Abstract
OBJECTIVE—Engraftment and function of human islet cell implants is considered to be dependent on their rapid and adequate revascularization. Studies with rodent islet grafts have shown that vascular endothelial growth factor (VEGF) expression by β-cells can promote this process. The present work examines whether human islet preparations produce VEGF as well as interleukin (IL)-8, another angiogenic protein, and assesses the role of contaminating duct cells in VEGF and IL-8–mediated angiogenesis.
RESEARCH DESIGN AND METHODS—Human islet and pancreatic duct cell preparations are compared for their respective expression and production of VEGF and IL-8 during culture as well as following transplantation in nonobese diabetic (NOD)/scid mice. The associated angiogenic effects are measured in an in vitro aortic ring assay and in an in vivo chick embryo chorioallantoic membrane assay.
RESULTS—Cultured pancreatic duct cells expressed 3- and 10-fold more VEGF and IL-8, respectively, than cultured human islet endocrine cells and released both proteins at angiogenic levels. The angiogenic effect of purified duct cells was higher than that of purified endocrine islet cells and was completely blocked by a combination of IL-8 and VEGF antibodies. Human duct cell implants under the kidney capsule of NOD/scid mice expressed higher levels of IL-8 and VEGF than human islet cell implants and induced circulating IL-8 and VEGF levels during the first day posttransplantation.
CONCLUSIONS—Human duct cell–released IL-8 and VEGF may help revascularization of currently used human islet cell grafts. Further work should examine whether and when this effect can prevail over other inflammatory and immune influences of this cell type.
Islet cell transplant protocols have been designed to restore type 1 diabetes in patients, but the function of the implants was found to progressively decrease, leading to <15% insulin-independent recipients after 5 years (1). Several variables can explain this loss in metabolic efficacy. We have observed that the functional β-cell mass in insulin-independent recipients at posttransplant month 12 is only 20–30% of that in normal control subjects (2). It is so far unknown to which extent this quantitative deficit results from an insufficient mass before injection, from poor engraftment, and/or from destruction by inflammation and immune reaction during the first year. There is evidence that β-cell loss is considerable during the first days posttransplantation (3), which has intensified attention to the process of revascularization in the islet cell implant (4,5).
In their normal pancreatic habitat, islet cells are embedded in a dense microvascular network that should meet the high oxygen and nutrient needs of the endocrine cells. The islet isolation process and the subsequent in vitro steps, however, disrupt capillary structures and their association with endocrine cells, making the formation of new vessels necessary for adequate graft survival. In rodent models, the islet blood flow appears restored within 1–2 weeks after transplantation (5); during the first posttransplant week, transplanted cells seem thus dependent on adequate diffusion of oxygen and nutrients, while mechanisms should be initiated to start sprouting of vessels into their intrahepatic sites. New-vessel formation can be induced and directed by factors released by the tissue to be irrigated (6). Islet β-cells are known to produce vascular endothelial growth factor (VEGF), and several studies have been reported on the beneficial effects of an increased VEGF expression or availability on survival of transplanted islets in rodents (7). However, it remains unclear to which extent VEGF release from normal β-cells succeeds in rapidly inducing an adequate blood supply to the implants. Moreover, such angiogenic action might be less pronounced in human islet cell preparations, since they contain much lower percentages of β-cells than isolated rodent islets (8). Our transplant protocols are conducted with cultured human islet cell grafts in which duct cells represent the main contaminant, often reaching percentages that approximate the percent of β-cells (2,8). To assess the possible role of duct cells in human islet cell implants, we have purified these cells from human donor organs and examined their properties in vitro as well as following transplantation in immune-deficient mice (9,10). It was thus found that human pancreatic duct cells react much more vigorously to cytokines than the islet endocrine cells when comparing cellular major histocompatibility complex II expression, induction of nitric oxide (NO) synthase followed by production of NO, and synthesis and subsequent release of the inflammatory cytokine tumor necrosis factor (TNF)-α (11–13). In addition, duct cells were shown to express CD40 that rapidly activated nuclear factor-κB upon ligation, lending further support to the view that duct cells may actively contribute to the inflammatory process leading to diabetes and/or to islet graft rejection (9,14). During these in vitro studies on human duct cells, we noticed that they could produce several cytokines, among which interleukin (IL)-8 (CXCL8) appeared more abundant. IL-8 is known as a potent inducer of angiogenesis (15,16), involving effects on chemotaxis, survival, and proliferation of tissue-specific endothelial cells through interaction with its cognate G protein–coupled receptors CXCR1 and CXCR2 (17,18). We therefore compared the production of IL-8 and VEGF by cultured human pancreatic duct cells and islet cell preparations and examined whether these duct cell products exhibit angiogenic properties in laboratory models.
RESEARCH DESIGN AND METHODS
Cell isolation and culture.
Human pancreatic duct cell and endocrine islet cell fractions were prepared from donor organs that were procured by transplant departments affiliated to Eurotransplant Foundation (Leiden, the Netherlands). The organs were sent to the Beta Cell Bank in Brussels for preparing β-cell grafts in an approved clinical trial (8); procedures for organ donation and for use of isolated fractions followed the guidelines of Eurotransplant and of the ethics committees of Brussels Free University and the Katholieke Universiteit Leuven. Endocrine islet cell fractions that did not fulfill the criteria for transplantation were only released for associated research projects when permission was granted; the latter was also needed for the duct cell fraction. The techniques for isolation and culture of both fractions have been previously described (8,13). Briefly, the pancreas was digested with collagenase and purified on a Ficoll gradient. The nonendocrine fraction was recovered and cultured for 3–7 days as suspension in serum-free HAM's F-10 medium (Life Technologies, Paisley, U.K.) supplemented with 6.5 mmol/l glucose (Merck, Darmstadt, Germany), 0.5% BSA (Cohn analog BSA; Sigma), 0.1 mg/ml streptomycin (Sigma Chemical, St. Louis, MO), 0.075 mg/ml penicillin (Continental Pharma, Brussels, Belgium), and 0.3 mg/ml l-glutamine (Sigma). At the end of this period, this fraction contained >90% CK19-positive duct cells, <10% endocrine cells, and <1% acinar cells (as indicated by amylase staining and electron microscopy). The endocrine islet cell fraction was cultured in the same medium (19); after 1 week of culture, it contained 50–80% cells that were positive for insulin or glucagon and 20–50% CK19-positive cells. For some experiments, fractions were enriched in either endocrine or duct cells; this procedure involved a trypsin dissociation of an islet fraction and subsequent flow cytometry (fluorescence-activated cell sorter [FACS]) for separation of cells with high (endocrine) or low (duct) cellular side scatter.
