Abstract
Five hybridomas that secrete monoclonal antibodies which neutralize the infectivity of lactate dehydrogenase-elevating virus (LDV) were isolated from BALB/c mice primed with Formalin-inactivated LDV. Competition analyses indicated that all five neutralizing monoclonal antibodies recognize contiguous, if not identical, epitopes on the envelope glycoprotein of LDV (VP-3) which are not recognized by nonneutralizing VP-3-specific monoclonal antibodies isolated from the same fusion. Despite the presence of neutralizing activity, polyclonal anti-LDV antibodies obtained from persistently infected mice did not compete for binding to LDV with four of the five neutralizing monoclonal antibodies tested. The results indicate that the envelope glycoprotein of LDV possesses a major neutralizing epitope which is poorly recognized, if at all, by mice during a natural infection but is rendered immunogenic by Formalin inactivation of the virus. The epitope was also not immunogenic in a rabbit, since its polyclonal LDV-neutralizing antibodies did not inhibit binding of the mouse monoclonal antibodies to LDV. Passive immunization with the neutralizing monoclonal antibodies did not protect mice from LDV infection and did not alter the course of infection. Neutralizing monoclonal antibodies have been used to select a neutralization escape variant by a novel combination of in vitro and in vivo isolation.
Full text
PDF

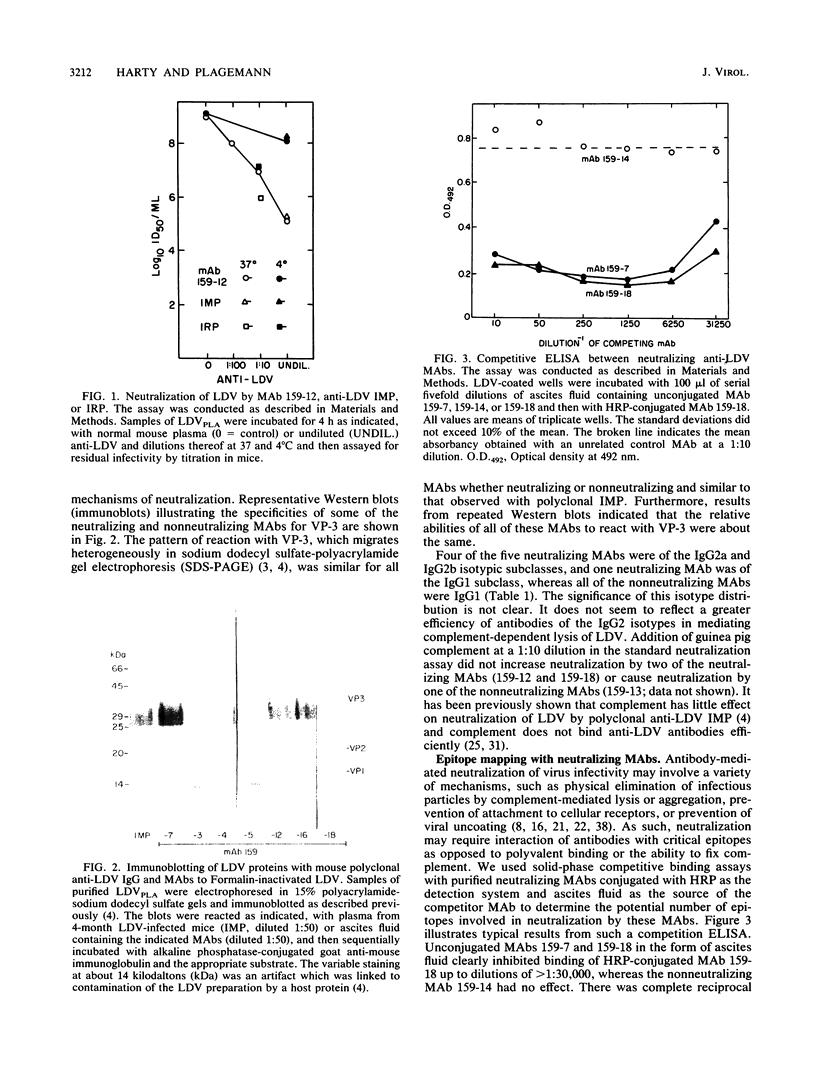
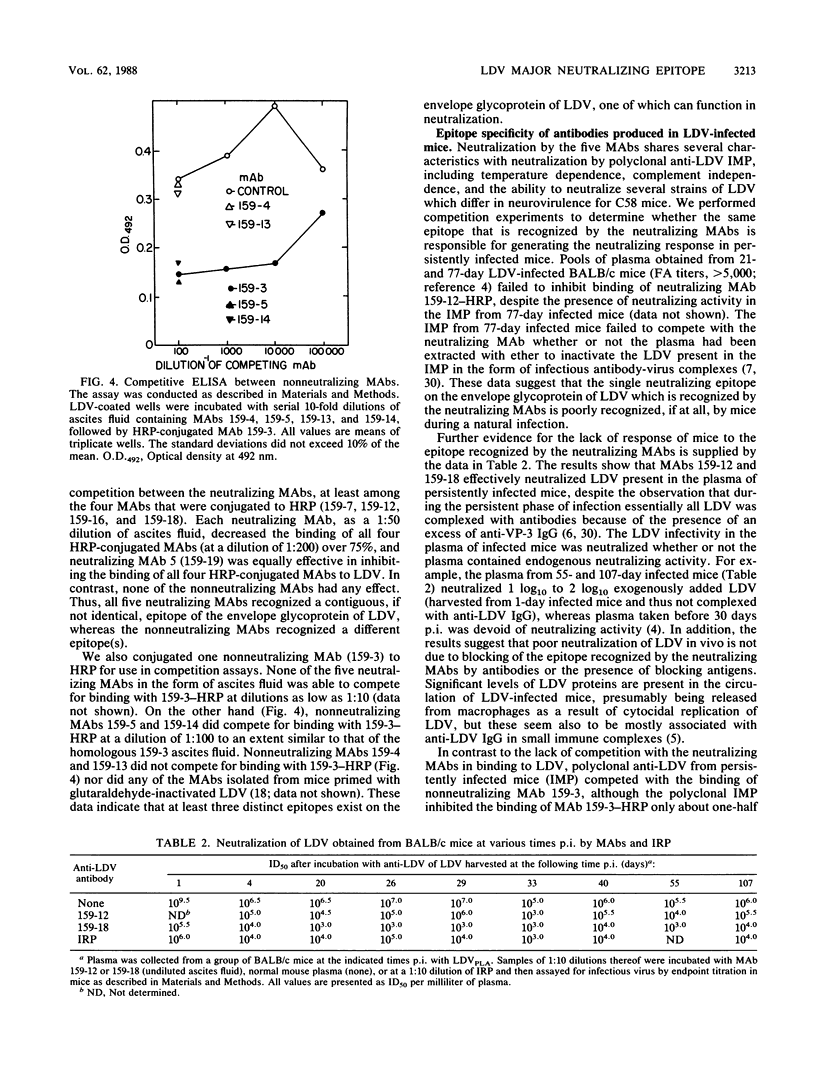



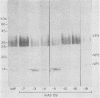
Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Berzofsky J. A. Intrinsic and extrinsic factors in protein antigenic structure. Science. 1985 Sep 6;229(4717):932–940. doi: 10.1126/science.2410982. [DOI] [PubMed] [Google Scholar]
- Berzofsky J. A. T-B reciprocity. An Ia-restricted epitope-specific circuit regulating T cell-B cell interaction and antibody specificity. Surv Immunol Res. 1983;2(3):223–229. doi: 10.1007/BF02918417. [DOI] [PubMed] [Google Scholar]
- Brinton-Darnell M., Plagemann P. G. Structure and chemical-physical characteristics of lactate dehydrogenase-elevating virus and its RNA. J Virol. 1975 Aug;16(2):420–433. doi: 10.1128/jvi.16.2.420-433.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cafruny W. A., Chan S. P., Harty J. T., Yousefi S., Kowalchyk K., McDonald D., Foreman B., Budweg G., Plagemann P. G. Antibody response of mice to lactate dehydrogenase-elevating virus during infection and immunization with inactivated virus. Virus Res. 1986 Sep;5(4):357–375. doi: 10.1016/0168-1702(86)90029-8. [DOI] [PubMed] [Google Scholar]
