Abstract
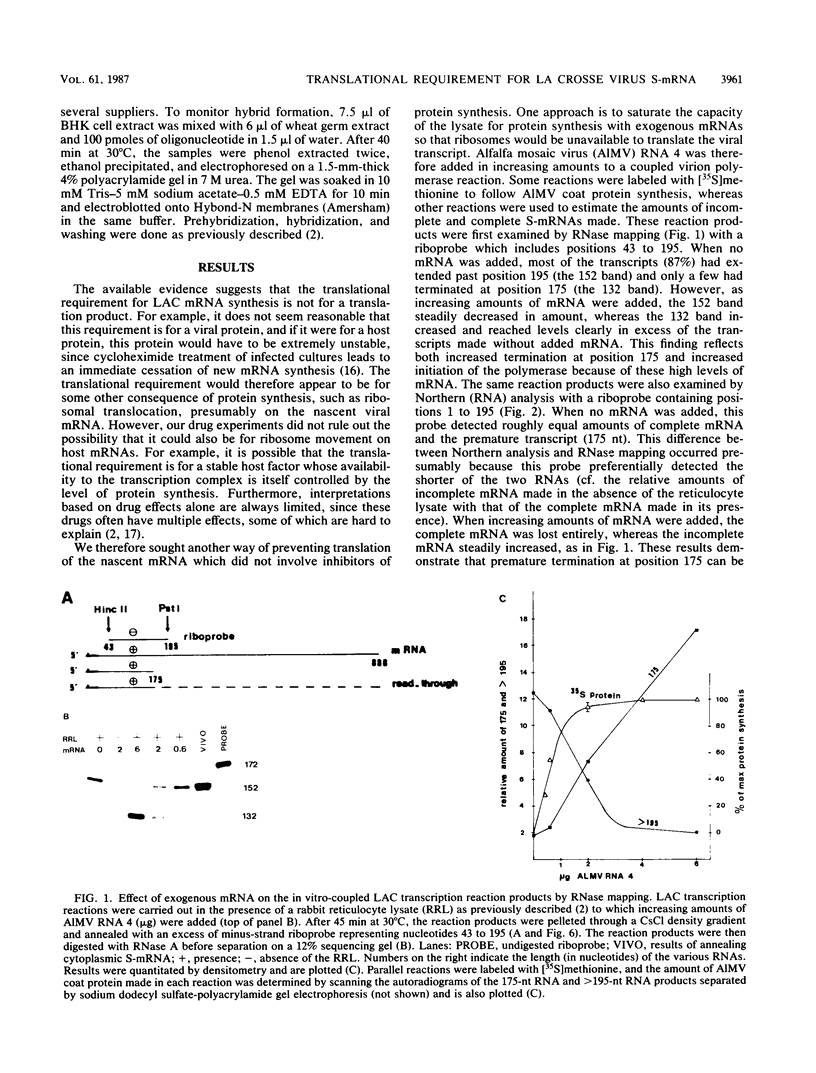
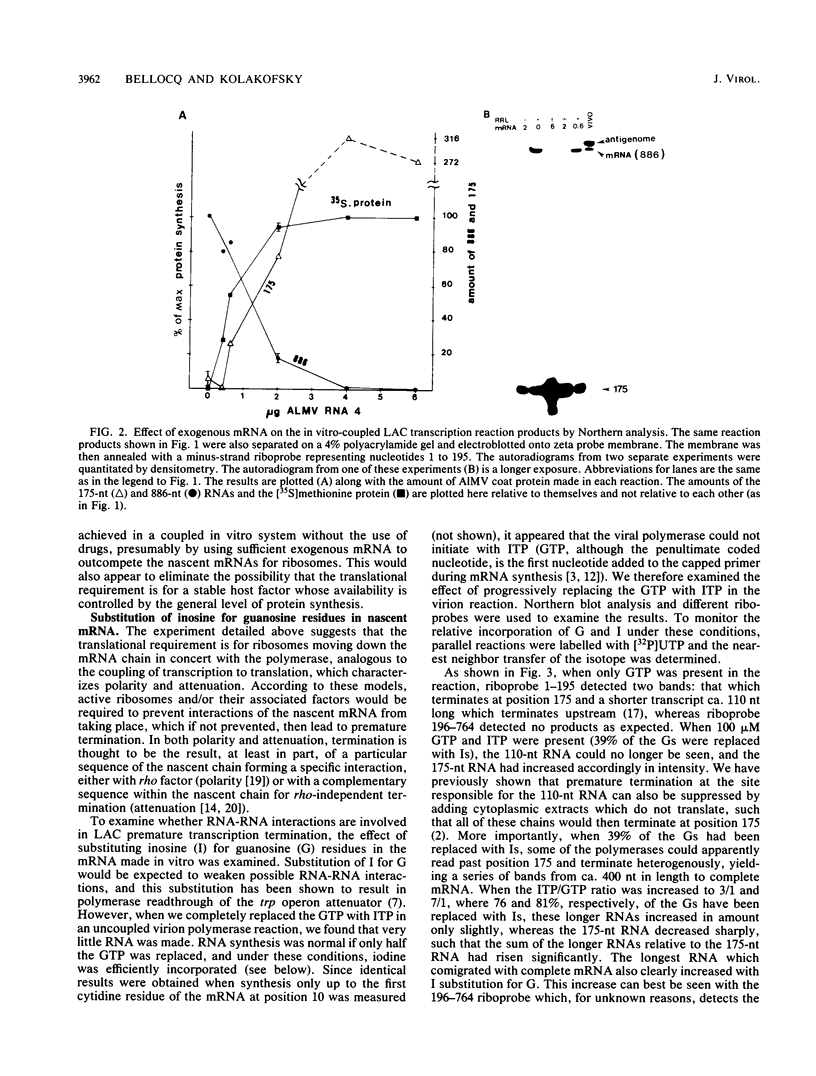
Ongoing protein synthesis is required for La Crosse S-mRNA synthesis in vivo, and complete S-mRNA can be made in vitro only in the presence of an active rabbit reticulocyte lysate. Using in vitro systems based on the polymerase activity of purified virions, we further support the notion that it is translation of the nascent mRNA that is required for complete transcription. Since replacement of guanosine with inosine in the nascent mRNA substitutes for the translational requirement, it appears that translation is required to prevent interactions of the nascent chain from taking place, which, if not prevented, lead to premature termination. These interactions appear to be between the nascent mRNA chain and its nucleocapsid template. A model for the translational requirement for complete S-mRNA synthesis is presented.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abraham G., Pattnaik A. K. Early RNA synthesis in Bunyamwera virus-infected cells. J Gen Virol. 1983 Jun;64(Pt 6):1277–1290. doi: 10.1099/0022-1317-64-6-1277. [DOI] [PubMed] [Google Scholar]
- Bellocq C., Raju R., Patterson J., Kolakofsky D. Translational requirement of La Crosse virus S-mRNA synthesis: in vitro studies. J Virol. 1987 Jan;61(1):87–95. doi: 10.1128/jvi.61.1.87-95.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bishop D. H., Gay M. E., Matsuoko Y. Nonviral heterogeneous sequences are present at the 5' ends of one species of snowshoe hare bunyavirus S complementary RNA. Nucleic Acids Res. 1983 Sep 24;11(18):6409–6418. doi: 10.1093/nar/11.18.6409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fisher R., Yanofsky C. Use of complementary DNA oligomers to probe trp leader transcript secondary structures involved in transcription pausing and termination. Nucleic Acids Res. 1984 Apr 11;12(7):3295–3302. doi: 10.1093/nar/12.7.3295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krug R. M. Priming of influenza viral RNA transcription by capped heterologous RNAs. Curr Top Microbiol Immunol. 1981;93:125–149. doi: 10.1007/978-3-642-68123-3_6. [DOI] [PubMed] [Google Scholar]