Conditioned medium preparation and cytokine measurement.
Duct cell–conditioned media were prepared for IL-8 and VEGF immunoassays and for the rat aortic ring assay. They were collected after 24-h suspension culture of the duct cell fraction at a density of 5 × 105 cells/ml in serum-free HAM's F10 (for enzyme-linked immunosorbent assay) or MCDB131 (for rat aortic ring assay; Life Technologies). Conditioned media were clarified by centrifugation and stored at −20°C. Cell samples were taken for total RNA isolation and quantitative real-time RT-PCR. A human-specific enzyme-linked immunosorbent assay (R&D systems, Minneapolis, MN) was used to measure IL-8 and VEGF concentrations at minimum detectable doses of 3.5 ng/ml and 3.3 pg/ml, respectively.
Assessment of angiogenic effects.
The angiogenic effect of cells or their conditioned media was assessed in two models. The in vitro aortic ring assay assesses the effect of solubilized agents on growth of endothelial cells and their assembly into vessel-like structures (20). The in vivo chick embryo chorioallantoic membrane (CAM) assay examines the effects of living cells on these processes (21); to this end, sponges are used as cell carrier and placed on the membrane, allowing the cellular secretory products to exert their effects; this model was not suitable for testing our conditioned media as too little volume can be inoculated.
Rat aortic ring assay.
The rat aortic ring assay was performed as previously described (20). In brief, aortas were removed from 6- to 10-week-old male Wistar rats (Janvier, Le Genest Saint-Isle, France), sectioned into 1-mm-long rings, and then embedded in gels of rat tail interstitial collagen (1.5 mg/ml, Collagen R; Serva, Heidelberg, Germany) that had polymerized in cylindrical agarose wells (type VII; Sigma-Aldrich, St. Louis, MO). Each gel was kept at 37°C in 60-mm diameter bacteriologic Petri dishes containing 6 ml control or conditioned medium. Positive controls consisted of culture medium with recombinant human IL-8 (1 ng/ml) or recombinant human VEGF-A165 (20 ng/ml); both proteins were purchased from Peprotech (Rocky Hill, NJ). Each condition was performed in triplicate; half of the medium volume was replaced every other day. After 10 days, photographs were taken under phase contrast (plan neofluar 5×/0.15; Axiovert 135 microscope equipped with Axiocam MRc camera) and analyzed with Axiovision 3.1 (Carl Zeiss, Jena, Germany) in order to count the number of microvessels and quantify their total length. This study was approved by the local ethics committee.
Chick CAM assay.
Fertilized white leghorn chicken eggs (Katholieke Universiteit Leuven animal house, Heverlee, Belgium) were incubated for 3 days at 37°C before removing 3 ml of albumen to detach the shell from the developing CAM. After making a window in the eggshell to expose the CAM, it was covered with cellophane. Eggs were then further incubated until day 9, when the test conditions were applied. Sterile absorbable gelatin sponges (1–2 mm3; Hospithera, Brussels, Belgium) were impregnated with the indicated pancreatic cell preparations and then placed on the CAM. PBS containing 0.1% BSA (1 mg/ml, ∼50 μg/embryo) was used as a negative control and recombinant human IL-8 (100 μg/ml, ∼5 μg/embryo) and recombinant human VEGF-A165 (100 μg/ml, ∼5 μg/embryo) as positive controls. The windows were again covered and the incubation continued until day 13, when angiogenesis was assessed. Membranes were fixed with 4% paraformaldehyde at room temperature for 2 h. A large area around the contact or insertion sites was removed, placed on a glass slide, and photographed under a Zeiss Axioplan 2 microscope (Carl Zeiss) with a Kontron Electronik (Eching bei München, Germany) image-analyzing system (KS400 version 3.00). To determine the number of blood vessels, a grid containing three concentric circles with diameters of 4, 5, and 6 mm was positioned on the surface of the CAM and all vessels radiating from the sample spot and intersecting the circles were blindly counted under a stereomicroscope. In some experiments, sponges with cells were preincubated with 500 ng of a neutralizing rabbit anti-human VEGF antibody (Chemicon International, Temecula, CA), a mouse anti-human IL-8 antibody, or a mouse anti-human CXCL1 antibody (R&D Systems) (30 min, 37°C, 5% CO2) before implantation on the CAM.
Transplantation.
Eight-week-old male normoglycemic nonobese diabetic (NOD)/scid mice were used as transplant recipients (Katholieke Universiteit Leuven breeding colony, Leuven, Belgium). Mice were kept under aseptic conditions and had free access to pellet food and water. Animals were anesthetized with intraperitoneal avertin (2,2,2-tribromoethanol, 500 mg/kg body wt; Sigma-Aldrich) and grafts implanted under the kidney capsule (22). Grafts consisted of a cultured human pancreatic duct cell fraction (74–88% purity with 4–16% endocrine cells) or a cultured human endocrine islet cell fraction (68–88% purity with 4–16% duct cells). Total cell number in each graft was 5 × 106. Animals were killed at 15 min or 2, 8, 24, or 144 h posttransplantation; graft-bearing kidneys were removed for mRNA analysis and/or serum collected for analysis. The same human-specific enzyme-linked immunosorbent assay kits (as described above) were used to measure human IL-8 and human VEGF concentrations in sera of graft recipients and sham controls. This study was approved by the local ethics committee.
RNA isolation and quantitative real-time RT-PCR.
