Abstract
The majority of recent preclinical gene therapy studies targeting the retina have used adeno-associated virus (AAV) as the gene transfer vector. However, AAV has several limitations including the ability to generate innate inflammatory responses, the ability to cause insertional mutagenesis at a frequency of up to 56% in some tissues and a limited cloning capacity of 4.8Kb. Furthermore, AAV is known to generate limiting immune responses in humans despite the absence of similar immune responses in preclinical canine and murine studies. Three clinical trials to treat Leber’s congenital amaurosis using AAV are under way. A clinical trial to treat Stargardt’s using lentivirus vectors has also been recently announced. However, very limited evidence currently exists that lentivirus vectors can efficiently transduce photoreceptor cells. In contrast, very few preclinical ocular gene therapy studies have utilized adenovirus as the gene therapy vector. Nonetheless, the only two ocular gene therapy clinical trials performed to date have each used adenovirus as the vector and more significantly, in these published trials there has been no observed serious adverse event. These trials appear to be poised for Phase II/III status. Activation of cytotoxic T lymphocytes limits duration of transgene expression in the retina from first generation adenovirus vectors. However, an advanced class of adenovirus vectors referred to as Helper-dependent Adenovirus (Hd-Ad) have recently been shown to be capable of expressing transgenes in ocular tissues for more than one year. Hd-Ad vectors have many properties that potentially warrant their inclusion in the retinal gene therapy toolbox for the treatment of retinal degenerative diseases.
Progress in Retinal Gene Therapy
During the previous decade, substantial progress has been made in preclinical ocular gene therapy. Of particular note are several studies demonstrating long-term correction of the visual defect in animal models of Leber’s congenital amaurosis (LCA) (Acland et al., 2001; Le Meur et al., 2007; Narfstrom et al., 2003). In each of these studies, an adeno-associated virus (AAV) vector was used to deliver the RPE65 cDNA to the retinal pigment epithelium (RPE) of LCA dogs. These studies have led to the initiation of two LCA clinical trials in the United States and one in the United Kingdom. The majority of additional gene therapy studies demonstrating long-term rescue of retinal degeneration in animal models have also focused on the delivery of transgenes to the RPE (Tschernutter et al., 2005). However, some disorders such as retinitis pigmentosa (RP), are much more common than LCA and are typically associated with degeneration of the rod photoreceptor cells due to mutations in genes expressed exclusively in those cells (Hartong et al., 2006). In contrast to RPE-associated diseases, there are no published studies thus far demonstrating evidence of efficacious and long-term somatic rescue of rod photoreceptor degeneration caused by mutations in photoreceptor specific genes. While the rods don’t play a significant role in daylight vision, their degeneration leads to the eventual loss of cones; hence the rods are important target cells for retinal gene therapy. Herein, I identify the current barriers and potential solutions for progress in gene therapy for rod photoreceptor degeneration and ocular gene therapy in general. In brief, given the availability of a large number of well-characterized animal models of retinal degeneration (Chang et al., 2005) along with our substantial knowledge of the genetic basis underlying retinal degeneration, the major obstacles for successful retinal gene therapy are a lack of appropriate gene transfer vectors that are capable of delivering transgenes efficiently and in the appropriate context to retinal cells. A variety of reviews (Auricchio and Rolling, 2005; Bainbridge et al., 2006; Rolling, 2004; Surace and Auricchio, 2007) summarizing successful preclinical retinal gene transfer have already been published and hence those studies are not discussed here. Instead, here we focus almost exclusively on issues that were least discussed previously, i.e. the major vector-associated barriers that need to be overcome in order to achieve safe and efficacious retinal gene therapy in the clinic.
Before a discussion of each specific barrier that needs to be overcome, it is worth addressing what are the properties of the ideal gene transfer vector for retinal cells. A case will be made for such a hypothetical vector as having the following properties: non integrating or episomal existence in the nucleus, high levels of tropism for the target cell in post mitotic retina, a large transgene cloning capacity, long-term transgene expression, lack of an immune response and the ability to re administer the vector if necessary. The discussion of current limitations will be focused primarily on the most commonly used vectors in ocular gene therapy thus far, namely AAV, Lentivirus (Lv) and Adenovirus (Ad). In contrast to previously published reviews, a case will be presented here for why Ad should also be considered as a potential gene therapy vector for ocular diseases. The objective is not to claim that Ad is superior to AAV or Lv but rather to highlight recent developments in Ad vector technology and its application in ocular tissues that warrant further consideration.
Integrating Vectors Cause Insertional Mutagenesis
The retina is generally considered to be comprised entirely of post-mitotic cells. Hence, dilutional loss of an episomal transgene due to cell division is not a significant barrier in retinal gene therapy. Historically, it has been assumed that long-term transgene expression requires integration of the transgene into the nuclear genome. As will be discussed, integration of the transgene is not a requirement for long-term transgene expression.
One of the most remarkable studies demonstrating that gene therapy is a viable approach for treating genetic disease in humans involved the successful treatment of eleven children suffering from Severe Combined Immunodeficiency Disease (SCID)-X1 (Cavazzana-Calvo et al., 2000). In those studies, the integrating Moloney retrovirus vector was utilized to deliver a cDNA encoding the γc cytokine receptor ex vivo. This integrating vector however, also caused insertional mutagenesis, leukemia in four and death in some of those same patients (Kaiser, 2003). Hence, based on at least these studies, the case for the absolute need for integrating vectors is weakened should persistence of gene expression be possible without integration. Indeed, after the clinical trials were initiated, it was discovered that mice treated with an analogous retrovirus vector also developed leukemia-like symptoms (Li et al., 2002). There is significant debate whether the human clinical trials were conducted prematurely given the data obtained from animals at a later date. An argument has been made that these unfortunate results were due to the unique transgene utilized in the vector, since other similar trials in the UK utilizing retrovirus based gene transfer did not have insertional mutagenesis noted in them. Accordingly, it must be noted that the dose of the virus administered (as well as the absolute number of successfully transduced stem cells infused into these patients) was at a level not previously achieved in other retrovirus trials. The UK trials were continued and those trials also ultimately led to T-cell leukemia in some patients. For purposes of this discussion, it should be noted however that gene transfer in these trials was performed ex vivo in proliferating cells, a condition not analogous to post mitotic retinal tissues. However, the need for cell proliferation is not a strict requirement for insertional mutagenesis.
The issue of insertional mutagenesis has now also become significant relative to AAV based gene transfer. Wild type AAV vectors naturally integrate at a unique site in human chromosome 19 known as AAVS1 (Kotin et al., 1990). Site-specific recombination is mediated through the AAV Rep proteins (Snyder et al., 1990). In order to make space for the transgene, recombinant AAV vectors lack Rep encoding genes. Despite loss of Rep, these vectors still integrate, but now randomly in human cells (Ponnazhagan et al., 1997). For example, in studies involving AAV mediated gene transfer to the liver, it has been shown that the predominant form of AAV genomes are present as episomal concatemers. Further studies revealed multiple integration sites in the hepatocyte genome-sites that have been mapped and found to be localized to active genes with integration adjacent to cancer-related genes occurring at a rate of 3.5% (Nakai et al., 2005). In addition, AAV vectors integrate on chromosome 1 and exist in the nucleus as mini circles (Schnepp et al., 2005).
Insertional mutagenesis caused by recombinant AAV vectors was suspected in some earlier studies involving the delivery of β-glucuronidase (GUSB) to mucopolysaccharidosis type VII (MPSVII) mice (Donsante et al., 2001). Three out of five mice treated with recombinant AAV developed hepatocellular carcinomas and angiosarcomas, indicating a possible correlation of tumor formation with AAV. The consensus in the AAV gene therapy community was that these data were likely specific to MPSVII mice, which may for unknown reasons be highly susceptible to carcinomas (Marshall, 2001). However, in some more recently published studies it has been found that normal mice injected with AAV expressing GUSB develop hepatocellular carcinoma at the alarmingly high rate of 56%. These investigators identified changes in gene expression profiles on chromosome 12 in the proximity of the mir-341 microRNA transcript and implicated insertional mutagenesis as the source of hepatocellular carcinomas (Donsante et al., 2007). No tumors were observed in transgenic mice over-expressing GUSB. Mice injected with AAV vectors devoid of a transgene-regulating promoter developed carcinomas at a rate of 33% (Donsante et al., 2007), directly implicating AAV as the potential source of the carcinomas. It should be mentioned however, that in this study normal uninjected mice also developed carcinomas, but at a substantially lower frequency of 8.3% (Donsante et al., 2007). This study has rekindled the debate about the safety of AAV vectors. The counter arguments offered are that the latest studies were performed in newborn mice that might be more susceptible to carcinomas due to the ease of whole body transduction (Kay, 2007). Relevant to our discussion, these observations do raise the question whether integrating vectors such as AAV or lentivirus are a good platform for retinal gene therapy if episomal vectors could efficiently express transgenes long term. Development of non-integrating Lv vectors is now being pursued (Yanez-Munoz et al., 2006), in part prompted by the insertional mutagenesis observed in the SCID-X1 clinical trials.
In July 2007, a patient being treated for rheumatoid arthritis by AAV-mediated delivery of tgAAC94, an inhibitor of tumor necrosis factor α, died unexpectedly, temporarily raising additional fears that vectors such as AAV are perhaps not as safe as initially presumed. An expert panel failed to definitively rule out the role of AAV in that death but also failed to find evidence directly implicating AAV (Kaiser, 2007). Hence, those trials have resumed.
While recombinant AAV and Lv integrate randomly into the genome of human cells, Ad is generally a non-integrating vector that persists in the nucleus as an episome (Russell, 2000). However, at high multiplicities of infection, Ad can also integrate (Neumann and Doerfler, 1981) but integration can in some cases be engineered to occur site-specifically (Wang and Lieber, 2006).
