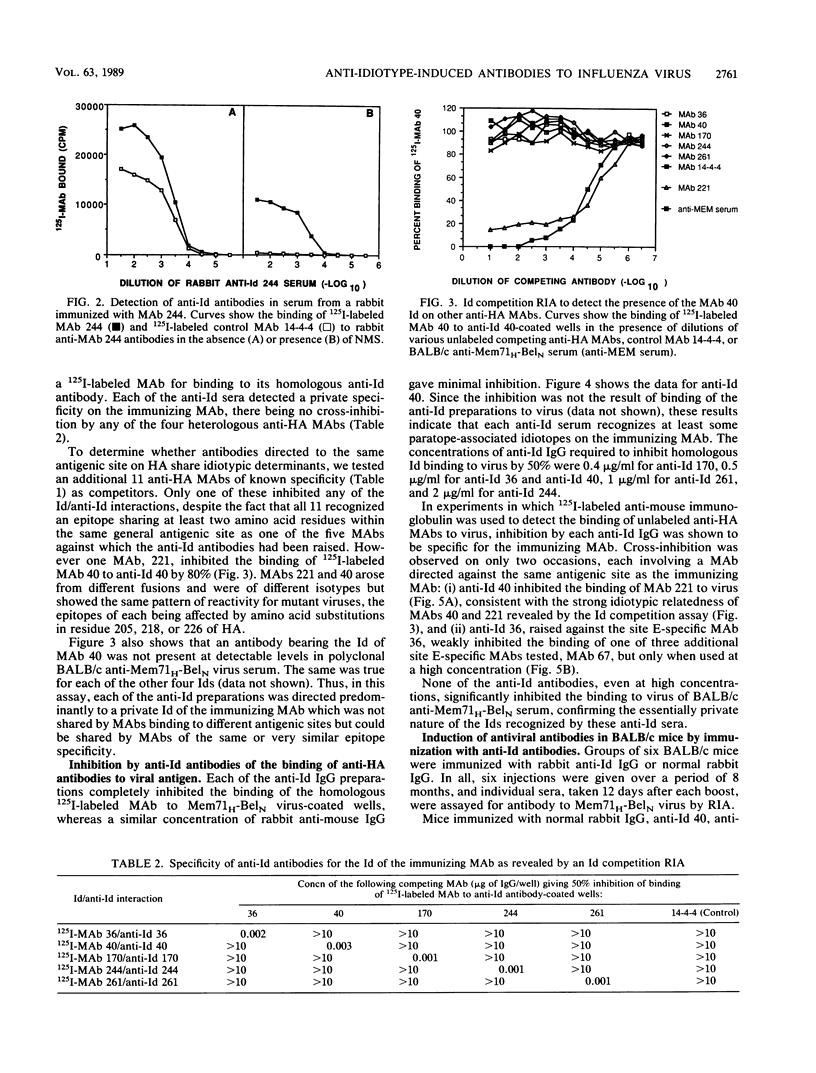
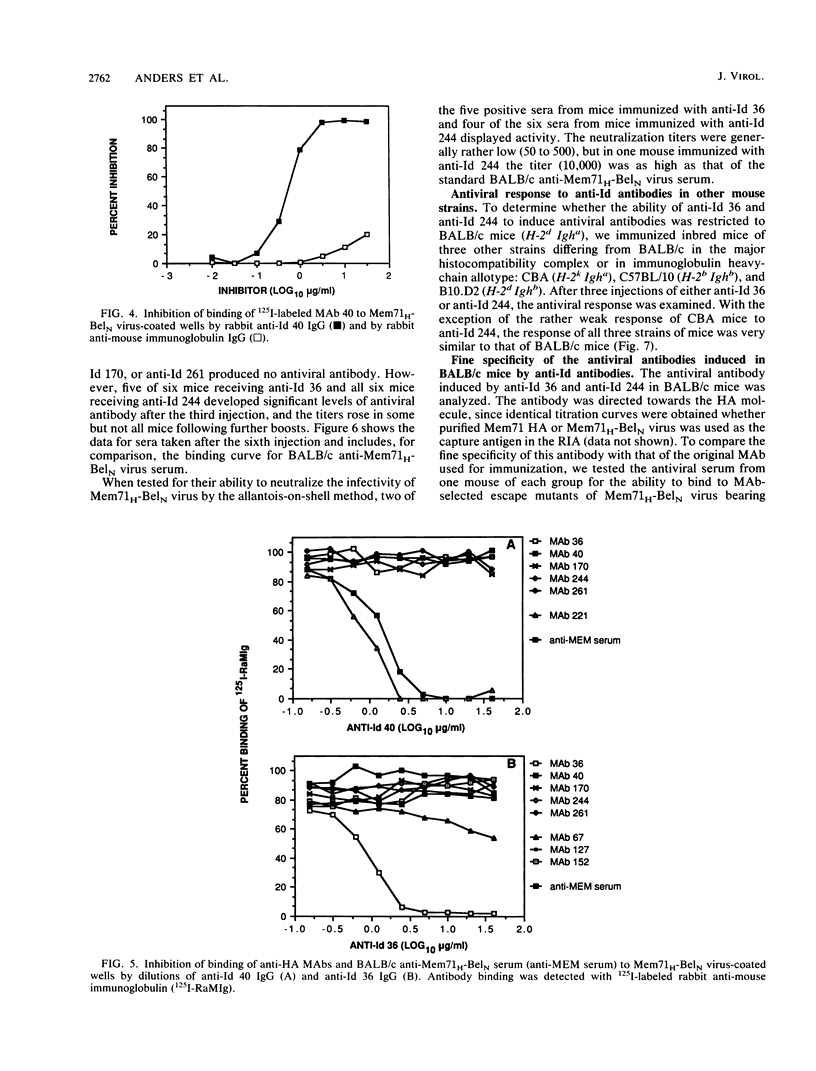
Abstract
Anti-idiotypic (anti-Id) antibodies were raised in rabbits against five monoclonal antibodies (MAbs) specific for different antigenic sites on the hemagglutinin (HA) of influenza virus Mem71H-BelN (H3N1) [A/Memphis/1/71 (H3N2) x A/Bel/42 (H1N1)]. Each of the anti-Id sera was directed predominantly towards a unique (private) idiotype of the immunizing MAb, none of the five idiotypes being detectable in pooled BALB/c antisera against Mem71H-BelN virus or on most other anti-HA MAbs tested. Partial idiotypic sharing was observed, however, between certain MAbs, from different mice, having the same or similar epitope specificity for HA. When used as immunogens in BALB/c mice, two of the five anti-Id preparations induced antibodies that reacted with Mem71H-BelN virus and displayed neutralizing activity. Mice of other inbred strains responded similarly, indicating that the response was not genetically restricted by the Igh locus. From their pattern of reactivity with mutants of Mem71H-BelN virus with known single amino acid substitutions in the HA molecule, the antiviral antibodies elicited by anti-Id antibodies were shown to be directed to the same antigenic site on A/Memphis/1/71 HA as the original immunizing MAb (site A or site E, respectively). However, several of these antisera were shown to contain additional distinct subpopulations of antibodies specific for heterologous influenza A virus strains, either of the H3 subtype or of a different HA subtype (H1 or H2). Since the induction of antibodies to HA of different subtypes is not a feature of the antibody response to influenza virus itself, their induction by anti-Id antibodies merits further investigation.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bluestone J. A., Sharrow S. O., Epstein S. L., Ozato K., Sachs D. H. Induction of anti-H-2 antibodies without alloantigen exposure by in vivo administration of anti-idiotype. Nature. 1981 May 21;291(5812):233–235. doi: 10.1038/291233a0. [DOI] [PubMed] [Google Scholar]
- Brown L. E., Ffrench R. A., Gawler J. M., Jackson D. C., Dyall-Smith M. L., Anders E. M., Tregear G. W., Duncan L., Underwood P. A., White D. O. Distinct epitopes recognized by I-Ad-restricted T-cell clones within antigenic site E on influenza virus hemagglutinin. J Virol. 1988 Jan;62(1):305–312. doi: 10.1128/jvi.62.1.305-312.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eichmann K., Rajewsky K. Induction of T and B cell immunity by anti-idiotypic antibody. Eur J Immunol. 1975 Oct;5(10):661–666. doi: 10.1002/eji.1830051002. [DOI] [PubMed] [Google Scholar]
- Ey P. L., Prowse S. J., Jenkin C. R. Isolation of pure IgG1, IgG2a and IgG2b immunoglobulins from mouse serum using protein A-sepharose. Immunochemistry. 1978 Jul;15(7):429–436. doi: 10.1016/0161-5890(78)90070-6. [DOI] [PubMed] [Google Scholar]