Total RNA was isolated from cultured cell fractions and from implants using Trizol (Life Technologies). After treatment with Superscript II reverse transcriptase (Life Technologies), real-time RT-PCR was performed for selected genes (23,24). PCR were performed in the ABI Prism 7700 Sequence Detector (Perkin-Elmer/Applied Biosystems, Foster City, CA) with a Gene-Amp PCR system 9600 (Perkin-Elmer). PCR amplifications were performed in triplicate. Human cDNA was determined using the following sequences: for IL-8: forward 5′-TGGCAGCCTTCCTGATTTCT-3′, reverse 5′-TTAGCACTCCTTGGCAAAACTG-3′, and TaqMan Probe 5′-CAGCTCTGTGTGAAGGT-3′; for human VEGF: forward 5′-ACCAAGGCCAGCACATAGAGA-3′, reverse 5′-CTTGCGCTTTCGTTTTTGC-3′, and TaqMan Probe 5′-CAGCACAACAAATGTGAATGCAGAC CAA-3′. Primers and probes for the housekeeping gene human hypoxanthinephosphoribosyltransferase 1 (HPRT) were previously described (13). Data are expressed as relative input copy number normalized to HPRT.
Statistics.
Results are expressed as means ± SE of n independent experiments. Statistical analysis was performed using the SPSS computer program. One-way ANOVA with post hoc least significant difference was applied to compare means of three or more groups. Differences were considered significant when P ≤ 0.05.
RESULTS
Human pancreatic duct cell preparations release VEGF and IL-8 during culture.
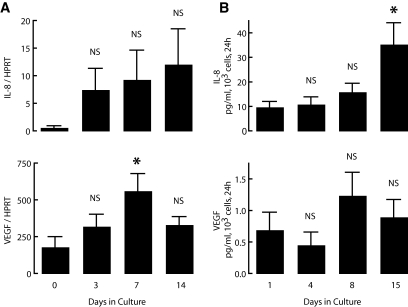
Cultured human pancreatic duct cell preparations were found to release IL-8 and VEGF from the day of isolation and to sustain this activity for at least 2 weeks; at day 15, the 24-h release of IL-8 was threefold higher than at start (Fig. 1). The cultured cells also exhibited a sustained mRNA expression for both proteins over this period, with average levels that tended to increase. When comparing individual donor cell preparations, we noticed a relatively large variability in absolute levels of protein expression and release, which we have not yet further examined in terms of possible underlying reasons at the level of the donor or of the composition of the isolate.
FIG. 1.
IL-8 and VEGF expression and release by human pancreatic duct cell preparations during culture. Human pancreatic duct cells were cultured for up to 15 days as described in research design and methods. Cell samples were taken for RNA isolation at indicated times. A: Expression levels of IL-8 and VEGF were measured by real-time PCR and normalized to HPRT. Medium was sampled at the indicated times and assayed for IL-8 and VEGF. B: Values are expressed as picograms per milliliter released by 103 cells over 24 h. Values are means of four independent experiments ± SE. P vs. start (day 0 or day 1): *P ≤ 0.05.
Duct cells induce VEGF- and IL-8–dependent neogenesis of blood vessels.
To assess the angiogenic effect of duct cell–released IL-8 and VEGF, we tested the angiogenic potential of duct cells as well as of duct cell–conditioned medium using recombinant human IL-8 and VEGF as control conditions.
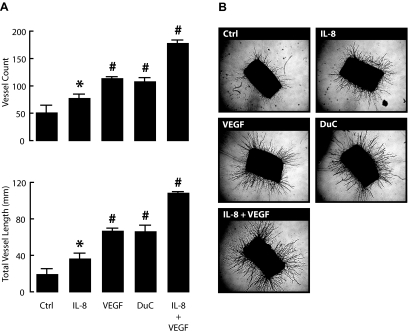
In the aortic ring assay, both recombinant human IL-8 and VEGF promote the outgrowth of vessel-like structures over a 10-day culture period (Fig. 2). In presence of IL-8 (1 ng/ml) or VEGF (20 ng/ml), the number, as well as the total length of vessels sprouting from the aortic rings, were significantly increased. Combination of both recombinant proteins resulted in a cumulative effect, leading to a threefold increase in the number of vessels and a fivefold increase in total vessel length. A stimulatory effect was also observed after addition of culture medium that had been conditioned for 24 h by duct cell aggregates; its amplitude corresponded to 61% of the effect (for both vessel count and total vessel length) measured for the combination of IL-8 at 1 ng/ml plus VEGF at 20 ng/ml and was comparable with that of VEGF alone at 20 ng/ml (Fig. 2).
FIG. 2.
Duct cells induce neogenesis of blood vessels in rat aortic ring assay. Isolated rat aortic rings were incubated for 10 days in control medium (Ctrl) or medium with recombinant human IL-8 (1 ng/ml), recombinant human VEGF-A165 (20 ng/ml), a combination of both, or with 24-h duct cell–conditioned medium (DuC). At day 10, rings were photographed. Total vessel numbers were counted and total vessel lengths were measured as described in research design and methods. A: Values are the average of three aortic rings per condition ± SE. P value vs. carrier alone (Ctrl); *P ≤ 0.05; #P ≤ 0.001. Results come from at least three independent experiments. Representative photographs of each condition are shown in B.
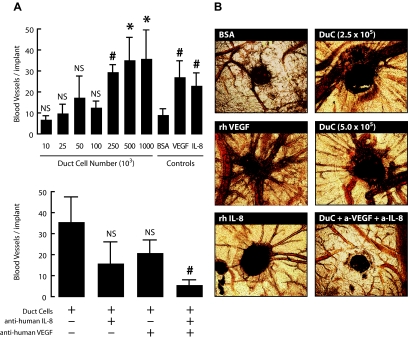
In the chicken embryonic CAM assay, inoculation of 5 μg recombinant human VEGF or 5 μg human IL-8 markedly increased the number of allantoic vessels directed toward the implant (Fig. 3). After 13 days, their number was 2.5- to 3-fold higher than for sponges containing 50 μg BSA. A similar increase was inducible with sponges containing 2.5 × 105 human pancreatic duct cells (Fig. 3). This duct cell effect was slightly stronger with 1 ×106 cells, inducing four- and fivefold more vessels than in the BSA control or in eggs with sponges containing 0.1 × 105 cells, respectively. Duct cell–induced allantoic vessel formation was reduced by 55 and 41%, respectively, in presence of an antibody against human VEGF or against human IL-8 but not by an antibody against human CXCL1 (95% of control, n = 3, data not shown); when expressed in absolute number of vessels these reductions did not reach statistical significance probably as a result of the large variation in the amplitude of IL-8 and VEGF release (section 1) and the related duct cell effects (Fig. 3). However, the combination of both antibodies completely suppressed duct cell–induced vessel formation (Fig. 3).