Photoreceptor Tropism is Dictated by Stage of Retinal Development
The majority of studies attempting to rescue rod photoreceptor degeneration in mouse models of RP have administered the vector during retinal development and differentiation between post-natal days 3 and 21 and not the post mitotic adult retina (Ali et al., 2000; Bennett et al., 1996; Jomary et al., 1997; Kumar-Singh and Farber, 1998; Pawlyk et al., 2005). While the need for this timing was based primarily on achieving therapeutic intervention prior to the onset of significant photoreceptor degeneration, results from these studies have limited relevance for gene therapy in the human retina unless these vectors can be demonstrated to also infect fully developed photoreceptors and not only photoreceptor precursor cells. This caveat is critical to understand because the tropism of viral vectors for different cell types is dictated by stage of retinal development. Retinal development at postnatal days 3 to 7 in mice is roughly equivalent to the 2nd trimester of retinal development in-utero in humans (Reh, 2006). Indeed, in newborn mice the retina is still undergoing significant cell proliferation, providing for the opportunity of vectors requiring cell division for transductional capability (i.e. retroviruses) to have a chance at showing efficacy. Rod birth in neonatal mice continues up to p11 (Cepko et al., 1996). This is in contrast to human retina where the entire central retina is free of cell division by fetal week 12 (Georges et al., 1999; Penfold and Provis, 1986) and the life-time supply of rod photoreceptors is largely completed in utero (Linberg and Fisher, 1990). Retrovirus vectors can infect only dividing cells and hence can be used to label photoreceptors (Price et al., 1987) and other retinal cells during development when the nuclear membrane is breaking down and allowing nuclear access to the retrovirus genome. These same retrovirus vectors do not transduce adult murine retina that is post mitotic. Similarly, while AAV, Ad and Lv can all transduce both photoreceptors and RPE very efficiently during murine retinal development, none of these vectors can efficiently transduce rod photoreceptors in the adult murine retina but are limited primarily to transducing the RPE after subretinal administration (Ali et al., 1996; Cashman et al., 2002; Pang et al., 2006; Pang et al., 2004). This significant shift in tropism coincides with the end of photoreceptor birth, at approximately p10 and complete differentiation of the murine retina by p21 (Cepko et al., 1996). Since each of these vectors can infect post mitotic cells, the lack of efficacy may be due to a down-regulation in the respective viral receptors present on the photoreceptor plasma membrane. Similar observations have been made for muscle cells, where myoblasts in neonatal mice are more amenable to infection by Ad relative to the more developed and specialized myotube fibers found in adults (Acsadi et al., 1994).
The limitation of RPE tropism can be overcome by the method of pseudotyping, involving exchange of the naturally occurring coat proteins of the viral vector with heterologous proteins from a virus that does have a tropism for photoreceptors. For example, AAV serotype 2 (AAV2) can be pseudotyped with AAV8 coat proteins to enhance photoreceptor tropism in adult mice (Allocca et al., 2007). Interestingly, while the majority of studies in mice and rats expressing GFP from a CMV promoter in the context of AAV2 indicate that transgene expression is primarily in the RPE and very limited in the photoreceptors, the same experiments in monkeys indicate that transduction is almost exclusively of the rod photoreceptors (Bennett et al., 1999). These data would indicate that either the CMV promoter is not active in the RPE of monkeys or that AAV2 does not transduce the RPE of monkeys. In a similar study, CMV regulated GFP in the context of AAV2 did not reveal exclusive transduction of rod photoreceptors by this vector (Le Meur et al., 2005). In this latter study, the investigators attributed low levels of GFP in the RPE due to quenching of GFP by pigment in the RPE (Le Meur et al., 2005). An alternative explanation for these observations is that high levels of RPE transduction by AAV2 is toxic and incidentally atrophy of RPE cells was reported in the former monkey studies (Bennett et al., 1999). More recently, dropout of GFP-positive RPE cells and retinal pathology has been observed in AAV-transduced RPE in monkeys (Vandenberghe et al., 2007). Clearly, data obtained using non-human primates does not correlate well with small animal studies or those performed in dogs. However, not all studies have been performed with GMP grade vectors and that may in part explain the varying results. Nonetheless, given that all currently ongoing LCA clinical trials are using AAV2 as the gene transfer vector, and the non-human primate eye is closer in structure and physiology to the human eye than are eyes of rodents or dogs, further studies on the performance of AAV2 in non-human primate retina may be warranted.
Lv has been used to transduce the retina of rat or mouse pups and found to efficiently deliver GFP to photoreceptors, but in adults, transduction primarily of the RPE is observed (Miyoshi et al., 1997; Pang et al., 2006). Such vectors have been used to slow rod photoreceptor degeneration in rd1 mice but only when the vector is introduced in neonates (Takahashi et al., 1999). Importantly, such vectors do not transduce adult murine photoreceptors efficiently (Bainbridge et al., 2001) and hence are not predicted to be very useful in human photoreceptor gene therapy except if they were to be administered in utero, currently an untenable task. In one study, photoreceptor transduction in adult murine retina has been achieved using a lentivirus vector pseudotyped with equine infectious anemia virus (EIAV) but the levels of photoreceptor transduction were described by the investigators as ‘variable’ (Balaggan et al., 2006). In another study, VSV-G pseudotyped lentivirus was also shown to transduce photoreceptors in adult rats but again the levels of transduction were described by the authors as ‘sporadic’ (Bemelmans et al., 2005). Nonetheless, a clinical trial for Stargardt’s disease using lentivirus vectors (Stargen®, Oxford Biomedica) has been announced to begin within the next one year despite the apparent failure of lentivirus mediated transduction of photoreceptors -the target cells in those clinical trials. A variety of promoter constructs for photoreceptor specific gene expression using EIAV vectors have been developed, but transgene expression from those vectors was tested by administration of vector in neonatal murine retina at p5 (Nicoud et al., 2007). Administration of lentivirus expressing GFP from a rhodopsin promoter into adult rats leads to poor gene expression in the photoreceptors, confirming that gene transfer is limited primarily at the level of transduction and not gene expression per se (Bemelmans et al., 2005; Miyoshi et al., 1997). Hence, currently available evidence may indicate that lentivirus vectors are not yet significantly developed for delivery of transgenes to photoreceptors and hence significant further development of these vectors may be necessary prior to the initiation of clinical trials targeting photoreceptors. Interestingly, the studies documenting ‘variable’ transduction of photoreceptors by Lv were also performed by Oxford Biomedica (Balaggan et al., 2006),(Nicoud et al., 2007). Hence, one assumes that Stargardt patients enrolled in a clinical trial will be treated with an as yet unpublished lentivirus vector that does efficiently transduce post mitotic photoreceptors.
Similar to AAV and Lv, Ad also transduces primarily the RPE in adult murine retina despite being able to transduce photoreceptors in neonatal retina (Pang et al., 2004). Indeed, some of the first studies demonstrating a delay in rod photoreceptor degeneration utilized Ad as the gene transfer vector (Bennett et al., 1996; Kumar-Singh and Farber, 1998). In those studies, Ad was administered prior to p10-a stage prior to the purported shift in tropism of Ad from photoreceptors/RPE to only RPE. Pseudotyping of Ad by exchanging the fiber protein has led to an improvement in tropism towards photoreceptors but those vectors still preferentially transduced the RPE, and antibodies against GFP were necessary to detect GFP in the photoreceptors (Mallam et al., 2004). In another study, photoreceptor transduction was achieved via the intravitreal route using an adenovirus vector pseudotyped with fiber from serotype 37 (Von Seggern et al., 2003). This is the only study demonstrating that the 80 nm Ad capsid can traverse the inner retina and reach the photoreceptors when administered via the intravitreal route. These data are unexpected and surprising given that AAV vectors with a diameter of 23 nm fail to transduce photoreceptors via the intravitreal route. Nonetheless, should the intravitreal route of photoreceptor transduction be confirmed, vector delivery for gene therapy of photoreceptor diseases would be significantly advanced, as intravitreal procedures are routine in the clinic and used currently for the administration of lucentis to patients suffering from age related macular degeneration. One study has recently shown that deletion of the integrin-binding RGD domain in Ad penton base allows Ad to infect photoreceptors, generally comparable to that seen with pseudotyped AAV vectors. While those studies were done with first generation Ad vectors that are hampered by a 7Kb cloning capacity, very strong levels of GFP was observed throughout the retina (Cashman et al., 2007) but transgene expression was still strongest in the RPE.
The Need for A Large Transgene Cloning Capacity
Any transgene being expressed in rod photoreceptors must be tightly regulated in terms of gene expression and cell specificity. It has been known for some time that five-fold expression of even normal gene products in rod photoreceptors leads to retinal degeneration (Olsson et al., 1992). In the case of the most commonly mutated gene in RP, namely rhodopsin, as little as 23% expression above normal levels of the wild type gene leads to retinal degeneration (Tan et al., 2001). Over-expression of peripherin/RDS delivered ectopically by an AAV vector to normal mice also led to retinal degeneration whereas AAV-delivered GFP did not (Sarra et al., 2001). Each of the above studies support the hypothesis that over-expression of normal transgenes in rod photoreceptors is deleterious. Examination of mice that are heterozygous for the rhodopsin null allele indicates that haploinsufficiency leads to disorganized photoreceptor outer segments and reduced electroretinograms and slow retinal degeneration (Liang et al., 2004). Hence, it would appear that there is a narrow range of gene expression that can be tolerated by rod photoreceptors to remain functional. One approach to control gene expression levels is the use of inducible systems that rely on an exogenous drug e.g. doxycycline to regulate the transgene-associated promoter (Goverdhana et al., 2005). While such approaches are very useful for studying temporally regulated expression of transgenes in animals including non-human primates (Stieger et al., 2006), it is unlikely that this approach will be a practical solution for RP patients. This is in part because such inducible systems are often immunogenic (Guiner et al., 2007; Lena et al., 2005) and because these approaches would rely on monitoring transgene expression on a regular basis in a non-invasive manner in order to gauge the amount of gene-inducer drug to administer. Practical and non-invasive methods to measure, for example, rhodopsin expression levels in RP patients are currently limited. One practical solution to this problem is the use of native gene regulatory elements to control gene expression. Such gene regulatory elements should theoretically participate in the natural feedback mechanisms present in the cell to regulate gene expression. Such gene control elements tend to be large relative to the limited cloning capacity of AAV-4.8Kb, Lv-7Kb or early generation Ad vectors-7Kb (Verma and Somia, 1997). In fact, once accommodation for the cDNA and polyadenylation sequences has been made, little room is left for large gene regulatory elements in any of these vectors.