- FAZEKAS DE ST GROTH S., WHITE D. O. An improved assay for the infectivity of in influenza viruses. J Hyg (Lond) 1958 Mar;56(1):151–162. doi: 10.1017/s0022172400037621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fazekas de St Groth, Webster R. G. Disquisitions of Original Antigenic Sin. I. Evidence in man. J Exp Med. 1966 Sep 1;124(3):331–345. doi: 10.1084/jem.124.3.331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Francotte M., Urbain J. Induction of anti-tobacco mosaic virus antibodies in mice by rabbit antiidiotypic antibodies. J Exp Med. 1984 Nov 1;160(5):1485–1494. doi: 10.1084/jem.160.5.1485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GREENWOOD F. C., HUNTER W. M., GLOVER J. S. THE PREPARATION OF I-131-LABELLED HUMAN GROWTH HORMONE OF HIGH SPECIFIC RADIOACTIVITY. Biochem J. 1963 Oct;89:114–123. doi: 10.1042/bj0890114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gaulton G. N., Sharpe A. H., Chang D. W., Fields B. N., Greene M. I. Syngeneic monoclonal internal image anti-idiotopes as prophylactic vaccines. J Immunol. 1986 Nov 1;137(9):2930–2936. [PubMed] [Google Scholar]
- Gerhard W., Webster R. G. Antigenic drift in influenza A viruses. I. Selection and characterization of antigenic variants of A/PR/8/34 (HON1) influenza virus with monoclonal antibodies. J Exp Med. 1978 Aug 1;148(2):383–392. doi: 10.1084/jem.148.2.383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goding J. W. Conjugation of antibodies with fluorochromes: modifications to the standard methods. J Immunol Methods. 1976;13(3-4):215–226. doi: 10.1016/0022-1759(76)90068-5. [DOI] [PubMed] [Google Scholar]
- Hackett C. J., Dietzschold B., Gerhard W., Ghrist B., Knorr R., Gillessen D., Melchers F. Influenza virus site recognized by a murine helper T cell specific for H1 strains. Localization to a nine amino acid sequence in the hemagglutinin molecule. J Exp Med. 1983 Aug 1;158(2):294–302. doi: 10.1084/jem.158.2.294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hurwitz J. L., Herber-Katz E., Hackett C. J., Gerhard W. Characterization of the murine TH response to influenza virus hemagglutinin: evidence for three major specificities. J Immunol. 1984 Dec;133(6):3371–3377. [PubMed] [Google Scholar]
- Jerne N. K., Roland J., Cazenave P. A. Recurrent idiotopes and internal images. EMBO J. 1982;1(2):243–247. doi: 10.1002/j.1460-2075.1982.tb01154.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kennedy R. C., Eichberg J. W., Lanford R. E., Dreesman G. R. Anti-idiotypic antibody vaccine for type B viral hepatitis in chimpanzees. Science. 1986 Apr 11;232(4747):220–223. doi: 10.1126/science.3952505. [DOI] [PubMed] [Google Scholar]