- Cafruny W. A., Heruth D. P., Jaqua M. J., Plagemann P. G. Immunoglobulins that bind to uncoated ELISA plate surfaces: appearance in mice during infection with lactate-dehydrogenase-elevating virus and in human anti-nuclear antibody-positive sera. J Med Virol. 1986 Jun;19(2):175–186. doi: 10.1002/jmv.1890190211. [DOI] [PubMed] [Google Scholar]
- Cafruny W. A., Plagemann P. G. Immune response to lactate dehydrogenase-elevating virus: isolation of infectious virus-immunoglobulin G complexes and quantitation of specific antiviral immunoglobulin G response in wild-type and nude mice. Infect Immun. 1982 Sep;37(3):1001–1006. doi: 10.1128/iai.37.3.1001-1006.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cafruny W. A., Plagemann P. G. Immune response to lactate dehydrogenase-elevating virus: serologically specific rabbit neutralizing antibody to the virus. Infect Immun. 1982 Sep;37(3):1007–1012. doi: 10.1128/iai.37.3.1007-1012.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coutelier J. P., Van Roost E., Lambotte P., Van Snick J. The murine antibody response to lactate dehydrogenase-elevating virus. J Gen Virol. 1986 Jun;67(Pt 6):1099–1108. doi: 10.1099/0022-1317-67-6-1099. [DOI] [PubMed] [Google Scholar]
- Dalziel R. G., Lampert P. W., Talbot P. J., Buchmeier M. J. Site-specific alteration of murine hepatitis virus type 4 peplomer glycoprotein E2 results in reduced neurovirulence. J Virol. 1986 Aug;59(2):463–471. doi: 10.1128/jvi.59.2.463-471.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diamond D. C., Jameson B. A., Bonin J., Kohara M., Abe S., Itoh H., Komatsu T., Arita M., Kuge S., Nomoto A. Antigenic variation and resistance to neutralization in poliovirus type 1. Science. 1985 Sep 13;229(4718):1090–1093. doi: 10.1126/science.2412292. [DOI] [PubMed] [Google Scholar]
- Engvall E. Enzyme immunoassay ELISA and EMIT. Methods Enzymol. 1980;70(A):419–439. doi: 10.1016/s0076-6879(80)70067-8. [DOI] [PubMed] [Google Scholar]
- Ey P. L., Prowse S. J., Jenkin C. R. Isolation of pure IgG1, IgG2a and IgG2b immunoglobulins from mouse serum using protein A-sepharose. Immunochemistry. 1978 Jul;15(7):429–436. doi: 10.1016/0161-5890(78)90070-6. [DOI] [PubMed] [Google Scholar]
- Fleming J. O., Trousdale M. D., el-Zaatari F. A., Stohlman S. A., Weiner L. P. Pathogenicity of antigenic variants of murine coronavirus JHM selected with monoclonal antibodies. J Virol. 1986 Jun;58(3):869–875. doi: 10.1128/jvi.58.3.869-875.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friguet B., Djavadi-Ohaniance L., Goldberg M. E. Some monoclonal antibodies raised with a native protein bind preferentially to the denatured antigen. Mol Immunol. 1984 Jul;21(7):673–677. doi: 10.1016/0161-5890(84)90053-1. [DOI] [PubMed] [Google Scholar]