Limited evidence exists for rod photoreceptor-specific transgene expression in the context of gene therapy. For example, short rhodopsin promoters of approximately 470 bp are known to express transgenes in both rods and cones (Glushakova et al., 2006). One study has reported rod specific transgene expression using a 235 bp opsin promoter (Khani et al., 2007). In the case of RP, it is plausible that ectopic and unregulated expression of genes such as rod opsin in the cones will lead to dominant negative phenotypes. Indeed, retinal degeneration in rd7 mice, a genetic model of enhanced Scone syndrome, is attributed to rod photoreceptors expressing both rod and cone specific genes (Corbo and Cepko, 2005). Rod or cone specific transgene expression has been achieved in the context of transgenic animals (Lem et al., 1991; Li et al., 2005) and the promoters utilized are typically larger than can be accommodated by currently available viral vectors. Despite the general need for cell-specific gene expression, there is a potential need for driving transgene expression in both rods and cones, such as for Xlinked RP (RP3) (Hong et al., 2000). While short rhodopsin kinase promoters (Khani et al., 2007) can express transgenes in both rods and cones, ultimately it may be preferable to use a promoter that is native to the transgene (for reasons discussed above). Recently, a 4.7 Kb rod opsin promoter was used to drive transgene expression in the context of an Ad vector but it remains to be determined whether even this length of promoter is rod specific in the context of gene therapy (Cashman et al., 2007). Furthermore, for some ocular diseases the cDNA of interest is simply too large to be accommodated in AAV vectors even without large gene regulatory elements. Examples include but are not limited to ABCR, CEP290 and myosin VIIa, each cDNA of which is greater than 4.8Kb. Clearly, in order to achieve rod photoreceptor-specific and regulated tansgene expression, vectors with larger cloning capacity than currently available are needed.
One potential solution to the problem of a limited cloning capacity is the approach of trans splicing of a gene split between multiple AAV vectors (Lai et al., 2005). At this time, trans splicing is relatively inefficient and requires gene sequence-specific modifications. Nonetheless, such approaches do warrant further investigation.
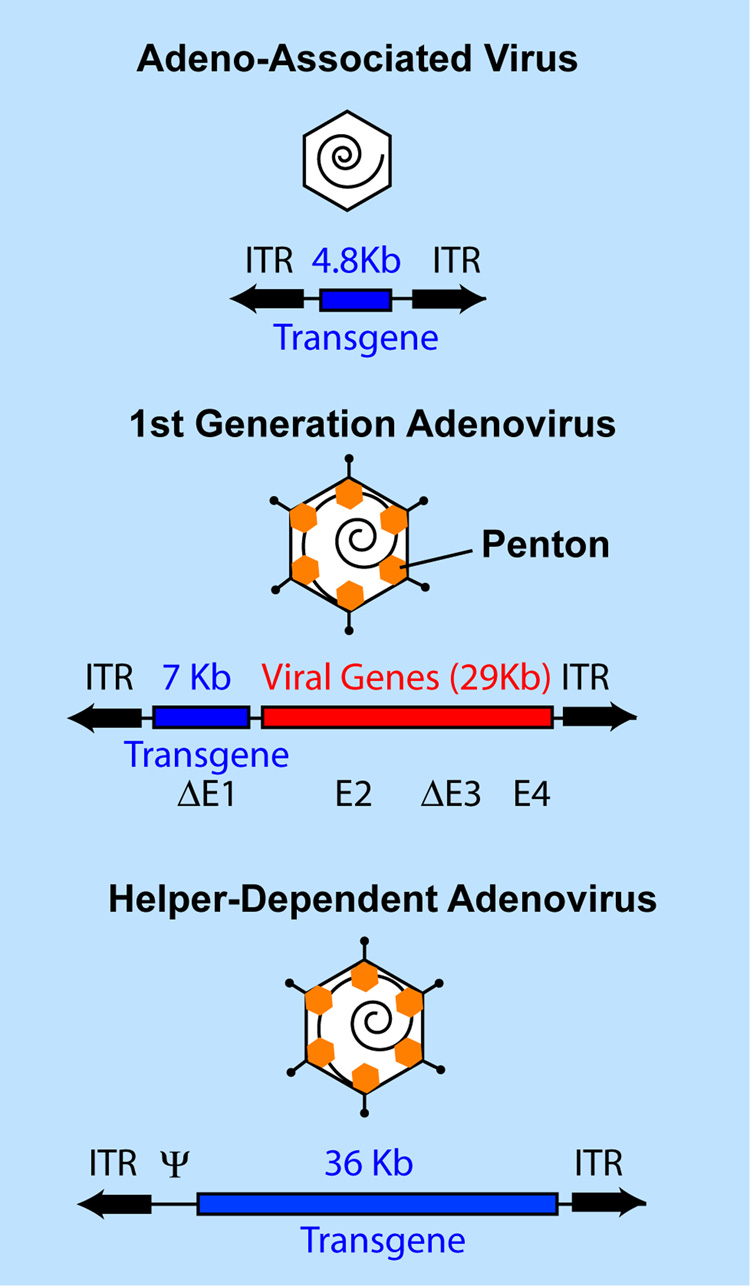
One potential solution to the barrier of a limited cloning capacity of AAV, Lv and Ad is the use of Helper-dependent Adenovirus (Hd-Ad) vectors that have a cloning capacity of 36 Kb (Parks et al., 1996). Such vectors are structurally analogous to recombinant AAV vectors since they contain only the transgene of interest, flanked by the Ad inverted terminal repeats (Figure 1). As will be discussed below, such vectors have many additional properties that render these vectors interesting for future gene therapy.
Figure 1.
Capsid and genomic structures of recombinant Adeno-associated Virus (AAV), 1st generation adenovirus and Helper-dependent adenovirus, emphasizing differences in cloning capacity for the recombinant transgene expression cassette. Whereas 1st generation Ad vectors have deletions in E1 and E3, second generation Ad vectors have deletions in E4, E2 etc. in addition to E1 and E3. ITR, inverted terminal repeat.
Vector Associated Immune Response
Long-term transgene expression is a requirement for treatment of slowly progressive degenerative diseases such as RP. If a vector is to achieve long-term transgene expression, it must evade the immune response. The immune response is usually directed towards viral proteins but may also be directed towards the transgene product if the immune system is naïve towards that protein-as could occur in for example rhodopsin-null patients carrying premature stop codons in the rhodopsin mRNA. Such patients will never have been exposed to wild-type rhodopsin epitopes and may generate immune responses against such proteins even in the immune privileged retina. This scenario may be even more relevant in the retina of patients with retinal degeneration that may be prone to leakage of retinal antigens due to disease. As gene therapy requires the delivery of the wild-type gene/ protein, we are limited at this time to addressing only the immune response associated with the vector. The general consensus in the retinal gene therapy field is that AAV is the least immunogenic of the viral gene transfer vectors available. General enthusiasm for AAV initially emanated from the observation that AAV could persistently express LacZ without activation of cytotoxic T lymphocytes (CTLs) after intramuscular injection into mice (Fisher et al., 1997; Kessler et al., 1996). Persistent transgene expression was subsequently observed in retinal gene transfer (reviewed in (Allocca et al., 2006)). However, there is significant evidence that AAV can cause an immune response that is limiting to transgene expression in humans even when similar experiments fail to generate such responses in dogs and mice. For example, while sustained factor IX expression was achieved over several years in dog and murine models of hemophilia B (Herzog et al., 1999; Snyder et al., 1999), transgene expression declined rapidly during clinical trials for hemophilia (Manno et al., 2006). No antibody to factor IX was found in those patients but T-cell responses to AAV capsid were documented, implicating the vector as the primary cause for loss of transgene expression. Partial immune privilege of the ocular environment might allow such studies to not be directly comparable to retinal gene therapy (Bennett, 2003). However, inflammatory responses, neutralizing antibodies and RPE atrophy following delivery of AAV into the subretinal space of non human primates has been documented (Bennett et al., 1999). T-cell responses to AAV capsid and local dropout of transgene expression (GFP) have also been documented after subretinal injection of AAV in non-human primates (Vandenberghe et al., 2007). Again, these data may need to be further considered prior to the initiation of additional clinical trials using AAV in ocular gene therapy.
Adenovirus vectors have received significant negative attention due to a serious adverse event resulting in the tragic death of a patient (Marshall, 1999). Several studies have demonstrated that a CTL mediated immune response limits transgene expression from Ad vectors in the ocular environment of mice (Hoffman et al., 1997; Reichel et al., 1998). Nonetheless, the only two published ocular gene therapy clinical trials performed to date have each used Ad as the gene therapy vector (Campochiaro et al., 2006; Chevez-Barrios et al., 2005). These studies warrant some discussion since it would appear that gene transfer in murine ocular tissues is not a good predictor of results achieved in humans.
In one ocular gene therapy clinical trial, twenty-eight patients with advanced age-related macular degeneration were injected intravitreally with a second-generation Ad vector (including deletions in E1, E3 as well as E4; Figure 1) expressing pigment epithelium-derived factor. The dose of Ad utilized ranged from 106 to 109.5 per patient and no dose-limiting toxicities were documented, although mild and transient intraocular inflammation that could be clinically managed was observed in 25% of patients (Campochiaro et al., 2006). In another ocular gene therapy clinical trial an early generation Ad expressing herpes simplex thymidine kinase was administered intravitreally to eight children suffering from retinoblastoma. Again, no dose limiting toxicities were observed even at concentrations as high as 1011 viral particles. Mild inflammation that could be managed was however documented (Chevez-Barrios et al., 2005).
Earlier studies performed in mice demonstrating ocular immune responses associated with Ad were performed with the so-called "first-generation" Ad vectors (Hoffman et al., 1997). These vectors retained approximately 80% of the adenovirus genome (Figure 1). Low-level expression of viral proteins from the major later promoter (MLP) was attributed to a CTL mediated immune response that led to loss of transgene expression. To address the barrier of vector associated immune responses, as mentioned above, novel adenovirus vector systems have been developed that do not generate a transgene-limiting immune response in animals. These vectors were initially referred to as ‘encapsidated adenovirus minichromosomes’, ‘gutted’ or ‘gutless’ vectors but they are now more generally referred to as Helper-dependent adenovirus (Hd-Ad) vectors (Parks et al., 1996). Hd-Ad vectors contain only the Ad inverted terminal repeats that are necessary for DNA replication and the Ad packaging signal necessary for encapsidation of the recombinant genome into preformed capsids. Hence, Hd-Ad vectors are devoid of all Ad genes and hence similar in structure to recombinant AAV but with a 36Kb cloning capacity instead of 4.8Kb (Figure 1). Importantly, the absence of viral genes should theoretically abrogate the low levels of gene expression from the MLP, products of which lead to activation of CTLs from first generation Ad vectors.