FIG. 3.
Duct cells induce neogenesis of blood vessels in the CAM assay. Duct cells were grafted on CAMs at indicated concentrations with or without anti-human IL-8 and anti-human VEGF neutralizing antibodies. BSA (50 μg/embryo) was used as negative control and recombinant human IL-8 (5 μg/embryo) and VEGF (5 μg/embryo) as positive controls. At day 13, the region around the implant was photographed. Values are the average of three implants per condition ± SE (of five independent experiments). P vs. negative control (50 μg/embryo; 0.1% BSA) (A, upper panel); P vs. 2.5 × 105 duct cell condition (A, lower panel). *P ≤ 0.05; #P ≤ 0.001. Representative photographs are shown in B. (Please see http://dx.doi.org/10.2337/db07-1705 for a high-quality digital representation of this figure.)
Contribution of duct cells to IL-8 and VEGF expression and to angiogenic effect in isolated human islet preparations.
When human duct cell fractions and islet cell–enriched fractions isolated from the same donors were compared for their IL-8 and VEGF expression, mRNA levels of both factors were consistently higher in duct cell preparations (Table 1). While each of the duct cell fractions exhibited a high purity in CK19-positive cells (>90%), their individual IL-8 and VEGF expression level varied markedly, with 20- to 60-fold higher values in one of the preparations. The corresponding islet-enriched fractions exhibited a similar variability in IL-8 and VEGF expression (Table 1). Since human islet-enriched fractions contain high percentages of nonendocrine cells that are CK19-positive, we examined whether this expression and its high variability was mainly located in the nonendocrine cell population. Islet-enriched fractions were dissociated and fluorescence-activated cell sorted in a population of nonendocrine cells (77–97% nongranulated cells in electron microscopy and 2–23% endocrine phenotype) and a population of larger endocrine cells (>91% cells with endocrine granules in electron microscopy and <9% nongranulated cells). Quantitative PCR analysis of IL-8 expression indicated that the nonendocrine cell population exhibits a considerably higher IL-8 expression than the endocrine cell population, and that it is also the cause for the high variability in this expression (from 3.2 to 6.3 versus a range from 0.1 to 0.8 in the endocrine cell population) (Table 1).
TABLE 1.
Correlation between presence of duct cells and expression of IL-8 and VEGF in human pancreatic cell preparations
| Donor pancreatic fraction | Cellular composition (%)
|
mRNA expression
|
||
|---|---|---|---|---|
| Endocrine cells | Duct cells | IL-8/HPRT | VEGF/HPRT | |
| No. 1 | ||||
| Duct cell enriched | 7 | 93 | 1.3 | 47.4 |
| Islet cell enriched | 79 | 21 | 0.1 | 9.9 |
| No. 2 | ||||
| Duct cell enriched | <1 | 98 | 0.6 | 25.4 |
| Islet cell enriched | 59 | 41 | 0.4 | 8.1 |
| No. 3 | ||||
| Duct cell enriched | <1 | 99 | 40.6 | 456.5 |
| Islet cell enriched | 50 | 50 | 11.0 | 319.1 |
| No. 4 | ||||
| Duct cell enriched | 2 | 97 | 3.2 | 36.2 |
| Islet cell enriched | 97 | 3 | 0.1 | 17.5 |
| No. 5 | ||||
| Duct cell enriched | 23 | 77 | 6.3 | 33.3 |
| Islet cell enriched | 96 | 4 | 0.7 | 8.8 |
| No. 6 | ||||
| Duct cell enriched | 5 | 95 | 4.6 | 14.4 |
| Islet cell enriched | 91 | 9 | 0.8 | 2.7 |
Pancreatic fractions enriched in duct cells or islet cells from the same donor were compared for their IL-8 and VEGF expression. Cells were retrieved from culture, total RNA extracted, and reverse transcribed to cDNA. IL-8 and VEGF expression levels were measured by real-time PCR and normalized to HPRT (donor numbers 1–3). Islet-enriched preparations were dissociated into single cells and fluorescence-activated cell sorted into duct cell–purified and endocrine cell–purified preparations prior to comparing their IL-8 and VEGF expression (donor numbers 4–6).
These observations led us to compare the angiogenic effect of FACS-purified duct and islet endocrine cells. For this experiment, we dissociated and fluorescence-activated cell-sorted transplant-grade islet preparations to obtain a duct cell–purified population with >90% CK19-positive cells and <5% endocrine cells and an endocrine cell–purified population with >80% endocrine islet cells and <20% CK19-positive cells. When tested at the same cell concentrations (1, 2.5, and 5 × 105 cells) in the chicken embryonic CAM assay, the duct cell fraction induced a significantly higher number of blood vessels than the endocrine fraction (Table 2).
TABLE 2.
Comparison of angiogenic effects of duct cell–purified and endocrine cell–purified fractions in the CAM assay
| Inoculate | Number of blood vessels per implant | P |
|---|---|---|
| Duct cell purified | ||
| 106 cells | ||
| 0.10 | 37.0 ± 0.0 | NS |
| 0.25 | 42.5 ± 3.5 | ≤0.05 |
| 0.50 | 41.0 ± 1.4 | ≤0.01 |
| Endocrine cell purified | ||
| 106 cells | ||
| 0.10 | 14.0 ± 15.6 | |
| 0.25 | 18.0 ± 8.7 | |
| 0.50 | 24.0 ± 1.4 | |
| Concentration (μg/ml) | ||
| BSA | ||
| 1,000 | 9.3 ± 2.7 | |
| Recombinant human VEGF | ||
| 100 | 27.3 ± 7.6 | ≤0.001* |
| Recombinant human IL-8 | ||
| 100 | 23.2 ± 5.9 | ≤0.001* |
Data are the average of three implants per condition ± SE (of two to five independent experiments). Islet preparations were dissociated to single cells and fluorescence-activated cell sorted into duct cell–purified and endocrine cell–purified preparations before inoculation on CAMs at the indicated cell numbers. At day 13, the region around the implant was photographed and the number of vessels was counted. P vs. endocrine cell–purified preparation.
P vs. negative control (1,000 μg/ml; 0.1% BSA).
Human duct cell implants express and release IL-8 and VEGF.