- Lamb J. R., Green N. Analysis of the antigen specificity of influenza haemagglutinin-immune human T lymphocyte clones: identification of an immunodominant region for T cells. Immunology. 1983 Dec;50(4):659–666. [PMC free article] [PubMed] [Google Scholar]
- Laver W. G., Air G. M., Webster R. G., Gerhard W., Ward C. W., Dopheide T. A. Antigenic drift in type A influenza virus: sequence differences in the hemagglutinin of Hong Kong (H3N2) variants selected with monoclonal hybridoma antibodies. Virology. 1979 Oct 15;98(1):226–237. doi: 10.1016/0042-6822(79)90540-3. [DOI] [PubMed] [Google Scholar]
- Liu Y. N., Bona C. A., Schulman J. L. Idiotypy of clonal responses to influenza virus hemagglutinin. J Exp Med. 1981 Nov 1;154(5):1525–1538. doi: 10.1084/jem.154.5.1525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKean D., Huppi K., Bell M., Staudt L., Gerhard W., Weigert M. Generation of antibody diversity in the immune response of BALB/c mice to influenza virus hemagglutinin. Proc Natl Acad Sci U S A. 1984 May;81(10):3180–3184. doi: 10.1073/pnas.81.10.3180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mills K. H., Skehel J. J., Thomas D. B. Conformational-dependent recognition of influenza virus hemagglutinin by murine T helper clones. Eur J Immunol. 1986 Mar;16(3):276–280. doi: 10.1002/eji.1830160312. [DOI] [PubMed] [Google Scholar]
- Moran T., Liu Y. C., Schulman J. L., Bona C. A. Shared idiotopes among monoclonal antibodies specific for A/PR/8/34 (H1N1) and X-31(H3N2) influenza viruses. Proc Natl Acad Sci U S A. 1984 Mar;81(6):1809–1812. doi: 10.1073/pnas.81.6.1809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nepom J. T., Weiner H. L., Dichter M. A., Tardieu M., Spriggs D. R., Gramm C. F., Powers M. L., Fields B. N., Greene M. I. Identification of a hemagglutinin-specific idiotype associated with reovirus recognition shared by lymphoid and neural cells. J Exp Med. 1982 Jan 1;155(1):155–167. doi: 10.1084/jem.155.1.155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newton S. E., Air G. M., Webster R. G., Laver W. G. Sequence of the hemagglutinin gene of influenza virus A/Memphis/1/71 and previously uncharacterized monoclonal antibody-derived variants. Virology. 1983 Jul 30;128(2):495–501. doi: 10.1016/0042-6822(83)90277-5. [DOI] [PubMed] [Google Scholar]
- Nisonoff A., Lamoyi E. Implications of the presence of an internal image of the antigen in anti-idiotypic antibodies: possible application to vaccine production. Clin Immunol Immunopathol. 1981 Dec;21(3):397–406. doi: 10.1016/0090-1229(81)90228-2. [DOI] [PubMed] [Google Scholar]
- Ozato K., Mayer N., Sachs D. H. Hybridoma cell lines secreting monoclonal antibodies to mouse H-2 and Ia antigens. J Immunol. 1980 Feb;124(2):533–540. [PubMed] [Google Scholar]