Hd-Ad vectors have been used to reduce the rate of photoreceptor degeneration in rd1 mice (Kumar-Singh and Farber, 1998). Initial versions of Hd-Ad vectors were contaminated with helper-virus that significantly contributed to immune-mediated clearance of transgene-containing cells. Those issues have been significantly resolved by improvements in vector design and purification techniques (Palmer and Ng, 2003). Moreover, since those earlier studies and other similar studies were performed in neonatal animals, they are not directly relevant to treatment of RP in humans, as was discussed above. Two recent studies using Hd-Ad vectors in the retina have had very significant success in terms of long term transgene expression, in one study lasting more than one year, the latest time point studied (Kreppel et al., 2002; Lamartina et al., 2007). Importantly, in non-ocular tissues, where Hd-Ad vectors have had the greatest application thus far, gene expression has been shown to persist for the 'life-time' of the animal (Kim et al., 2001). Moreover, Hd-Ad vectors persist in the brain and can be readministered even in a preimmunized animal (Barcia et al., 2007). Ironically, while the retinal gene therapy field generally focuses on the necessity of long-term transgene expression, the ability to readminister the vector makes available the opportunity to explore the possibility of designing safer Phase I/II clinical trials. This may be achieved by delivering therapeutic genes with the intention of short term transgene expression that will naturally be ‘turned off’, should adverse events begin to take place.
AAV and lentivirus vectors are popular in part because of the apparent lack of a significant immune response relative to first generation Ad vectors. However, the development of Hd-Ad vectors that persist in the retina in terms of transgene expression begs the question that the field of ocular gene therapy once again takes a close look at Ad, and more specifically at Hd-Ads. Significantly, a case is not being presented here for exclusive use of Hd-Ads instead of AAV or Lv, but rather that Hd-Ad may be an alternative vector to add to the ocular gene therapy vector toolbox.
Non Viral Vectors
Nobel Laureate Peter Medawar described a virus as a piece of nucleic acid surrounded by bad news (Oldstone, 1998). While the discussions above address the barriers to generating an optimal viral gene transfer vector, introduction of large amounts of foreign protein such as the viral coat proteins whether they be from Ad, AAV or Lv are likely to generate transient inflammatory responses that could exacerbate retinal disease and further compromise this delicate neuronal tissue. Hence, a preferred approach would include the use of a non-viral vector that could efficiently transduce retinal cells and achieve persistent transgene expression. Such vectors are likely not to be as restrictive in transgene cloning capacity and hence delivery of entire genomic fragments containing all native gene regulatory elements may be considered. Relative to viral vectors, non viral gene transfer to ocular or to any post mitotic cells in vivo is currently extremely inefficient and attempts to rescue models of retinal degeneration using such vectors have thus far not been successful. However, non viral reporter gene transfer rivaling levels achieved by viral gene transfer has been reported by two groups thus far, one approach using DNA encapsulating liposomes (Zhang et al., 2003; Zhu et al., 2002; Zhu et al., 2004) and another using a DNA-compacting peptide (Farjo et al., 2006). Interestingly, both these studies yielded data that needs to be somehow reconciled with previous studies describing the use of viral vectors in the retina. As discussed below, in both these approaches it appears that localization of the transgene product delivered by non-viral vectors is either different from that observed using viral vectors or there are as yet some unexplainable factors at play.
Zhang et al. intravenously delivered 85 nm immunoliposomes targeted to the human insulin receptor to achieve non-invasive transfer of genes across the blood retinal barrier of non-human primates (Zhang et al., 2003). These immunoliposomes encapsulated a lacZ gene regulated by a 2.2 Kb bovine rod opsin promoter. The study reported β-galactosidase activity in a large number of ocular tissues including RPE, photoreceptors, lens, iris, cornea, ganglion cells etc. Indeed, β-galactosidase activity was present to varying degrees in a large number of ocular cells and absent in non-ocular cells or non ocular tissues such as brain, heart and lung. While there has been considerable debate whether a 2.2Kb opsin promoter is rod specific, this is the only study describing organ specific gene expression from an otherwise previously considered photoreceptor specific promoter. These same investigators also utilized 85 nm immunoliposomes targeting the transferrin receptor to deliver genes across the blood retinal barrier in mice (Zhu et al., 2004). In this latter case the investigators localized β-galactosidase activity to the RPE, iris and ciliary body when using the same 2.2Kb bovine opsin promoter. Use of the GFAP promoter also led to unanticipated localization of β-galactosidase activity in the RPE (Zhu et al., 2002). While extremely promising, immunoliposome mediated gene delivery across the blood retinal or blood brain barrier appears to be highly technically demanding and has hence thus far not been widely replicated or used by other investigators.
Recently, Farjo et al. (Farjo et al., 2006) demonstrated non-viral gene transfer to photoreceptors at levels equivalent to or greater than viral gene transfer. In this study the investigators compacted DNA into nanoparticles using a peptide containing a cysteine residue coupled to poly ethylene glycol (PEG) followed by 30 lysines (CK30). Such nanoparticles appeared as rods with a minor diameter of less than 8nm, significantly less than the 25 nm diameter of the nuclear membrane pore (Dworetzky and Feldherr, 1988; Feldherr and Akin, 1991), allowing the nanoparticles to readily enter the nucleus (Liu et al., 2003). Following subretinal administration of CK30, GFP localization by direct fluorescence indicated that GFP was present extensively in the outer nuclear layer (photoreceptor cell bodies) and photoreceptor outer segments and absent from the inner photoreceptor segments (Farjo et al., 2006). This pattern of GFP localization contrasts with that observed using viral vectors, where typically the inner segments are also GFP-positive and more so than the outer segments (Cashman et al., 2007; Khani et al., 2007). Transgenic mice expressing GFP in photoreceptors also contain GFP in the inner segments (Nour et al., 2004). Furthermore, antibody mediated detection (immunofluorescence) of GFP indicated that CK30-compacted GFP delivered to the subretinal space resulted in GFP expression exclusively to the outer nuclear layer with no antibody signal in the outer or inner segments. Moreover, while immunofluorescence indicated a perinuclear location for recombinant GFP, direct fluorescence indicated a nuclear location for this same protein. Clearly this study would suggest that there are major differences in patterns and locations of transgene expression between viral vectors and some non-viral vectors in the retina. These differences between viral and non-viral gene delivery methods are intriguing and further investigation is necessary to reconcile these apparent contradictions. CK30 particles appear to be able to transduce non-dividing cells in culture (Liu et al., 2003), however, evidence that they do so in vivo in post mitotic tissues is extremely limited. Nonetheless, the studies performed in the retina are interesting given that CK30 nanoparticles are currently being utilized in clinical trials for treatment of cystic fibrosis (Konstan et al., 2004).
Another potential non-viral approach to deliver nucleic acids to the retina includes the use of peptides with cell penetrating properties such as peptide for ocular delivery (POD). Recently, Johnson et al. have shown that POD can rapidly translocate across cell plasma membranes in vitro and in vivo and deliver small and large molecules into the photoreceptors, RPE, ganglion cells etc. (Johnson et al., 2007). Delivery of large molecules such as DNA into the nucleus has thus far only been achieved in human embryonic retinoblasts in culture. The limitations in vivo appear to be the intact nuclear membrane, which disintegrates during mitosis in vitro to allow access of DNA to the nucleus. Similar to POD, other peptides tested in ocular tissues for potential of delivery of heterologous molecules include the protein transduction domain of human immunodeficiency virus (HIV) Tat (Barnett et al., 2006; Cashman et al., 2003; Schorderet et al., 2005) and Herpes Simplex Virus VP22 (Cashman et al., 2002; Kretz et al., 2003).
In addition to CK30, a variety of strategies to compact DNA and reduce its gyration radius to below the limiting diameter of the nuclear membrane pore have been employed. For example, use of poly(lactide-co-glycolide)/ PLGA that can form nanoparticles of approximately 140 nm has been shown to transduce RPE (Bejjani et al., 2005). How such particles cross the intact nuclear membrane pore of approximately 25nm is not yet understood. In non-ocular tissues the polymer polyethylenimine (PEI) has been one of the most successful non viral gene transfer vectors available (Boussif et al., 1995), but again, the efficiency has been too low to achieve therapeutic levels of gene expression in vivo. Other promising non-viral methods include in vivo electroporation (Chalberg et al., 2005) or electron avalanche transfection (Chalberg et al., 2006). Again, each of these methods, while promising, need significant improvements prior to their potential use in the clinic.
Finally, while non-viral gene delivery has significant advantages over viral gene delivery, the therapeutic DNA is still likely to face challenges from the immune system. Several sensors of single stranded and double stranded DNA are present as part of the innate immune response repertoires of cells. These sensors are located in the endosomes e.g. Toll-like receptor (TLR) 9 (Akira et al., 2006) as well as cytoplasmically, e.g. DAI (Takaoka et al., 2007). Engagement of TLRs initiates a signaling cascade of events that leads to induction of interferon-regulated factors and transcription factors such as NF-κB. This cascade ultimately leads to the production of type I interferon and proinflammatory cytokines. The methods employed by host DNA to protect itself from factors such as DAI are not yet understood. Caveats regarding these well known responses need to be considered, especially in the context of non-viral gene delivery to the retina.
