positive regulatory elements associated with a class I major histocompatibility complex gene. Mol Cell Biol. 1988 Feb;8(2):695–703. doi: 10.1128/mcb.8.2.695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ehrlich R., Sharrow S. O., Maguire J. E., Singer D. S. Expression of a class I MHC transgene: effects of in vivo alpha/beta-interferon treatment. Immunogenetics. 1989;30(1):18–26. doi: 10.1007/BF02421465. [DOI] [PubMed] [Google Scholar]
- Frels W. I., Bluestone J. A., Hodes R. J., Capecchi M. R., Singer D. S. Expression of a microinjected porcine class I major histocompatibility complex gene in transgenic mice. Science. 1985 May 3;228(4699):577–580. doi: 10.1126/science.3885396. [DOI] [PubMed] [Google Scholar]
- Friedman D. J., Ricciardi R. P. Adenovirus type 12 E1A gene represses accumulation of MHC class I mRNAs at the level of transcription. Virology. 1988 Jul;165(1):303–305. doi: 10.1016/0042-6822(88)90689-7. [DOI] [PubMed] [Google Scholar]
- Goodenow R. S., Vogel J. M., Linsk R. L. Histocompatibility antigens on murine tumors. Science. 1985 Nov 15;230(4727):777–783. doi: 10.1126/science.2997918. [DOI] [PubMed] [Google Scholar]
- Green M., Wold W. S. Human adenoviruses: growth, purification, and transfection assay. Methods Enzymol. 1979;58:425–435. doi: 10.1016/s0076-6879(79)58157-9. [DOI] [PubMed] [Google Scholar]
- Haddada H., Lewis A. M., Jr, Sogn J. A., Coligan J. E., Cook J. L., Walker T. A., Levine A. S. Tumorigenicity of hamster and mouse cells transformed by adenovirus types 2 and 5 is not influenced by the level of class I major histocompatibility antigens expressed on the cells. Proc Natl Acad Sci U S A. 1986 Dec;83(24):9684–9688. doi: 10.1073/pnas.83.24.9684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hämmerling G. J., Klar D., Pülm W., Momburg F., Moldenhauer G. The influence of major histocompatibility complex class I antigens on tumor growth and metastasis. Biochim Biophys Acta. 1987 Nov 25;907(3):245–259. doi: 10.1016/0304-419x(87)90008-4. [DOI] [PubMed] [Google Scholar]
- Katoh S., Ozawa K., Kondoh S., Soeda E., Israel A., Shiroki K., Fujinaga K., Itakura K., Gachelin G., Yokoyama K. Identification of sequences responsible for positive and negative regulation by E1A in the promoter of H-2Kbm1 class I MHC gene. EMBO J. 1990 Jan;9(1):127–135. doi: 10.1002/j.1460-2075.1990.tb08088.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lalanne J. L., Delarbre C., Gachelin G., Kourilsky P. A cDNA clone containing the entire coding sequence of a mouse H-2Kd histocompatibility antigen. Nucleic Acids Res. 1983 Mar 11;11(5):1567–1577. doi: 10.1093/nar/11.5.1567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lewis A. M., Jr, Cook J. L. Spectrum of tumorigenic phenotypes among adenovirus 2-, adenovirus 12-, and simian virus 40-transformed Syrian hamster cells defined by host cellular immune-tumor cell interactions. Cancer Res. 1982 Mar;42(3):939–944. [PubMed] [Google Scholar]
- Meijer I., Jochemsen A. G., de Wit C. M., Bos J. L., Morello D., van der Eb A. J. Adenovirus type 12 E1A down regulates expression of a transgene under control of a major histocompatibility complex class I promoter: evidence for transcriptional control. J Virol. 1989 Sep;63(9):4039–4042. doi: 10.1128/jvi.63.9.4039-4042.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mellow G. H., Föhring B., Dougherty J., Gallimore P. H., Raska K., Jr Tumorigenicity of adenovirus-transformed rat cells and expression of class I major histocompatibility antigen. Virology. 1984 Apr 30;134(2):460–465. doi: 10.1016/0042-6822(84)90313-1. [DOI] [PubMed] [Google Scholar]
- Ozato K., Sachs D. H. Monoclonal antibodies to mouse MHC antigens. III. Hybridoma antibodies reacting to antigens of the H-2b haplotype reveal genetic control of isotype expression. J Immunol. 1981 Jan;126(1):317–321. [PubMed] [Google Scholar]
- Rosenthal A., Wright S., Quade K., Gallimore P., Cedar H., Grosveld F. Increased MHC H-2K gene transcription in cultured mouse embryo cells after adenovirus infection. Nature. 1985 Jun 13;315(6020):579–581. doi: 10.1038/315579a0. [DOI] [PubMed] [Google Scholar]
- Schrier P. I., Bernards R., Vaessen R. T., Houweling A., van der Eb A. J. Expression of class I major histocompatibility antigens switched off by highly oncogenic adenovirus 12 in transformed rat cells. 1983 Oct 27-Nov 2Nature. 305(5937):771–775. doi: 10.1038/305771a0. [DOI] [PubMed] [Google Scholar]
- Singer D. S., Camerini-Otero R. D., Satz M. L., Osborne B., Sachs D., Rudikoff S. Characterization of a porcine genomic clone encoding a major histocompatibility antigen: expression in mouse L cells. Proc Natl Acad Sci U S A. 1982 Mar;79(5):1403–1407. doi: 10.1073/pnas.79.5.1403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanaka K., Isselbacher K. J., Khoury G., Jay G. Reversal of oncogenesis by the expression of a major histocompatibility complex class I gene. Science. 1985 Apr 5;228(4695):26–30. doi: 10.1126/science.3975631. [DOI] [PubMed] [Google Scholar]
- Vaessen R. T., Houweling A., van der Eb A. J. Post-transcriptional control of class I MHC mRNA expression in adenovirus 12-transformed cells. Science. 1987 Mar 20;235(4795):1486–1488. doi: 10.1126/science.3823900. [DOI] [PubMed] [Google Scholar]
- Vasavada R., Eager K. B., Barbanti-Brodano G., Caputo A., Ricciardi R. P. Adenovirus type 12 early region 1A proteins repress class I HLA expression in transformed human cells. Proc Natl Acad Sci U S A. 1986 Jul;83(14):5257–5261. doi: 10.1073/pnas.83.14.5257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yewdell J. W., Bennink J. R., Eager K. B., Ricciardi R. P. CTL recognition of adenovirus-transformed cells infected with influenza virus: lysis by anti-influenza CTL parallels adenovirus-12-induced suppression of class I MHC molecules. Virology. 1988 Jan;162(1):236–238. doi: 10.1016/0042-6822(88)90413-8. [DOI] [PubMed] [Google Scholar]
- Zinkernagel R. M., Doherty P. C. MHC-restricted cytotoxic T cells: studies on the biological role of polymorphic major transplantation antigens determining T-cell restriction-specificity, function, and responsiveness. Adv Immunol. 1979;27:51–177. doi: 10.1016/s0065-2776(08)60262-x. [DOI] [PubMed] [Google Scholar]