Since human islet cell grafts can contain a high percentages of duct cells, it is conceivable that this “contaminant” influences revascularization through its expression and release of angiogenic factors like IL-8 and VEGF. Thus, we measured expression and release of both compounds in human duct cell grafts (5 × 106 cells per graft) that had been implanted under the renal capsule of NOD/scid mice.
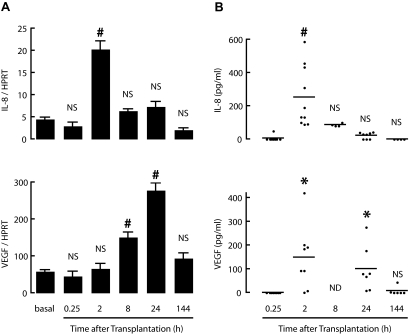
Implants were removed at different time points and analyzed for their human IL-8 and VEGF expression by quantitative real-time RT-PCR. Expression of both genes increased during the first day posttransplantation. IL-8 was 7- and 2.5-fold higher after 2 and 24 h, respectively, and VEGF progressively increased to fivefold higher levels (Fig. 4). Six days after transplantation, the IL-8 mRNA level was back to its start level, whereas the VEGF mRNA level was still 61% higher, although not significantly different from basal control (Fig. 4).
FIG. 4.
Human duct cell grafts express IL-8 and VEGF after transplantation. Human duct cells were transplanted under the kidney capsule of NOD/scid mice. A: At indicated times, grafts were explanted and total RNA extracted and reverse transcribed to cDNA. IL-8 and VEGF gene expression levels were assessed by real-time PCR and normalized to HPRT. Values are means of four animals per time point. Figure represents three independent experiments. P vs. basal condition: *P ≤ 0.05; #P ≤ 0.001. At indicated time points, blood was taken from transplanted animals and serum assayed for human IL-8 and VEGF. Each dot represents an individual serum concentration value. Horizontal lines represent median. B: P vs. initial time point: *P ≤ 0.05; #P ≤ 0.001.
Serum of recipients of duct cell grafts was assayed for human IL-8 and VEGF. None of the proteins were detected in sham-operated animals (data not shown) or at 15 min posttransplantation. At 120 min, all recipients presented markedly elevated human IL-8 levels (100–600 pg/ml), but these decreased thereafter (Fig. 4). In six of eight recipients, human VEGF was also clearly detected in the serum, at levels ranging from 100 to 400 pg/ml, and remained present in the 24-h samples (Fig. 4). The failure to detect human IL-8 and VEGF at 15 min posttransplantation might reflect in part the delay of diffusion from the fresh nonvascularized implant to the blood. For IL-8, upregulation of mRNA during the first posttransplant hours seems also related to the appearance of the protein in the circulation.
The IL-8 and VEGF expression in transplanted duct cells was higher than that in islet cell–enriched implants prepared from the same donors, which is consistent with the differences observed in the isolated preparations (Fig. 5).
FIG. 5.
Contribution of duct cells to IL-8 and VEGF expression in transplanted human islet grafts. Endocrine-enriched (21% duct cells, 79% endocrine cells) and duct cell–enriched (93% duct cells, 7% endocrine cells) fractions from the same donor were transplanted under the renal capsule of NOD/scid mice. Grafts were removed at indicated times and total RNA extracted and reverse transcribed into cDNA. IL-8 and VEGF expression was measured by real-time PCR. For basal condition expression levels, a cell sample of each preparation was taken before transplantation. □, endocrine-enriched fraction; ▪, duct cell–enriched fraction. Figure represents three independent experiments; each data point is the mean ± SE of at least three animals. P vs. matched islet graft: §P ≤ 0.01; #P ≤ 0.001.
DISCUSSION
The present study demonstrates that human pancreatic duct cells can produce IL-8 and VEGF, two compounds that are known for their angiogenic activity (15,25). Both proteins are released at biologically relevant levels during culture of duct cell–enriched fractions that are prepared from human donor organs. To assess the angiogenic effect of duct cell–released IL-8 and VEGF, we tested duct cells and duct cell–conditioned medium in the aortic ring assay and in the chick embryo CAM assay. In both models, addition of IL-8 or VEGF induces the formation of microvessels with an increased potency when two factors were combined. Duct cells and their medium were found to induce similar effects and these appeared mediated by IL-8 and VEGF, since they were suppressed by neutralizing antibodies to these cytokines.
The IL-8 and VEGF release by isolated human duct cells is not reactive to the preparative procedure but is constitutive, as shown by the persistent expression and release of both proteins during culture. The two cytokines remained also expressed following transplantation of a cultured duct cell fraction under the kidney capsule of immune-deficient NOD/scid mice. They were even detected in the circulation of these animals, be it only during the first day posttransplantation. This short episode of elevated circulating levels does not exclude that both proteins remain produced and locally released at later times but indicates that the immediate posttransplant period favors their release, possibly as an effect of the hypoxic and/or inflammatory environment, which is known to induce IL-8 and VEGF production (26,27). When comparing data from independent experiments, we observed a high variation in the mRNA expression level of both proteins. This might reflect interindividual variations as seen in vitro (present study) and in other studies. VEGF production appears for example related to polymorphism in its gene promoter (28) and IL-8, usually barely detectable in normal tissue, was found to increase 10- to 100-fold under stress or proinflammatory cytokines (29). The present preparations were also obtained from donors of different age and sex, following various disease histories and organ preservation conditions. Finally, although the duct cell preparations appeared quite pure in light of their CK19-expressing cells, they might still be composed of different subpopulations with consequences on the rates of IL-8 and VEGF production.
IL-8 belongs to the family of CXC chemokines that are inducible by inflammatory mediators such as IL-1β, TNF-α, and bacterial products (30,31) and that have been implicated in early phases of tissue repair (32). Besides its angiogenic effect, IL-8 was found to recruit and activate monocytes to vascular lesions (33). Tissue infiltration by lymphocytes and macrophages is known to mediate local expression of VEGF, which promotes endothelial cell proliferation, migration, and microvascular networking (34). These processes were not investigated in the transplanted mice; the revascularization process is difficult to quantify and it is also uncertain whether immune-deficient recipients represent an adequate model for studying inflammatory and repair reactions.