A Significant Need for Further Development of Retinal Gene Transfer Vectors
Although the barriers referred to above do not exclude the use of currently available viral vectors in human ocular gene therapy and clinical trials are already under way, they do point to one thing: there is an urgent need for better and safer gene delivery vectors. This opinion is echoed in the 1995 Report and recommendations of the panel to assess the NIH investment in research on gene therapy by Stuart Orkin and Arno Motulsky (available on NIH website http://www.nih.gov/news/panelrep.html). The authors of this report concluded that a major obstacle to gene therapy is the "shortcomings in all current gene transfer vectors". Their recommendations are that "efforts need to be applied to improving vectors for gene delivery, enhancing and maintaining high level expression of genes transferred to somatic cells, achieving tissue-specific and regulated expression of transferred genes, and directing gene transfer to specific cell types." As discussed above, there is no evidence as yet that currently available vectors are ideal for gene transfer in humans. Despite this, significantly fewer resources are invested in vector development relative to attempted rescue of retinal disease models using compromised vectors that are readily available or simply using vectors that were developed by investigators with interests in non-ocular diseases with a goal of addressing only the needs of those other organ systems. Consideration of retinal physiology is critical for the development of vectors specifically for the retina, as was applied recently in the design of Ad vectors aimed at improving photoreceptor transduction by systematically reducing the interactions between Ad coat proteins and RPE (Cashman et al., 2007).
A case for inclusion of Hd-Ads in the ocular gene therapy vector toolbox
Helper dependent adenovirus vectors have a 36Kb cloning capacity, do not generally integrate and hence, theoretically, should not cause insertional mutagenesis (Kumar-Singh and Chamberlain, 1996; Kumar-Singh and Farber, 1998; Morsy et al., 1998; Parks et al., 1996). Hd-Ad vectors can be designed to integrate site-specifically (Wang and Lieber, 2006) or allow for long-term episomal transgene expression in the retina (Lamartina et al., 2007) and can be readministered if necessary (Barcia et al., 2007). Hd-Ad vectors have been found to be safe in non-human primates (Morral et al., 1999). At least two human ocular gene therapy trials have validated first generation Ad as being a safe vector (Campochiaro et al., 2006; Chevez-Barrios et al., 2005). Data from several hundred additional Ad-mediatd gene transfer clinical trials in non ocular tissues indicate that it is a relatively safe vector (Edelstein et al., 2007). However, Hd-Ad vectors transduce primarily the RPE and not the photoreceptors. First generation Ad5 vectors can be engineered to target photoreceptors (Cashman et al., 2007) but these do not persist in transgene expression. Ad5 capsid proteins are typically used during the construction of Hd-Ad vectors and hence Hd-Ad vectors based on Ad5 are potential candidates for gene delivery to the RPE (Kreppel et al., 2002). It is tempting to suggest that retargeting of Hd-Ad vectors to the photoreceptors where necessary can be simply achieved by using a helper virus derived from an Ad5 modified to target photoreceptors. Then, one may have in hand a vector with all of the properties of the ideal gene transfer vector for RPE as well as photoreceptor cells.
Concluding Remarks
Here I have described just some of the major vector-associated barriers faced by investigators in the retinal gene therapy field. As of now, there is no evidence that one vector is favorable over any other, although a case is presented here for further consideration of Ad as a potential vector for ocular gene therapy. The case for Ad is presented not to claim that Ad is superior to AAV or Lv but simply to reintroduce a vector that has not been generally discussed or well utilized by the ocular gene therapy community. Clearly, there are risks involved in any new endeavor and developing genetic therapy for substantially debilitating diseases such as blindness warrants pushing the envelope of risk. However, there is substantial data in the retinal gene therapy literature that needs to be reconciled in order to move the field forward. Much of this data indicates that preclinical success is focused on utilizing readily available vectors to rescue animal models at the expense of vector development that are relevant in the long term to human ocular gene therapy. General enthusiasm in the retinal gene therapy community that gene therapy for some rare retinal degenerations is imminent is well founded but whether the current studies will lead to rapid development of therapies for ocular gene therapy in general will require novel solutions to the barriers described herein.
ACKNOWLEDGEMENTS
Research in the RK-S laboratory is supported by the NIH/NEI, (EY014991 & EY013887), Foundation Fighting Blindness, The Ellison Foundation and grants to the Department of Ophthalmology at Tufts University School of Medicine from the Lions Eye Foundation and Research To Prevent Blindness. The author wishes to acknowledge useful feedback during the preparation of this article from Siobhan Cashman (Tufts University) and Andrea Amalfitano (Michigan State University).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Note added in proof: Recently, Phase I data for two of the three LCA clinical trials has been published (Bainbridge et al., 2008; Maguire et al., 2008). Also, a recent study suggests that the limited 4.8Kb capacity of AAV2 can be exceeded by AAV5 (Allocca et al., 2008).
REFERENCES
- Acland GM, Aguirre GD, Ray J, Zhang Q, Aleman TS, Cideciyan AV, Pearce-Kelling SE, Anand V, Zeng Y, Maguire AM, et al. Gene therapy restores vision in a canine model of childhood blindness. Nat Genet. 2001;28:92–95. doi: 10.1038/ng0501-92. [DOI] [PubMed] [Google Scholar]
- Acsadi G, Jani A, Massie B, Simoneau M, Holland P, Blaschuk K, Karpati G. A differential efficiency of adenovirus-mediated in vivo gene transfer into skeletal muscle cells of different maturity. HumMolGenet. 1994;3:579–584. doi: 10.1093/hmg/3.4.579. [DOI] [PubMed] [Google Scholar]
- Akira S, Uematsu S, Takeuchi O. Pathogen recognition and innate immunity. Cell. 2006;124:783–801. doi: 10.1016/j.cell.2006.02.015. [DOI] [PubMed] [Google Scholar]
- Ali RR, Reichel MB, Thrasher AJ, Levinsky RJ, Kinnon C, Kanuga N, Hunt DM, Bhattacharya SS. Gene transfer into the mouse retina mediated by an adeno-associated viral vector. Hum Mol Genet. 1996;5:591–594. doi: 10.1093/hmg/5.5.591. [DOI] [PubMed] [Google Scholar]
- Ali RR, Sarra GM, Stephens C, Alwis MD, Bainbridge JW, Munro PM, Fauser S, Reichel MB, Kinnon C, Hunt DM, et al. Restoration of photoreceptor ultrastructure and function in retinal degeneration slow mice by gene therapy. Nat Genet. 2000;25:306–310. doi: 10.1038/77068. [see comments] [DOI] [PubMed] [Google Scholar]
- Allocca M, Mussolino C, Garcia-Hoyos M, Sanges D, Iodice C, Petrillo M, Vandenberghe LH, Wilson JM, Marigo V, Surace EM, Auricchio A. Novel adeno-associated virus serotypes efficiently transduce murine photoreceptors. J Virol. 2007;81:11372–11380. doi: 10.1128/JVI.01327-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allocca M, Tessitore A, Cotugno G, Auricchio A. AAV-mediated gene transfer for retinal diseases. Expert Opin Biol Ther. 2006;6:1279–1294. doi: 10.1517/14712598.6.12.1279. [DOI] [PubMed] [Google Scholar]
- Auricchio A, Rolling F. Adeno-associated viral vectors for retinal gene transfer and treatment of retinal diseases. Curr Gene Ther. 2005;5:339–348. doi: 10.2174/1566523054065020. [DOI] [PubMed] [Google Scholar]
- Bainbridge JW, Stephens C, Parsley K, Demaison C, Halfyard A, Thrasher AJ, Ali RR. In vivo gene transfer to the mouse eye using an HIV-based lentiviral vector; efficient long-term transduction of corneal endothelium and retinal pigment epithelium. Gene Ther. 2001;8:1665–1668. doi: 10.1038/sj.gt.3301574. [DOI] [PubMed] [Google Scholar]
- Bainbridge JW, Tan MH, Ali RR. Gene therapy progress and prospects: the eye. Gene Ther. 2006;13:1191–1197. doi: 10.1038/sj.gt.3302812. [DOI] [PubMed] [Google Scholar]
- Balaggan KS, Binley K, Esapa M, Iqball S, Askham Z, Kan O, Tschernutter M, Bainbridge JW, Naylor S, Ali RR. Stable and efficient intraocular gene transfer using pseudotyped EIAV lentiviral vectors. J Gene Med. 2006;8:275–285. doi: 10.1002/jgm.845. [DOI] [PubMed] [Google Scholar]
- Barcia C, Jimenez-Dalmaroni M, Kroeger KM, Puntel M, Rapaport AJ, Larocque D, King GD, Johnson SA, Liu C, Xiong W, et al. One-year Expression From High-capacity Adenoviral Vectors in the Brains of Animals With Preexisting Anti-adenoviral Immunity: Clinical Implications. Mol Ther. 2007 doi: 10.1038/sj.mt.6300305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnett EM, Elangovan B, Bullok KE, Piwnica-Worms D. Selective cell uptake of modified Tat peptide-fluorophore conjugates in rat retina in ex vivo and in vivo models. Invest Ophthalmol Vis Sci. 2006;47:2589–2595. doi: 10.1167/iovs.05-1470. [DOI] [PubMed] [Google Scholar]