- Reagan K. J., Wunner W. H., Wiktor T. J., Koprowski H. Anti-idiotypic antibodies induce neutralizing antibodies to rabies virus glycoprotein. J Virol. 1983 Dec;48(3):660–666. doi: 10.1128/jvi.48.3.660-666.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sacks D. L. Molecular mimicry of parasite antigens using anti-idiotypic antibodies. Curr Top Microbiol Immunol. 1985;119:45–55. doi: 10.1007/978-3-642-70675-2_4. [DOI] [PubMed] [Google Scholar]
- Schulman J. L., Moran T. M., Reale M., Beilin Y., Sigal N., Bona C. A. Effects of immunization with syngeneic monoclonal anti-idiotype antibody on antibody responses to influenza viruses. Monogr Allergy. 1987;22:143–149. [PubMed] [Google Scholar]
- Sharpe A. H., Gaulton G. N., McDade K. K., Fields B. N., Greene M. I. Syngeneic monoclonal antiidiotype can induce cellular immunity to reovirus. J Exp Med. 1984 Oct 1;160(4):1195–1205. doi: 10.1084/jem.160.4.1195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sigal N. H., Chan M., Reale M. A., Moran T., Beilin Y., Schulman J. L., Bona C. The human and murine influenza-specific B cell repertoires share a common idiotope. J Immunol. 1987 Sep 15;139(6):1985–1990. [PubMed] [Google Scholar]
- Staudt L. M., Gerhard W. Generation of antibody diversity in the immune response of BALB/c mice to influenza virus hemagglutinin. I. Significant variation in repertoire expression between individual mice. J Exp Med. 1983 Feb 1;157(2):687–704. doi: 10.1084/jem.157.2.687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- UytdeHaag F. G., Bunschoten H., Weijer K., Osterhaus A. D. From Jenner to Jerne: towards idiotype vaccines. Immunol Rev. 1986 Apr;90:93–113. doi: 10.1111/j.1600-065x.1986.tb01479.x. [DOI] [PubMed] [Google Scholar]
- Ward C. W. Structure of the influenza virus hemagglutinin. Curr Top Microbiol Immunol. 1981;94-95:1–74. doi: 10.1007/978-3-642-68120-2_1. [DOI] [PubMed] [Google Scholar]
- Webster R. G., Laver W. G. Determination of the number of nonoverlapping antigenic areas on Hong Kong (H3N2) influenza virus hemagglutinin with monoclonal antibodies and the selection of variants with potential epidemiological significance. Virology. 1980 Jul 15;104(1):139–148. doi: 10.1016/0042-6822(80)90372-4. [DOI] [PubMed] [Google Scholar]
- Wiley D. C., Skehel J. J. The structure and function of the hemagglutinin membrane glycoprotein of influenza virus. Annu Rev Biochem. 1987;56:365–394. doi: 10.1146/annurev.bi.56.070187.002053. [DOI] [PubMed] [Google Scholar]
- Wiley D. C., Wilson I. A., Skehel J. J. Structural identification of the antibody-binding sites of Hong Kong influenza haemagglutinin and their involvement in antigenic variation. Nature. 1981 Jan 29;289(5796):373–378. doi: 10.1038/289373a0. [DOI] [PubMed] [Google Scholar]
- Wilson I. A., Skehel J. J., Wiley D. C. Structure of the haemagglutinin membrane glycoprotein of influenza virus at 3 A resolution. Nature. 1981 Jan 29;289(5796):366–373. doi: 10.1038/289366a0. [DOI] [PubMed] [Google Scholar]


