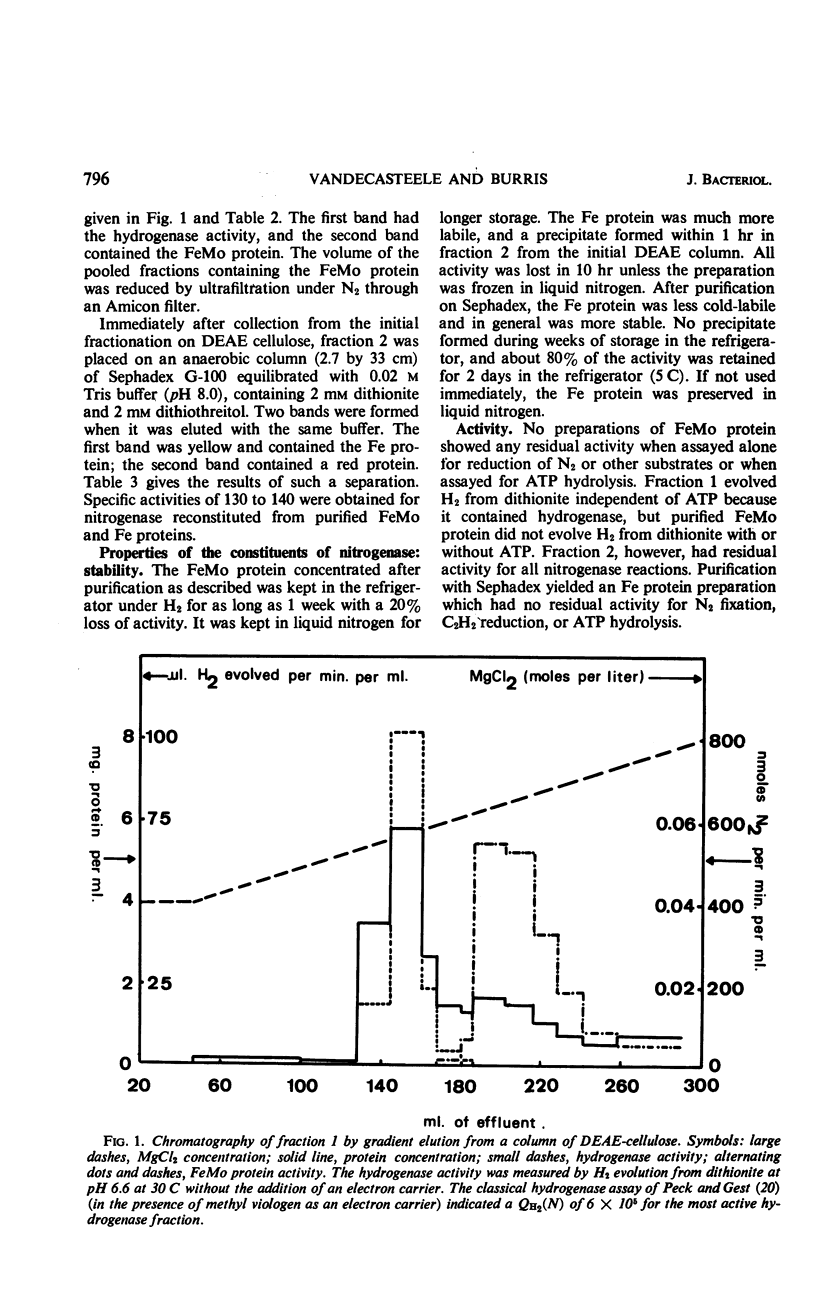
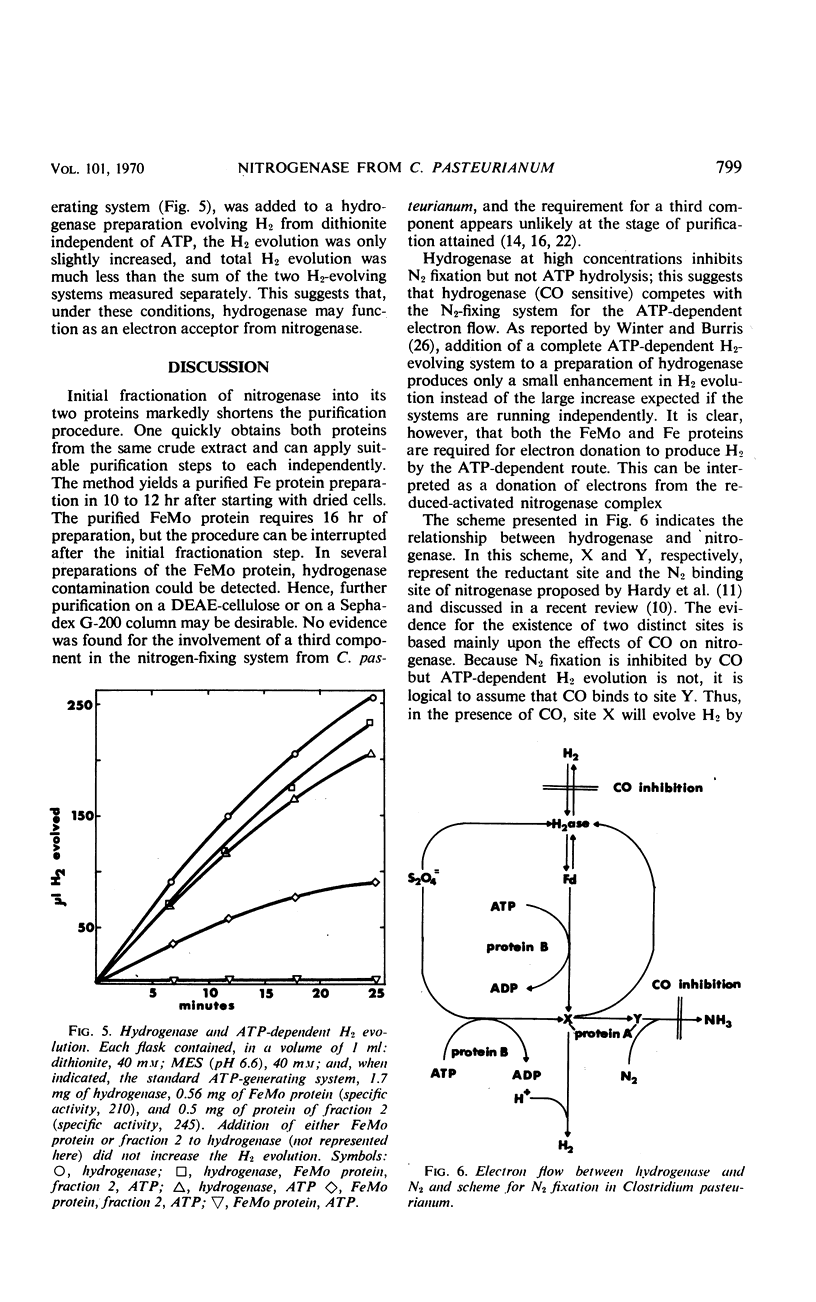
Abstract
A new procedure for a rapid and extensive purification of the FeMo protein and the Fe protein of the nitrogenase complex from Clostridium pasteurianum is described. Specific activities of 345 and 460 nmoles of N2 reduced per mg of protein per min for the FeMo protein and for the Fe protein, respectively, have been obtained. Preparations of the FeMo protein contained 0.96 atom of molybdenum and 15 atoms of iron per molecule, whereas those of the Fe protein contained 2.86 atoms of iron per molecule. Experiments suggest that a definite association of two Fe proteins and one FeMo protein is functional in the active enzyme complex. No individual role could be ascribed to either of the two proteins, but the fact that hydrogenase inhibits N2 fixation but not the reductant-dependent adenosine triphosphate hydrolysis supports the idea that there are two distinct sites on nitrogenase, one concerned with N2 activation and the other with activated electron transport.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bui P. T., Mortenson L. E. Mechanism of the enzymic reduction of N2: the binding of adenosine 5'-triphosphate and cyanide to the N2-reducing system. Proc Natl Acad Sci U S A. 1968 Nov;61(3):1021–1027. doi: 10.1073/pnas.61.3.1021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bulen W. A., LeComte J. R. The nitrogenase system from Azotobacter: two-enzyme requirement for N2 reduction, ATP-dependent H2 evolution, and ATP hydrolysis. Proc Natl Acad Sci U S A. 1966 Sep;56(3):979–986. doi: 10.1073/pnas.56.3.979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CARNAHAN J. E., MORTENSON L. E., MOWER H. F., CASTLE J. E. Nitrogen fixation in cell-free extracts of Clostridium pasteurianum. Biochim Biophys Acta. 1960 Nov 18;44:520–535. doi: 10.1016/0006-3002(60)91606-1. [DOI] [PubMed] [Google Scholar]