- Bejjani RA, BenEzra D, Cohen H, Rieger J, Andrieu C, Jeanny JC, Gollomb G, Behar-Cohen FF. Nanoparticles for gene delivery to retinal pigment epithelial cells. Mol Vis. 2005;11:124–132. [PubMed] [Google Scholar]
- Bemelmans AP, Bonnel S, Houhou L, Dufour N, Nandrot E, Helmlinger D, Sarkis C, Abitbol M, Mallet J. Retinal cell type expression specificity of HIV-1-derived gene transfer vectors upon subretinal injection in the adult rat: influence of pseudotyping and promoter. J Gene Med. 2005;7:1367–1374. doi: 10.1002/jgm.788. [DOI] [PubMed] [Google Scholar]
- Bennett J. Immune response following intraocular delivery of recombinant viral vectors. Gene Ther. 2003;10:977–982. doi: 10.1038/sj.gt.3302030. [DOI] [PubMed] [Google Scholar]
- Bennett J, Maguire AM, Cideciyan AV, Schnell M, Glover E, Anand V, Aleman TS, Chirmule N, Gupta AR, Huang Y, et al. Stable transgene expression in rod photoreceptors after recombinant adeno-associated virus-mediated gene transfer to monkey retina. Proc Natl Acad Sci U S A. 1999;96:9920–9925. doi: 10.1073/pnas.96.17.9920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bennett J, Tanabe T, Sun D, Zeng Y, Kjeldbye H, Gouras P, Maguire AM. Photoreceptor cell rescue in retinal degeneration (rd) mice by in vivo gene therapy. Nat Med. 1996;2:649–654. doi: 10.1038/nm0696-649. [DOI] [PubMed] [Google Scholar]
- Boussif O, Lezoualc'h F, Zanta MA, Mergny MD, Scherman D, Demeneix B, Behr JP. A versatile vector for gene and oligonucleotide transfer into cells in culture and in vivo: polyethylenimine. Proc Natl Acad Sci U S A. 1995;92:7297–7301. doi: 10.1073/pnas.92.16.7297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campochiaro PA, Dong Nguyen Q, Mahmood Shah S, Klein ML, Holz E, Frank RN, Saperstein DA, Gupta A, Stout JT, Macko J, Dibartolomeo R. Adenoviral Vector-Delivered Pigment Epithelium-Derived Factor for Neovascular Age-Related Macular Degeneration: Results of a Phase I Clinical Trial. Hum Gene Ther. 2006;17:167–176. doi: 10.1089/hum.2006.17.167. [DOI] [PubMed] [Google Scholar]
- Cashman SM, McCullough L, Kumar-Singh R. Improved Retinal Transduction In Vivo and Photoreceptor-specific Transgene Expression Using Adenovirus Vectors With Modified Penton Base. Mol Ther. 2007;15:1640–1646. doi: 10.1038/sj.mt.6300203. [DOI] [PubMed] [Google Scholar]
- Cashman SM, Morris DJ, Kumar-Singh R. Evidence of protein transduction but not intercellular transport by proteins fused to HIV tat in retinal cell culture and in vivo. Mol Ther. 2003;8:130–142. doi: 10.1016/s1525-0016(03)00131-x. [DOI] [PubMed] [Google Scholar]
- Cashman SM, Sadowski SL, Morris DJ, Frederick J, Kumar-Singh R. Intercellular Trafficking of Adenovirus-Delivered HSV VP22 from the Retinal Pigment Epithelium to the Photoreceptors-Implications for Gene Therapy. Mol Ther. 2002;6:813–823. doi: 10.1006/mthe.2002.0806. [DOI] [PubMed] [Google Scholar]
- Cavazzana-Calvo M, Hacein-Bey S, de Saint Basile G, Gross F, Yvon E, Nusbaum P, Selz F, Hue C, Certain S, Casanova JL, et al. Gene therapy of human severe combined immunodeficiency (SCID)-X1 disease. Science. 2000;288:669–672. doi: 10.1126/science.288.5466.669. [see comments] [DOI] [PubMed] [Google Scholar]
- Cepko CL, Austin CP, Yang X, Alexiades M, Ezzeddine D. Cell fate determination in the vertebrate retina. Proc Natl Acad Sci U S A. 1996;93:589–595. doi: 10.1073/pnas.93.2.589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chalberg TW, Genise HL, Vollrath D, Calos MP. phiC31 integrase confers genomic integration and long-term transgene expression in rat retina. Invest Ophthalmol Vis Sci. 2005;46:2140–2146. doi: 10.1167/iovs.04-1252. [DOI] [PubMed] [Google Scholar]
- Chalberg TW, Vankov A, Molnar FE, Butterwick AF, Huie P, Calos MP, Palanker DV. Gene transfer to rabbit retina with electron avalanche transfection. Invest Ophthalmol Vis Sci. 2006;47:4083–4090. doi: 10.1167/iovs.06-0092. [DOI] [PubMed] [Google Scholar]
- Chang B, Hawes NL, Hurd RE, Wang J, Howell D, Davisson MT, Roderick TH, Nusinowitz S, Heckenlively JR. Mouse models of ocular diseases. Vis Neurosci. 2005;22:587–593. doi: 10.1017/S0952523805225075. [DOI] [PubMed] [Google Scholar]
- Chevez-Barrios P, Chintagumpala M, Mieler W, Paysse E, Boniuk M, Kozinetz C, Hurwitz MY, Hurwitz RL. Response of retinoblastoma with vitreous tumor seeding to adenovirus-mediated delivery of thymidine kinase followed by ganciclovir. J Clin Oncol. 2005;23:7927–7935. doi: 10.1200/JCO.2004.00.1883. [DOI] [PubMed] [Google Scholar]
- Corbo JC, Cepko CL. A hybrid photoreceptor expressing both rod and cone genes in a mouse model of enhanced S-cone syndrome. PLoS Genet. 2005;1:11. doi: 10.1371/journal.pgen.0010011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Donsante A, Miller DG, Li Y, Vogler C, Brunt EM, Russell DW, Sands MS. AAV vector integration sites in mouse hepatocellular carcinoma. Science. 2007;317:477. doi: 10.1126/science.1142658. [DOI] [PubMed] [Google Scholar]
- Donsante A, Vogler C, Muzyczka N, Crawford JM, Barker J, Flotte T, Campbell-Thompson M, Daly T, Sands MS. Observed incidence of tumorigenesis in long-term rodent studies of rAAV vectors. Gene Ther. 2001;8:1343–1346. doi: 10.1038/sj.gt.3301541. [DOI] [PubMed] [Google Scholar]
- Dworetzky SI, Feldherr CM. Translocation of RNA-coated gold particles through the nuclear pores of oocytes. J Cell Biol. 1988;106:575–584. doi: 10.1083/jcb.106.3.575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edelstein ML, Abedi MR, Wixon J. Gene therapy clinical trials worldwide to 2007--an update. J Gene Med. 2007;9:833–842. doi: 10.1002/jgm.1100. [DOI] [PubMed] [Google Scholar]
- Farjo R, Skaggs J, Quiambao AB, Cooper MJ, Naash MI. Efficient non-viral ocular gene transfer with compacted DNA nanoparticles. PLoS ONE. 2006;1:38. doi: 10.1371/journal.pone.0000038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feldherr CM, Akin D. Signal-mediated nuclear transport in proliferating and growth-arrested BALB/c 3T3 cells. J Cell Biol. 1991;115:933–939. doi: 10.1083/jcb.115.4.933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fisher KJ, Jooss K, Alston J, Yang Y, Haecker SE, High K, Pathak R, Raper SE, Wilson JM. Recombinant adeno-associated virus for muscle directed gene therapy. NatMed. 1997;3:306–312. doi: 10.1038/nm0397-306. [DOI] [PubMed] [Google Scholar]
- Georges P, Madigan MC, Provis JM. Apoptosis during development of the human retina: relationship to foveal development and retinal synaptogenesis. J Comp Neurol. 1999;413:198–208. [PubMed] [Google Scholar]
- Glushakova LG, Timmers AM, Issa TM, Cortez NG, Pang J, Teusner JT, Hauswirth WW. Does recombinant adeno-associated virus-vectored proximal region of mouse rhodopsin promoter support only rod-type specific expression in vivo? Mol Vis. 2006;12:298–309. [PubMed] [Google Scholar]
- Goverdhana S, Puntel M, Xiong W, Zirger JM, Barcia C, Curtin JF, Soffer EB, Mondkar S, King GD, Hu J, et al. Regulatable gene expression systems for gene therapy applications: progress and future challenges. Mol Ther. 2005;12:189–211. doi: 10.1016/j.ymthe.2005.03.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guiner CL, Stieger K, Snyder RO, Rolling F, Moullier P. Immune responses to gene product of inducible promoters. Curr Gene Ther. 2007;7:334–346. doi: 10.2174/156652307782151461. [DOI] [PubMed] [Google Scholar]
- Hartong DT, Berson EL, Dryja TP. Retinitis pigmentosa. Lancet. 2006;368:1795–1809. doi: 10.1016/S0140-6736(06)69740-7. [DOI] [PubMed] [Google Scholar]
- Herzog RW, Yang EY, Couto LB, Hagstrom JN, Elwell D, Fields PA, Burton M, Bellinger DA, Read MS, Brinkhous KM, et al. Long-term correction of canine hemophilia B by gene transfer of blood coagulation factor IX mediated by adeno-associated viral vector. Nat Med. 1999;5:56–63. doi: 10.1038/4743. [DOI] [PubMed] [Google Scholar]
- Hoffman LM, Maguire AM, Bennett J. Cell-mediated immune responsez and stability of intraocular transgene expression after adenovirus-mediated delivery. InvestOphthalmolVisSci. 1997;38:2224–2233. [PubMed] [Google Scholar]
- Hong DH, Pawlyk BS, Shang J, Sandberg MA, Berson EL, Li T. A retinitis pigmentosa GTPase regulator (RPGR)-deficient mouse model for X-linked retinitis pigmentosa (RP3) Proc Natl Acad Sci U S A. 2000;97:3649–3654. doi: 10.1073/pnas.060037497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson LN, Cashman SM, Kumar-Singh R. Cell-penetrating Peptide for Enhanced Delivery of Nucleic Acids and Drugs to Ocular Tissues Including Retina and Cornea. Mol Ther. 2007 doi: 10.1038/sj.mt.6300324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jomary C, Vincent KA, Grist J, Neal MJ, Jones SE. Rescue of photoreceptor function by AAV-mediated gene transfer in a mouse model of inherited retinal degeneration. Gene Ther. 1997;4:683–690. doi: 10.1038/sj.gt.3300440. [DOI] [PubMed] [Google Scholar]
