Abstract
A double-blind, randomized phase I clinical trial was carried out to compare mefloquine with sulfadoxine—pyrimethamine for safety and tolerance. Twenty adult male Brazilian subjects from areas endemic for malaria were studied for a period of 66 days, which included 2 days of basal studies and a 63-day follow-up after drug administration. Subjects received either mefloquine, given as a single oral dose of 1000 mg (4 × 250-mg tablets) or sulfadoxine—pyrimethamine (2 tablets, each containing 500 mg of sulfadoxine plus 25 mg of pyrimethamine). Clinical examination, electrocardiogram, chest X-ray, and haematological, biochemical, stool, and urine analyses were carried out before drug administration and at various intervals afterwards. Peripheral blood smears were examined for malarial parasites.
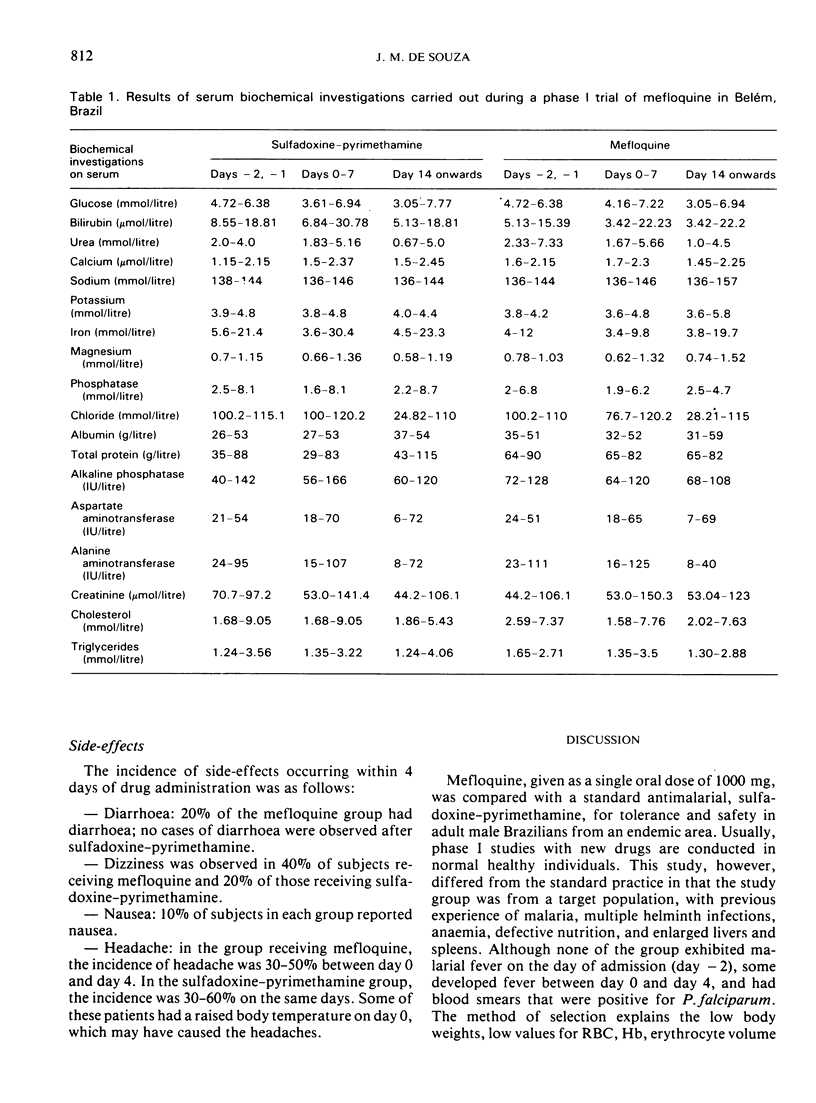
Both drugs were well tolerated and safe as seen from the absence of drug-induced changes in the various laboratory assay results. There was an improvement in body weight, red blood cell count, haemoglobin, and erythrocyte volume fraction values for all patients during the study. In subjects who had positive smears for Plasmodium falciparum, mefloquine produced complete clearance on day 1 with an S-type response (3 cases). Sulfadoxine—pyrimethamine produced complete clearance on day 2 in 5 subjects, but a delayed RI-type response (recrudescence) was observed in 2 cases and an early RI response in one case. P. vivax relapses occurred in both groups. Side-effects of mefloquine included mild diarrhoea (20%) and dizziness (40%); dizziness was also observed with sulfadoxine—pyrimethamine (20%). In both groups, side-effects were mild, short-lived and needed no specific treatment.
Thus, mefloquine in an oral dose of 1000 mg was found to be well tolerated and safe in adult male Brazilian volunteers from endemic areas. No drug-related adverse reactions were observed. In cases where P. falciparum infection was present, there was a complete parasite clearance with no recrudescence.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Clyde D. F., McCarthy V. C., Miller R. M., Hornick R. B. Suppressive activity of mefloquine in sporozoite-induced human malaria. Antimicrob Agents Chemother. 1976 Mar;9(3):384–386. doi: 10.1128/aac.9.3.384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doberstyn E. B., Phintuyothin P., Noeypatimanondh S., Teerakiartkamjorn C. Single-dose therapy of falciparum malaria with mefloquine or pyrimethamine-sulfadoxine. Bull World Health Organ. 1979;57(2):275–279. [PMC free article] [PubMed] [Google Scholar]
- Hall A. P., Doberstyn E. B., Karnchanachetanee C., Samransamruajkit S., Laixuthai B., Pearlman E. J., Lampe R. M., Miller C. F., Phintuyothin P. Sequential treatment with quinine and mefloquine or quinine and pyrimethamine-sulfadoxine for falciparum malaria. Br Med J. 1977 Jun 25;1(6077):1626–1628. doi: 10.1136/bmj.1.6077.1626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rozman R. S., Canfield C. J. New experimental antimalarial drugs. Adv Pharmacol Chemother. 1979;16:1–43. doi: 10.1016/s1054-3589(08)60241-0. [DOI] [PubMed] [Google Scholar]
- da SILVA J., LOPES P. F., FERREIRA L. F., MORTEO R., NAVEIRA J. B. [Resistance of P. falciparum to the action of chloroquine (preliminary note)]. Hospital (Rio J) 1961 Nov;60:581–594. [PubMed] [Google Scholar]