- Gollins S. W., Porterfield J. S. A new mechanism for the neutralization of enveloped viruses by antiviral antibody. Nature. 1986 May 15;321(6067):244–246. doi: 10.1038/321244a0. [DOI] [PubMed] [Google Scholar]
- Harty J. T., Chan S. P., Plagemann P. G. Characteristics of monoclonal antibodies to the lactate dehydrogenase-elevating virus. Intervirology. 1987;27(1):53–60. doi: 10.1159/000149715. [DOI] [PubMed] [Google Scholar]
- Hollander Z., Katchalski-Katzir E. Use of monoclonal antibodies to detect conformational alterations in lactate dehydrogenase isoenzyme 5 on heat denaturation and on adsorption to polystyrene plates. Mol Immunol. 1986 Sep;23(9):927–933. doi: 10.1016/0161-5890(86)90122-7. [DOI] [PubMed] [Google Scholar]
- Hu S. L., Fultz P. N., McClure H. M., Eichberg J. W., Thomas E. K., Zarling J., Singhal M. C., Kosowski S. G., Swenson R. B., Anderson D. C. Effect of immunization with a vaccinia-HIV env recombinant on HIV infection of chimpanzees. Nature. 1987 Aug 20;328(6132):721–723. doi: 10.1038/328721a0. [DOI] [PubMed] [Google Scholar]
- Lachmann P. J. Antibody and complement in viral infections. Br Med Bull. 1985 Jan;41(1):3–6. doi: 10.1093/oxfordjournals.bmb.a072020. [DOI] [PubMed] [Google Scholar]
- Martinez D., Brinton M. A., Tachovsky T. G., Phelps A. H. Identification of lactate dehydrogenase-elevating virus as the etiological agent of genetically restricted, age-dependent polioencephalomyelitis of mice. Infect Immun. 1980 Mar;27(3):979–987. doi: 10.1128/iai.27.3.979-987.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McDonald T. L. Isolation of Clq-binding virus-antibody immune complexes from lactic dehydrogenase virus (LDV)-infected mice. Immunology. 1982 Feb;45(2):365–370. [PMC free article] [PubMed] [Google Scholar]
- Mierendorf R. C., Jr, Dimond R. L. Functional heterogeneity of monoclonal antibodies obtained using different screening assays. Anal Biochem. 1983 Nov;135(1):221–229. doi: 10.1016/0003-2697(83)90754-6. [DOI] [PubMed] [Google Scholar]
- Minor P. D., Evans D. M., Ferguson M., Schild G. C., Westrop G., Almond J. W. Principal and subsidiary antigenic sites of VP1 involved in the neutralization of poliovirus type 3. J Gen Virol. 1985 May;66(Pt 5):1159–1165. doi: 10.1099/0022-1317-66-5-1159. [DOI] [PubMed] [Google Scholar]