The finding that human pancreatic duct cells can produce and release IL-8 and VEGF at levels that can stimulate angiogenesis might be relevant in human islet cell transplantation. In our experience, cultured human islet cell grafts contain 13–40% nongranulated CK19-positive cells that correspond to duct cells (8). This contaminating fraction is the main source of the IL-8 and VEGF production by the islet-enriched fraction. When compared with the expression levels in purified human endocrine islet cells, purified human duct cells generate 6- to 28-fold more IL-8 and 2- to 5-fold more VEGF. This difference was also reflected following transplantation in mice.
It is generally accepted that local VEGF production is important for the revascularization and survival of implanted β-cells (7). So far, β-cells have been considered as the driver of such process (35), but the evidence is still incomplete. Rat islet grafts were indeed shown to express increased VEGF levels during the first days following transplantation (36), which can be explained by a hypoxia-induced transcriptional activation and mRNA stabilization in the β-cells (37). That β-cell release of VEGF may promote angiogenesis was demonstrated by the improved vascularization and function of rodent islet grafts in which an increased VEGF expression was under control of the insulin promoter or had been induced by gene transfer (7,38). Surprisingly, blockade of VEGF signaling by an antagonist of VEGF-KDR/flk-1 did not impair the revascularization of transplanted islets (39), which was not sufficient to disregard a VEGF-regulated process but suggestive for further investigating the mechanisms underlying the revascularization of islet cell grafts. In fact, mouse islet implants under the kidney capsule or in the liver or spleen exhibit a much lower vascular density than pancreatic islets (40); they are, however, surrounded by a well-vascularized connective tissue that may provide a richer source of proangiogenic compounds. The present study shows that human pancreatic duct cells are a more potent source of VEGF than β-cells and that this is also the case for IL-8, another angiogenic factor. Whether, and if so, to which extent, the environment of contaminating duct cells can help the revascularization of a human β-cell implant is not yet known. It is likely that the microenvironment of the implant will not only regulate the production of angiogenic factors such as IL-8 and VEGF but also that of as-yet-unidentified angiostatins and of other inflammatory substances that can influence the survival of the β-cells. Previous work has shown that human duct cells can produce other inflammatory products like NO and TNF-α (12,13) and participate in the coagulation process (41). Angiogenic effects at the site of implantation can also be influenced by immune suppressive agents. Since rapamycin has been shown to suppress IL-8 expression (42) and to block VEGF effects (43), it needs to be assessed for possible negative influences on the revascularization of human islet grafts; its antiangiogenic effect might even be involved in its inhibitory action on β-cell regeneration (44), which is known to depend on interactions with vascular components (45). The currently used in vitro and in vivo models provide the tools to investigate these questions. Our observations demonstrate that duct cells should not be considered as passive bystanders in this process and further support their earlier proposed role in islet transplantation (44).
Acknowledgments
This study was supported by the Juvenile Diabetes Research Foundation (Center Grant 4-2005-1327); the European Commission (Sixth FP Grant no. LSHB-CT-2005-512145); the Research Program of the Research Foundation-Flanders (FWO) (G.0233.04 and G.0552.06); the Interuniversity Attraction Poles Program, Belgian State, Belgian Science Policy (IAP P5/17 and P6/40); and the Katholieke Universiteit Leuven (GOA 2004/10). B.M. has been and D.J. is a PhD fellow of FWO. C.G. is a postdoctoral fellow and C.M. is a senior clinical investigator of FWO, Belgium. B.M. and C.G. are partners of the Juvenile Diabetes Research Foundation Center for Beta Cell Therapy in Diabetes.
The authors thank Krista Suenens, Johan Guns, Natascha Caluwé, Geert Stangé, and Dirk Valckx for their excellent assistance; Dr. Karin Vanderkerken for valuable advice on rat aortic ring assay; and the Eurotransplant Foundation and the Beta Cell Bank for providing human pancreatic cell preparations.
Published ahead of print at http://diabetes.diabetesjournals.org on 20 May 2008.
B.M. and C.G. contributed equally to this article.
The costs of publication of this article were defrayed in part by the payment of page charges. This article must therefore be hereby marked “advertisement” in accordance with 18 U.S.C. Section 1734 solely to indicate this fact.
REFERENCES
- 1.Ryan EA, Paty BW, Senior PA, Bigam D, Alfadhli E, Kneteman NM, Lakey JR, Shapiro AM: Five-year follow-up after clinical islet transplantation. Diabetes 54: 2060–2069, 2005 [DOI] [PubMed] [Google Scholar]
- 2.Keymeulen B, Gillard P, Mathieu C, Movahedi B, Maleux G, Delvaux G, Ysebaert D, Roep B, Vandemeulebroucke E, Marichal M, In 't Veld P, Bogdani M, Hendrieckx C, Gorus F, Ling Z, van Rood J, Pipeleers D: Correlation between beta cell mass and glycemic control in type 1 diabetic recipients of islet cell graft. Proc Natl Acad Sci U S A 103: 17444–17449, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Davalli AM, Scaglia L, Zangen DH, Hollister J, Bonner-Weir S, Weir GC: Vulnerability of islets in the immediate posttransplantation period: dynamic changes in structure and function. Diabetes 45: 1161–1167, 1996 [DOI] [PubMed] [Google Scholar]