- DAVIS B. J. DISC ELECTROPHORESIS. II. METHOD AND APPLICATION TO HUMAN SERUM PROTEINS. Ann N Y Acad Sci. 1964 Dec 28;121:404–427. doi: 10.1111/j.1749-6632.1964.tb14213.x. [DOI] [PubMed] [Google Scholar]
- Dilworth M. J., Subramanian D., Munson T. O., Burris R. H. The adenosine triphosphate requirement for nitrogen fixation in cell-free extracts of Clostridium pasteurianum. Biochim Biophys Acta. 1965 Jun 22;99(3):486–503. doi: 10.1016/s0926-6593(65)80202-8. [DOI] [PubMed] [Google Scholar]
- Eggleton P., Elsden S. R., Gough N. The estimation of creatine and of diacetyl. Biochem J. 1943;37(5):526–529. doi: 10.1042/bj0370526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ennor A. H., Stocken L. A. The preparation of sodium phosphocreatine. Biochem J. 1948;43(2):190–191. [PMC free article] [PubMed] [Google Scholar]
- GOA J. A micro biuret method for protein determination; determination of total protein in cerebrospinal fluid. Scand J Clin Lab Invest. 1953;5(3):218–222. doi: 10.3109/00365515309094189. [DOI] [PubMed] [Google Scholar]
- HOCH G. E., SCHNEIDER K. C., BURRIS R. H. Hydrogen evolution and exchange, and conversion of N2O to N2 by soybean root nodules. Biochim Biophys Acta. 1960 Jan 15;37:273–279. doi: 10.1016/0006-3002(60)90234-1. [DOI] [PubMed] [Google Scholar]
- Hardy R. W., Knight E., Jr, D'Eustachio A. J. An energy-dependent hydrogen-evolution from dithionite in nitrogen-fixing extracts of Clostridium pasteurianum. Biochem Biophys Res Commun. 1965 Sep 8;20(5):539–544. doi: 10.1016/0006-291x(65)90431-6. [DOI] [PubMed] [Google Scholar]
- Jeng D. Y., Devanathan T., Mortenson L. E. Components of cell-free extracts of clostridium pasteurianum W5 required for acetylene reduction and N2 fixation. Biochem Biophys Res Commun. 1969 Jun 6;35(5):625–633. doi: 10.1016/0006-291x(69)90450-1. [DOI] [PubMed] [Google Scholar]