- Kaiser J. Gene therapy. Seeking the cause of induced leukemias in X-SCID trial. Science. 2003;299:495. doi: 10.1126/science.299.5606.495. [DOI] [PubMed] [Google Scholar]
- Kaiser J. Clinical trials. Gene transfer an unlikely contributor to patient's death. Science. 2007;318:1535. doi: 10.1126/science.318.5856.1535. [DOI] [PubMed] [Google Scholar]
- Kay MA. AAV vectors and tumorigenicity. Nat Biotechnol. 2007;25:1111–1113. doi: 10.1038/nbt1007-1111. [DOI] [PubMed] [Google Scholar]
- Kessler PD, Podsakoff GM, Chen X, McQuiston SA, Colosi PC, Matelis LA, Kurtzman GJ, Byrne BJ. Gene delivery to skeletal muscle results in sustained expression and systemic delivery of a therapeutic protein. Proc Natl Acad Sci U S A. 1996;93:14082–14087. doi: 10.1073/pnas.93.24.14082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khani SC, Pawlyk BS, Bulgakov OV, Kasperek E, Young JE, Adamian M, Sun X, Smith AJ, Ali RR, Li T. AAV-mediated expression targeting of rod and cone photoreceptors with a human rhodopsin kinase promoter. Invest Ophthalmol Vis Sci. 2007;48:3954–3961. doi: 10.1167/iovs.07-0257. [DOI] [PubMed] [Google Scholar]
- Kim IH, Jozkowicz A, Piedra PA, Oka K, Chan L. Lifetime correction of genetic deficiency in mice with a single injection of helper-dependent adenoviral vector. Proc Natl Acad Sci U S A. 2001;98:13282–13287. doi: 10.1073/pnas.241506298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konstan MW, Davis PB, Wagener JS, Hilliard KA, Stern RC, Milgram LJ, Kowalczyk TH, Hyatt SL, Fink TL, Gedeon CR, et al. Compacted DNA nanoparticles administered to the nasal mucosa of cystic fibrosis subjects are safe and demonstrate partial to complete cystic fibrosis transmembrane regulator reconstitution. Hum Gene Ther. 2004;15:1255–1269. doi: 10.1089/hum.2004.15.1255. [DOI] [PubMed] [Google Scholar]
- Kotin RM, Siniscalco M, Samulski RJ, Zhu XD, Hunter L, Laughlin CA, McLaughlin S, Muzyczka N, Rocchi M, Berns KI. Site-specific integration by adeno-associated virus. Proc Natl Acad Sci U S A. 1990;87:2211–2215. doi: 10.1073/pnas.87.6.2211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kreppel F, Luther TT, Semkova I, Schraermeyer U, Kochanek S. Long-term transgene expression in the RPE after gene transfer with a high-capacity adenoviral vector. Invest Ophthalmol Vis Sci. 2002;43:1965–1970. [PubMed] [Google Scholar]
- Kretz A, Wybranietz WA, Hermening S, Lauer UM, Isenmann S. HSV-1 VP22 augments adenoviral gene transfer to CNS neurons in the retina and striatum in vivo. Mol Ther. 2003;7:659–669. doi: 10.1016/s1525-0016(03)00062-5. [DOI] [PubMed] [Google Scholar]
- Kumar-Singh R, Chamberlain JS. Encapsidated adenovirus minichromosomes allow delivery and expression of a 14 kb dystrophin cDNA to muscle cells. HumMolGenet. 1996;5:913–921. doi: 10.1093/hmg/5.7.913. [DOI] [PubMed] [Google Scholar]
- Kumar-Singh R, Farber DB. Encapsidated adenovirus mini-chromosome-mediated delivery of genes to the retina: application to the rescue of photoreceptor degeneration. HumMolGenet. 1998;7:1893–1900. doi: 10.1093/hmg/7.12.1893. [DOI] [PubMed] [Google Scholar]
- Lai Y, Yue Y, Liu M, Ghosh A, Engelhardt JF, Chamberlain JS, Duan D. Efficient in vivo gene expression by trans-splicing adeno-associated viral vectors. Nat Biotechnol. 2005;23:1435–1439. doi: 10.1038/nbt1153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamartina S, Cimino M, Roscilli G, Dammassa E, Lazzaro D, Rota R, Ciliberto G, Toniatti C. Helper-dependent adenovirus for the gene therapy of proliferative retinopathies: stable gene transfer, regulated gene expression and therapeutic efficacy. J Gene Med. 2007;9:862–874. doi: 10.1002/jgm.1083. [DOI] [PubMed] [Google Scholar]
- Le Meur G, Stieger K, Smith AJ, Weber M, Deschamps JY, Nivard D, Mendes-Madeira A, Provost N, Pereon Y, Cherel Y, et al. Restoration of vision in RPE65-deficient Briard dogs using an AAV serotype 4 vector that specifically targets the retinal pigmented epithelium. Gene Ther. 2007;14:292–303. doi: 10.1038/sj.gt.3302861. [DOI] [PubMed] [Google Scholar]
- Le Meur G, Weber M, Pereon Y, Mendes-Madeira A, Nivard D, Deschamps JY, Moullier P, Rolling F. Postsurgical assessment and long-term safety of recombinant adeno-associated virus-mediated gene transfer into the retinas of dogs and primates. Arch Ophthalmol. 2005;123:500–506. doi: 10.1001/archopht.123.4.500. [DOI] [PubMed] [Google Scholar]
- Lem J, Applebury ML, Falk JD, Flannery JG, Simon MI. Tissue-specific and developmental regulation of rod opsin chimeric genes in transgenic mice. Neuron. 1991;6:201–210. doi: 10.1016/0896-6273(91)90356-5. [DOI] [PubMed] [Google Scholar]
- Lena AM, Giannetti P, Sporeno E, Ciliberto G, Savino R. Immune responses against tetracycline-dependent transactivators affect long-term expression of mouse erythropoietin delivered by a helper-dependent adenoviral vector. J Gene Med. 2005;7:1086–1096. doi: 10.1002/jgm.758. [DOI] [PubMed] [Google Scholar]
- Li S, Chen D, Sauve Y, McCandless J, Chen YJ, Chen CK. Rhodopsin-iCre transgenic mouse line for Cre-mediated rod-specific gene targeting. Genesis. 2005;41:73–80. doi: 10.1002/gene.20097. [DOI] [PubMed] [Google Scholar]
- Li Z, Dullmann J, Schiedlmeier B, Schmidt M, von Kalle C, Meyer J, Forster M, Stocking C, Wahlers A, Frank O, et al. Murine leukemia induced by retroviral gene marking. Science. 2002;296:497. doi: 10.1126/science.1068893. [DOI] [PubMed] [Google Scholar]
- Liang Y, Fotiadis D, Maeda T, Maeda A, Modzelewska A, Filipek S, Saperstein DA, Engel A, Palczewski K. Rhodopsin signaling and organization in heterozygote rhodopsin knockout mice. J Biol Chem. 2004;279:48189–48196. doi: 10.1074/jbc.M408362200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linberg KA, Fisher SK. A burst of differentiation in the outer posterior retina of the eleven-week human fetus: an ultrastructural study. Vis Neurosci. 1990;5:43–60. doi: 10.1017/s0952523800000067. [DOI] [PubMed] [Google Scholar]
- Liu G, Li D, Pasumarthy MK, Kowalczyk TH, Gedeon CR, Hyatt SL, Payne JM, Miller TJ, Brunovskis P, Fink TL, et al. Nanoparticles of compacted DNA transfect postmitotic cells. J Biol Chem. 2003;278:32578–32586. doi: 10.1074/jbc.M305776200. [DOI] [PubMed] [Google Scholar]
- Mallam JN, Hurwitz MY, Mahoney T, Chevez-Barrios P, Hurwitz RL. Efficient gene transfer into retinal cells using adenoviral vectors: dependence on receptor expression. Invest Ophthalmol Vis Sci. 2004;45:1680–1687. doi: 10.1167/iovs.03-0730. [DOI] [PubMed] [Google Scholar]
- Manno CS, Pierce GF, Arruda VR, Glader B, Ragni M, Rasko JJ, Ozelo MC, Hoots K, Blatt P, Konkle B, et al. Successful transduction of liver in hemophilia by AAV-Factor IX and limitations imposed by the host immune response. Nat Med. 2006;12:342–347. doi: 10.1038/nm1358. [DOI] [PubMed] [Google Scholar]
- Marshall E. Gene therapy death prompts review of adenovirus vector. Science. 1999;286:2244–2245. doi: 10.1126/science.286.5448.2244. [DOI] [PubMed] [Google Scholar]
- Marshall E. Gene therapy. Viral vectors still pack surprises. Science. 2001;294:1640. doi: 10.1126/science.294.5547.1640. [DOI] [PubMed] [Google Scholar]
- Miyoshi H, Takahashi M, Gage FH, Verma IM. Stable and efficient gene transfer into the retina using an HIV-based lentiviral vector. Proc Natl Acad Sci U S A. 1997;94:10319–10323. doi: 10.1073/pnas.94.19.10319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morral N, O'Neal W, Rice K, Leland M, Kaplan J, Piedra PA, Zhou H, Parks RJ, Velji R, Aguilar-Cordova E, et al. Administration of helper-dependent adenoviral vectors and sequential delivery of different vector serotype for long-term liver-directed gene transfer in baboons. Proc Natl Acad Sci U S A. 1999;96:12816–12821. doi: 10.1073/pnas.96.22.12816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morsy MA, Gu M, Motzel S, Zhao J, Lin J, Su Q, Allen H, Franlin L, Parks RJ, Graham FL, et al. An adenoviral vector deleted for all viral coding sequences results in enhanced safety and extended expression of a leptin transgene. Proc Natl Acad Sci U S A. 1998;95:7866–7871. doi: 10.1073/pnas.95.14.7866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakai H, Wu X, Fuess S, Storm TA, Munroe D, Montini E, Burgess SM, Grompe M, Kay MA. Large-scale molecular characterization of adeno-associated virus vector integration in mouse liver. J Virol. 2005;79:3606–3614. doi: 10.1128/JVI.79.6.3606-3614.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Narfstrom K, Katz ML, Bragadottir R, Seeliger M, Boulanger A, Redmond TM, Caro L, Lai CM, Rakoczy PE. Functional and structural recovery of the retina after gene therapy in the RPE65 null mutation dog. Invest Ophthalmol Vis Sci. 2003;44:1663–1672. doi: 10.1167/iovs.02-0595. [DOI] [PubMed] [Google Scholar]