- 4.Andersson A, Korsgren O, Jansson L: Intraportally transplanted pancreatic islets revascularized from hepatic arterial system. Diabetes 38 (Suppl. 1): 192–195, 1989 [DOI] [PubMed] [Google Scholar]
- 5.Jansson L, Carlsson PO: Graft vascular function after transplantation of pancreatic islets. Diabetologia 45: 749–763, 2002 [DOI] [PubMed] [Google Scholar]
- 6.Brissova M, Shostak A, Shiota M, Wiebe PO, Poffenberger G, Kantz J, Chen Z, Carr C, Jerome WG, Chen J, Baldwin HS, Nicholson W, Bader DM, Jetton T, Gannon M, Powers AC: Pancreatic islet production of vascular endothelial growth factor-α is essential for islet vascularization, revascularization, and function. Diabetes 55: 2974–2985, 2006 [DOI] [PubMed] [Google Scholar]
- 7.Zhang N, Richter A, Suriawinata J, Harbaran S, Altomonte J, Cong L, Zhang H, Song K, Meseck M, Bromberg J, Dong H: Elevated vascular endothelial growth factor production in islets improves islet graft vascularization. Diabetes 53: 963–970, 2004 [DOI] [PubMed] [Google Scholar]
- 8.Keymeulen B, Ling Z, Gorus FK, Delvaux G, Bouwens L, Grupping A, Hendrieckx C, Pipeleers-Marichal M, Van Schravendijk C, Salmela K, Pipeleers DG: Implantation of standardized beta-cell grafts in a liver segment of IDDM patients: graft and recipients characteristics in two cases of insulin-independence under maintenance immunosuppression for prior kidney graft. Diabetologia 41: 452–459, 1998 [DOI] [PubMed] [Google Scholar]
- 9.Pipeleers D, Hoorens A, Marichal-Pipeleers M, Van de Casteele M, Bouwens L, Ling Z: Role of pancreatic β-cells in the process of β-cell death. Diabetes 50 (Suppl. 1): S52–S57, 2001 [DOI] [PubMed] [Google Scholar]
- 10.Bogdani M, Lefebvre V, Buelens N, Bock T, Pipeleers-Marichal M, In't Veld P, Pipeleers D: Formation of insulin-positive cells in implants of human pancreatic duct cell preparations from young donors. Diabetologia 46: 830–838, 2003 [DOI] [PubMed] [Google Scholar]
- 11.Pavlovic D, van de Winkel M, van der Auwera B, Chen MC, Schuit F, Bouwens L, Pipeleers D: Effect of interferon-gamma and glucose on major histocompatibility complex class I and class II expression by pancreatic beta- and non-beta-cells. J Clin Endocrinol Metab 82: 2329–2336, 1997 [DOI] [PubMed] [Google Scholar]
- 12.Pavlovic D, Chen MC, Bouwens L, Eizirik DL, Pipeleers D: Contribution of ductal cells to cytokine responses by human pancreatic islets. Diabetes 48: 29–33, 1999 [DOI] [PubMed] [Google Scholar]
- 13.Movahedi B, Van de Casteele M, Caluwe N, Stange G, Breckpot K, Thielemans K, Vreugdenhil G, Mathieu C, Pipeleers D: Human pancreatic duct cells can produce tumour necrosis factor-alpha that damages neighbouring beta cells and activates dendritic cells. Diabetologia 47: 998–1008, 2004 [DOI] [PubMed] [Google Scholar]
- 14.Vosters O, Beuneu C, Nagy N, Movahedi B, Aksoy E, Salmon I, Pipeleers D, Goldman M, Verhasselt V: CD40 expression on human pancreatic duct cells: role in nuclear factor-kappa B activation and production of pro-inflammatory cytokines. Diabetologia 47: 660–668, 2004 [DOI] [PubMed] [Google Scholar]
- 15.Koch AE, Polverini PJ, Kunkel SL, Harlow LA, DiPietro LA, Elner VM, Elner SG, Strieter RM: Interleukin-8 as a macrophage-derived mediator of angiogenesis. Science 258: 1798–1801, 1992 [DOI] [PubMed] [Google Scholar]
- 16.Strieter RM, Burdick MD, Gomperts BN, Belperio JA, Keane MP: CXC chemokines in angiogenesis. Cytokine Growth Factor Rev 16: 593–609, 2005 [DOI] [PubMed] [Google Scholar]
- 17.Heidemann J, Ogawa H, Dwinell MB, Rafiee P, Maaser C, Gockel HR, Otterson MF, Ota DM, Lugering N, Domschke W, Binion DG: Angiogenic effects of interleukin 8 (CXCL8) in human intestinal microvascular endothelial cells are mediated by CXCR2. J Biol Chem 278: 8508–8515, 2003 [DOI] [PubMed] [Google Scholar]
- 18.Li A, Dubey S, Varney ML, Dave BJ, Singh RK: IL-8 directly enhanced endothelial cell survival, proliferation, and matrix metalloproteinases production and regulated angiogenesis. J Immunol 170: 3369–3376, 2003 [DOI] [PubMed] [Google Scholar]
- 19.Ling Z, Pipeleers DG: Prolonged exposure of human beta cells to elevated glucose levels results in sustained cellular activation leading to a loss of glucose regulation. J Clin Invest 98: 2805–2812, 1996 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Van Valckenborgh E, De Raeve H, Devy L, Blacher S, Munaut C, Noel A, Van Marck E, Van Riet I, Van Camp B, Vanderkerken K: Murine 5T multiple myeloma cells induce angiogenesis in vitro and in vivo. Br J Cancer 86: 796–802, 2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pliskin ME, Ginsberg SM, Carp N: Induction of neovascularization by mitogen-activated spleen cells and their supernatants. Transplantation 29: 255–258, 1980 [DOI] [PubMed] [Google Scholar]
- 22.Gysemans CA, Waer M, Valckx D, Laureys JM, Mihkalsky D, Bouillon R, Mathieu C: Early graft failure of xenogeneic islets in NOD mice is accompanied by high levels of interleukin-1 and low levels of transforming growth factor-β mRNA in the grafts. Diabetes 49: 1992–1997, 2000 [DOI] [PubMed] [Google Scholar]
- 23.Overbergh L, Giulietti A, Valckx D, Decallonne R, Bouillon R, Mathieu C: The use of real-time reverse transcriptase PCR for the quantification of cytokine gene expression. J Biomol Tech 14: 33–43, 2003 [PMC free article] [PubMed] [Google Scholar]
- 24.Giulietti A, Overbergh L, Valckx D, Decallonne B, Bouillon R, Mathieu C: An overview of real-time quantitative PCR: applications to quantify cytokine gene expression. Methods 25: 386–401, 2001 [DOI] [PubMed] [Google Scholar]