- Murphy W. H., Nawrocki J. F., Pease L. R. Age-dependent paralytic viral infection in C58 mice: possible implications in human neurologic disease. Prog Brain Res. 1983;59:291–303. doi: 10.1016/S0079-6123(08)63874-1. [DOI] [PubMed] [Google Scholar]
- Notkins A. L. Enzymatic and immunologic alterations in mice infected with lactic dehydrogenase virus. Am J Pathol. 1971 Sep;64(3):733–746. [PMC free article] [PubMed] [Google Scholar]
- Notkins A. L., Mahar S., Scheele C., Goffman J. Infectious virus-antibody complex in the blood of chronically infected mice. J Exp Med. 1966 Jul 1;124(1):81–97. doi: 10.1084/jem.124.1.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oldstone M. B., Buchmeier M. J., Doyle M. V., Tishon A. Virus-induced immune complex disease: specific anti-viral antibody and C1q binding material in the circulation during persistent lymphocytic choriomeningitis virus infection. J Immunol. 1980 Feb;124(2):831–838. [PubMed] [Google Scholar]
- Ritzi D. M., Holth M., Smith M. S., Swart W. J., Cafruny W. A., Plagemann G. W., Stueckemann J. A. Replication of lactate dehydrogenase-elevating virus in macrophages. 1. Evidence for cytocidal replication. J Gen Virol. 1982 Apr;59(Pt 2):245–262. doi: 10.1099/0022-1317-59-2-245. [DOI] [PubMed] [Google Scholar]
- Roivainen M., Hovi T. Intestinal trypsin can significantly modify antigenic properties of polioviruses: implications for the use of inactivated poliovirus vaccine. J Virol. 1987 Dec;61(12):3749–3753. doi: 10.1128/jvi.61.12.3749-3753.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rowson K. E., Mahy B. W. Lactate dehydrogenase-elevating virus. J Gen Virol. 1985 Nov;66(Pt 11):2297–2312. doi: 10.1099/0022-1317-66-11-2297. [DOI] [PubMed] [Google Scholar]
- Stec D. S., Waddell A., Schmaljohn C. S., Cole G. A., Schmaljohn A. L. Antibody-selected variation and reversion in Sindbis virus neutralization epitopes. J Virol. 1986 Mar;57(3):715–720. doi: 10.1128/jvi.57.3.715-720.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stueckemann J. A., Holth M., Swart W. J., Kowalchyk K., Smith M. S., Wolstenholme A. J., Cafruny W. A., Plagemann P. G. Replication of lactate dehydrogenase-elevating virus in macrophages. 2. Mechanism of persistent infection in mice and cell culture. J Gen Virol. 1982 Apr;59(Pt 2):263–272. doi: 10.1099/0022-1317-59-2-263. [DOI] [PubMed] [Google Scholar]
- Thomas A. A., Vrijsen R., Boeyé A. Relationship between poliovirus neutralization and aggregation. J Virol. 1986 Aug;59(2):479–485. doi: 10.1128/jvi.59.2.479-485.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]