- Neumann R, Doerfler W. Integration of adenovirus type 2 DNA at a limited number of cellular sites in productively infected cells. J Virol. 1981;37:887–892. doi: 10.1128/jvi.37.3.887-892.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nicoud M, Kong J, Iqball S, Kan O, Naylor S, Gouras P, Allikmets R, Binley K. Development of photoreceptor-specific promoters and their utility to investigate EIAV lentiviral vector mediated gene transfer to photoreceptors. J Gene Med. 2007;9(12):1015–1023. doi: 10.1002/jgm.1115. [DOI] [PubMed] [Google Scholar]
- Nour M, Quiambao AB, Al-Ubaidi MR, Naash MI. Absence of functional and structural abnormalities associated with expression of EGFP in the retina. Invest Ophthalmol Vis Sci. 2004;45:15–22. doi: 10.1167/iovs.03-0663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oldstone MBA. Introduction to the principles of virology. In: Oldstone MBA, editor. Viruses, plagues, & history. New York, NY: Oxford University Press; 1998. pp. 8–15. [Google Scholar]
- Olsson JE, Gordon JW, Pawlyk BS, Roof D, Hayes A, Molday RS, Mukai S, Cowley GS, Berson EL, Dryja TP. Transgenic mice with a rhodopsin mutation (Pro23His): a mouse model of autosomal dominant retinitis pigmentosa. Neuron. 1992;9:815–830. doi: 10.1016/0896-6273(92)90236-7. [DOI] [PubMed] [Google Scholar]
- Palmer D, Ng P. Improved system for helper-dependent adenoviral vector production. Mol Ther. 2003;8:846–852. doi: 10.1016/j.ymthe.2003.08.014. [DOI] [PubMed] [Google Scholar]
- Pang J, Cheng M, Haire SE, Barker E, Planelles V, Blanks JC. Efficiency of lentiviral transduction during development in normal and rd mice. Mol Vis. 2006;12:756–767. [PubMed] [Google Scholar]
- Pang J, Cheng M, Stevenson D, Trousdale MD, Dorey CK, Blanks JC. Adenoviral-mediated gene transfer to retinal explants during development and degeneration. Exp Eye Res. 2004;79:189–201. doi: 10.1016/j.exer.2004.03.010. [DOI] [PubMed] [Google Scholar]
- Parks RJ, Chen L, Anton M, Sankar U, Rudnicki MA, Graham FL. A helper-dependent adenovirus vector system: removal of helper virus by Cre-mediated excision of the viral packaging signal. Proc Natl Acad Sci U S A. 1996;93:13565–13570. doi: 10.1073/pnas.93.24.13565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pawlyk BS, Smith AJ, Buch PK, Adamian M, Hong DH, Sandberg MA, Ali RR, Li T. Gene replacement therapy rescues photoreceptor degeneration in a murine model of Leber congenital amaurosis lacking RPGRIP. Invest Ophthalmol Vis Sci. 2005;46:3039–3045. doi: 10.1167/iovs.05-0371. [DOI] [PubMed] [Google Scholar]
- Penfold PL, Provis JM. Cell death in the development of the human retina: phagocytosis of pyknotic and apoptotic bodies by retinal cells. Graefes Arch Clin Exp Ophthalmol. 1986;224:549–553. doi: 10.1007/BF02154744. [DOI] [PubMed] [Google Scholar]
- Ponnazhagan S, Erikson D, Kearns WG, Zhou SZ, Nahreini P, Wang XS, Srivastava A. Lack of site-specific integration of the recombinant adeno-associated virus 2 genomes in human cells. Hum Gene Ther. 1997;8:275–284. doi: 10.1089/hum.1997.8.3-275. [DOI] [PubMed] [Google Scholar]
- Price J, Turner D, Cepko C. Lineage analysis in the vertebrate nervous system by retrovirus-mediated gene transfer. Proc Natl Acad Sci U S A. 1987;84:156–160. doi: 10.1073/pnas.84.1.156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reh TA. Neurobiology: right timing for retina repair. Nature. 2006;444:156–157. doi: 10.1038/444156a. [DOI] [PubMed] [Google Scholar]
- Reichel MB, Ali RR, Thrasher AJ, Hunt DM, Bhattacharya SS, Baker D. Immune responses limit adenovirally mediated gene expression in the adult mouse eye. Gene Ther. 1998;5:1038–1046. doi: 10.1038/sj.gt.3300691. [DOI] [PubMed] [Google Scholar]
- Rolling F. Recombinant AAV-mediated gene transfer to the retina: gene therapy perspectives. Gene Ther. 2004;11 Suppl 1:S26–S32. doi: 10.1038/sj.gt.3302366. [DOI] [PubMed] [Google Scholar]
- Russell WC. Update on adenovirus and its vectors. J Gen Virol. 2000;81:2573–2604. doi: 10.1099/0022-1317-81-11-2573. [DOI] [PubMed] [Google Scholar]
- Sarra GM, Stephens C, de Alwis M, Bainbridgex JW, Smith AJ, Thrasher AJ, Ali RR. Gene replacement therapy in the retinal degeneration slow (rds) mouse: the effect on retinal degeneration following partial transduction of the retina. Hum Mol Genet. 2001;10:2353–2361. doi: 10.1093/hmg/10.21.2353. [DOI] [PubMed] [Google Scholar]
- Schnepp BC, Jensen RL, Chen CL, Johnson PR, Clark KR. Characterization of adeno-associated virus genomes isolated from human tissues. J Virol. 2005;79:14793–14803. doi: 10.1128/JVI.79.23.14793-14803.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schorderet DF, Manzi V, Canola K, Bonny C, Arsenijevic Y, Munier FL, Maurer F. D-TAT transporter as an ocular peptide delivery system. Clin Experiment Ophthalmol. 2005;33:628–635. doi: 10.1111/j.1442-9071.2005.01108.x. [DOI] [PubMed] [Google Scholar]
- Snyder RO, Im DS, Muzyczka N. Evidence for covalent attachment of the adeno-associated virus (AAV) rep protein to the ends of the AAV genome. J Virol. 1990;64:6204–6213. doi: 10.1128/jvi.64.12.6204-6213.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snyder RO, Miao C, Meuse L, Tubb J, Donahue BA, Lin HF, Stafford DW, Patel S, Thompson AR, Nichols T, et al. Correction of hemophilia B in canine and murine models using recombinant adeno-associated viral vectors. Nat Med. 1999;5:64–70. doi: 10.1038/4751. [DOI] [PubMed] [Google Scholar]
- Stieger K, Le Meur G, Lasne F, Weber M, Deschamps JY, Nivard D, Mendes-Madeira A, Provost N, Martin L, Moullier P, Rolling F. Long-term doxycycline-regulated transgene expression in the retina of nonhuman primates following subretinal injection of recombinant AAV vectors. Mol Ther. 2006;13:967–975. doi: 10.1016/j.ymthe.2005.12.001. [DOI] [PubMed] [Google Scholar]
- Surace EM, Auricchio A. Versatility of AAV vectors for retinal gene transfer. Vision Res. 2007 doi: 10.1016/j.visres.2007.07.027. [DOI] [PubMed] [Google Scholar]
- Takahashi M, Miyoshi H, Verma IM, Gage FH. Rescue from photoreceptor degeneration in the rd mouse by human immunodeficiency virus vector-mediated gene transfer. J Virol. 1999;73:7812–7816. doi: 10.1128/jvi.73.9.7812-7816.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takaoka A, Wang Z, Choi MK, Yanai H, Negishi H, Ban T, Lu Y, Miyagishi M, Kodama T, Honda K, et al. DAI (DLM-1/ZBP1) is a cytosolic DNA sensor and an activator of innate immune response. Nature. 2007;448:501–505. doi: 10.1038/nature06013. [DOI] [PubMed] [Google Scholar]
- Tan E, Wang Q, Quiambao AB, Xu X, Qtaishat NM, Peachey NS, Lem J, Fliesler SJ, Pepperberg DR, Naash MI, Al-Ubaidi MR. The relationship between opsin overexpression and photoreceptor degeneration. Invest Ophthalmol Vis Sci. 2001;42:589–600. [PubMed] [Google Scholar]
- Tschernutter M, Schlichtenbrede FC, Howe S, Balaggan KS, Munro PM, Bainbridge JW, Thrasher AJ, Smith AJ, Ali RR. Long-term preservation of retinal function in the RCS rat model of retinitis pigmentosa following lentivirus-mediated gene therapy. Gene Ther. 2005;12:694–701. doi: 10.1038/sj.gt.3302460. [DOI] [PubMed] [Google Scholar]
- Vandenberghe L, Bell P, Calcedo R, Wang L, Rebecca G, Irvin T, Hopkins TB, Maguire AM, Wilson JM, Bennett J. Adeno_Associated Virus (AAV)-Mediated Transduction of the Macula in Non-Human Primates (NHPs): Cell-Specificity, Stability and Safety of Transduction After Subretinal Delivery of AAV2 and AAV2/8 Vectors. Invest Ophthalmol Vis Sci. 2007;48 Abstract 1983. [Google Scholar]
- Verma IM, Somia N. Gene therapy -- promises, problems and prospects. Nature. 1997;389:239–242. doi: 10.1038/38410. [DOI] [PubMed] [Google Scholar]
- Von Seggern DJ, Aguilar E, Kinder K, Fleck SK, Gonzalez Armas JC, Stevenson SC, Ghazal P, Nemerow GR, Friedlander M. In vivo transduction of photoreceptors or ciliary body by intravitreal injection of pseudotyped adenoviral vectors. Mol Ther. 2003;7:27–34. doi: 10.1016/s1525-0016(02)00030-8. [DOI] [PubMed] [Google Scholar]
- Wang H, Lieber A. A helper-dependent capsid-modified adenovirus vector expressing adeno-associated virus rep78 mediates site-specific integration of a 27- kilobase transgene cassette. J Virol. 2006;80:11699–11709. doi: 10.1128/JVI.00779-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanez-Munoz RJ, Balaggan KS, MacNeil A, Howe SJ, Schmidt M, Smith AJ, Buch P, MacLaren RE, Anderson PN, Barker SE, et al. Effective gene therapy with nonintegrating lentiviral vectors. Nat Med. 2006;12:348–353. doi: 10.1038/nm1365. [DOI] [PubMed] [Google Scholar]
- Zhang Y, Schlachetzki F, Li JY, Boado RJ, Pardridge WM. Organspecific gene expression in the rhesus monkey eye following intravenous non-viral gene transfer. Mol Vis. 2003;9:465–472. [PubMed] [Google Scholar]
- Zhu C, Zhang Y, Pardridge WM. Widespread expression of an exogenous gene in the eye after intravenous administration. Invest Ophthalmol Vis Sci. 2002;43:3075–3080. [PubMed] [Google Scholar]
- Zhu C, Zhang Y, Zhang YF, Yi Li J, Boado RJ, Pardridge WM. Organ-specific expression of the lacZ gene controlled by the opsin promoter after intravenous gene administration in adult mice. J Gene Med. 2004;6:906–912. doi: 10.1002/jgm.575. [DOI] [PubMed] [Google Scholar]