- Mortenson L. E. Components of cell-free extracts of Clostridium pasteurianum required for ATP-dependent H2 evolution from dithionite and for N2 fixation. Biochim Biophys Acta. 1966 Sep 26;127(1):18–25. doi: 10.1016/0304-4165(66)90470-3. [DOI] [PubMed] [Google Scholar]
- Moustafa E., Mortenson L. E. Properties of azoferredoxin purified from nitrogen-fixing extracts of Clostridium pasteurianum. Biochim Biophys Acta. 1969 Jan 14;172(1):106–115. doi: 10.1016/0005-2728(69)90095-4. [DOI] [PubMed] [Google Scholar]
- Munson T. O., Dilworth M. J., Burris R. H. Method for demonstrating cofactor requirements for nitrogen fixation. Biochim Biophys Acta. 1965 Jun 15;104(1):278–281. doi: 10.1016/0304-4165(65)90245-x. [DOI] [PubMed] [Google Scholar]
- PECK H. D., Jr, GEST H. A new procedure for assay of bacterial hydrogenases. J Bacteriol. 1956 Jan;71(1):70–80. doi: 10.1128/jb.71.1.70-80.1956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stewart W. D., Fitzgerald G. P., Burris R. H. In situ studies on N2 fixation using the acetylene reduction technique. Proc Natl Acad Sci U S A. 1967 Nov;58(5):2071–2078. doi: 10.1073/pnas.58.5.2071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor K. B. The enzymology of nitrogen fixation in cell-free extracts of Clostridium pasteurianum. J Biol Chem. 1969 Jan 10;244(1):171–179. [PubMed] [Google Scholar]
- Van de Bogart M., Beinert H. Micro methods for the quantitative determination of iron and copper in biological material. Anal Biochem. 1967 Aug;20(2):325–334. doi: 10.1016/0003-2697(67)90038-3. [DOI] [PubMed] [Google Scholar]
- WESTLAKE D. W., WILSON P. W. Molecular hydrogen and nitrogen fixation by Clostridium pasteurianum. Can J Microbiol. 1959 Dec;5:617–620. doi: 10.1139/m59-075. [DOI] [PubMed] [Google Scholar]
- Winter H. C., Burris R. H. Stoichiometry of the adenosine triphosphate requirement for N2 fixation and H2 evolution by a partially purified preparation of Clostridium pasteurianum. J Biol Chem. 1968 Mar 10;243(5):940–944. [PubMed] [Google Scholar]