- 25.Leung DW, Cachianes G, Kuang WJ, Goeddel DV, Ferrara N: Vascular endothelial growth factor is a secreted angiogenic mitogen. Science 246: 1306–1309, 1989 [DOI] [PubMed] [Google Scholar]
- 26.Kukielka GL, Smith CW, LaRosa GJ, Manning AM, Mendoza LH, Daly TJ, Hughes BJ, Youker KA, Hawkins HK, Michael LH, et al.: Interleukin-8 gene induction in the myocardium after ischemia and reperfusion in vivo. J Clin Invest 95: 89–103, 1995 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Fava RA, Olsen NJ, Spencer-Green G, Yeo KT, Yeo TK, Berse B, Jackman RW, Senger DR, Dvorak HF, Brown LF: Vascular permeability factor/endothelial growth factor (VPF/VEGF): accumulation and expression in human synovial fluids and rheumatoid synovial tissue. J Exp Med 180: 341–346, 1994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Watson CJ, Webb NJ, Bottomley MJ, Brenchley PE: Identification of polymorphisms within the vascular endothelial growth factor (VEGF) gene: correlation with variation in VEGF protein production. Cytokine 12: 1232–1235, 2000 [DOI] [PubMed] [Google Scholar]
- 29.Hoffmann E, Dittrich-Breiholz O, Holtmann H, Kracht M: Multiple control of interleukin-8 gene expression. J Leukoc Biol 72: 847–855, 2002 [PubMed] [Google Scholar]
- 30.Matsushima K, Morishita K, Yoshimura T, Lavu S, Kobayashi Y, Lew W, Appella E, Kung HF, Leonard EJ, Oppenheim JJ: Molecular cloning of a human monocyte-derived neutrophil chemotactic factor (MDNCF) and the induction of MDNCF mRNA by interleukin 1 and tumor necrosis factor. J Exp Med 167: 1883–1893, 1988 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yoshimura T, Matsushima K, Tanaka S, Robinson EA, Appella E, Oppenheim JJ, Leonard EJ: Purification of a human monocyte-derived neutrophil chemotactic factor that has peptide sequence similarity to other host defense cytokines. Proc Natl Acad Sci U S A 84: 9233–9237, 1987 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rennekampff HO, Hansbrough JF, Kiessig V, Dore C, Sticherling M, Schroder JM: Bioactive interleukin-8 is expressed in wounds and enhances wound healing. J Surg Res 93: 41–54, 2000 [DOI] [PubMed] [Google Scholar]
- 33.Gerszten RE, Garcia-Zepeda EA, Lim YC, Yoshida M, Ding HA, Gimbrone MA, Jr, Luster AD, Luscinskas FW, Rosenzweig A: MCP-1 and IL-8 trigger firm adhesion of monocytes to vascular endothelium under flow conditions. Nature 398: 718–723, 1999 [DOI] [PubMed] [Google Scholar]
- 34.Pober JS, Sessa WC: Evolving functions of endothelial cells in inflammation. Nat Rev Immunol 7: 803–815, 2007 [DOI] [PubMed] [Google Scholar]
- 35.Kuroda M, Oka T, Oka Y, Yamochi T, Ohtsubo K, Mori S, Watanabe T, Machinami R, Ohnishi S: Colocalization of vascular endothelial growth factor (vascular permeability factor) and insulin in pancreatic islet cells. J Clin Endocrinol Metab 80: 3196–3200, 1995 [DOI] [PubMed] [Google Scholar]
- 36.Vasir B, Jonas JC, Steil GM, Hollister-Lock J, Hasenkamp W, Sharma A, Bonner-Weir S, Weir GC: Gene expression of VEGF and its receptors Flk-1/KDR and Flt-1 in cultured and transplanted rat islets. Transplantation 71: 924–935, 2001 [DOI] [PubMed] [Google Scholar]
- 37.Vasir B, Aiello LP, Yoon KH, Quickel RR, Bonner-Weir S, Weir GC: Hypoxia induces vascular endothelial growth factor gene and protein expression in cultured rat islet cells. Diabetes 47: 1894–1903, 1998 [DOI] [PubMed] [Google Scholar]
- 38.Lai Y, Schneider D, Kidszun A, Hauck-Schmalenberger I, Breier G, Brandhorst D, Brandhorst H, Iken M, Brendel MD, Bretzel RG, Linn T: Vascular endothelial growth factor increases functional beta-cell mass by improvement of angiogenesis of isolated human and murine pancreatic islets. Transplantation 79: 1530–1536, 2005 [DOI] [PubMed] [Google Scholar]
- 39.Schramm R, Yamauchi J, Vollmar B, Vajkoczy P, Menger MD: Blockade of in vivo VEGF-KDR/flk-1 signaling does not affect revascularization of freely transplanted pancreatic islets. Transplantation 75: 239–242, 2003 [DOI] [PubMed] [Google Scholar]
- 40.Mattsson G, Jansson L, Carlsson PO: Decreased vascular density in mouse pancreatic islets after transplantation. Diabetes 51: 1362–1366, 2002 [DOI] [PubMed] [Google Scholar]
- 41.Beuneu C, Vosters O, Movahedi B, Remmelink M, Salmon I, Pipeleers D, Pradier O, Goldman M, Verhasselt V: Human pancreatic duct cells exert tissue factor–dependent procoagulant activity: relevance to islet transplantation. Diabetes 53: 1407–1411, 2004 [DOI] [PubMed] [Google Scholar]
- 42.Schmidbauer G, Hancock WW, Wasowska B, Badger AM, Kupiec-Weglinski JW: Abrogation by rapamycin of accelerated rejection in sensitized rats by inhibition of alloantibody responses and selective suppression of intragraft mononuclear and endothelial cell activation, cytokine production, and cell adhesion. Transplantation 57: 933–941, 1994 [DOI] [PubMed] [Google Scholar]
- 43.Guba M, von Breitenbuch P, Steinbauer M, Koehl G, Flegel S, Hornung M, Bruns CJ, Zuelke C, Farkas S, Anthuber M, Jauch KW, Geissler EK: Rapamycin inhibits primary and metastatic tumor growth by antiangiogenesis: involvement of vascular endothelial growth factor. Nat Med 8: 128–135, 2002 [DOI] [PubMed] [Google Scholar]
- 44.Pipeleers D, Keymeulen B, Chatenoud L, Hendrieckx C, Ling Z, Mathieu C, Roep B, Ysebaert D: A view on beta cell transplantation in diabetes. Ann N Y Acad Sci 958: 69–76, 2002 [DOI] [PubMed] [Google Scholar]
- 45.Nikolova G, Jabs N, Konstantinova I, Domogatskaya A, Tryggvason K, Sorokin L, Fassler R, Gu G, Gerber HP, Ferrara N, Melton DA, Lammert E: The vascular basement membrane: a niche for insulin gene expression and Beta cell proliferation. Dev Cell 10: 397–405, 2006 [DOI] [PubMed] [Google Scholar]