- Lee F., Yanofsky C. Transcription termination at the trp operon attenuators of Escherichia coli and Salmonella typhimurium: RNA secondary structure and regulation of termination. Proc Natl Acad Sci U S A. 1977 Oct;74(10):4365–4369. doi: 10.1073/pnas.74.10.4365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Obijeski J. F., Bishop D. H., Palmer E. L., Murphy F. A. Segmented genome and nucleocapsid of La Crosse virus. J Virol. 1976 Dec;20(3):664–675. doi: 10.1128/jvi.20.3.664-675.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Obijeski J. F., Murphy F. A. Bunyaviridae: recent biochemical developments. J Gen Virol. 1977 Oct;37(1):1–14. doi: 10.1099/0022-1317-37-1-1. [DOI] [PubMed] [Google Scholar]
- Patterson J. L., Holloway B., Kolakofsky D. La Crosse virions contain a primer-stimulated RNA polymerase and a methylated cap-dependent endonuclease. J Virol. 1984 Oct;52(1):215–222. doi: 10.1128/jvi.52.1.215-222.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patterson J. L., Kolakofsky D. Characterization of La Crosse virus small-genome transcripts. J Virol. 1984 Mar;49(3):680–685. doi: 10.1128/jvi.49.3.680-685.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pattnaik A. K., Abraham G. Identification of four complementary RNA species in Akabane virus-infected cells. J Virol. 1983 Sep;47(3):452–462. doi: 10.1128/jvi.47.3.452-462.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Platt T. Transcription termination and the regulation of gene expression. Annu Rev Biochem. 1986;55:339–372. doi: 10.1146/annurev.bi.55.070186.002011. [DOI] [PubMed] [Google Scholar]
- Raju R., Kolakofsky D. Inhibitors of protein synthesis inhibit both La Crosse virus S-mRNA and S genome syntheses in vivo. Virus Res. 1986 Jul;5(1):1–9. doi: 10.1016/0168-1702(86)90061-4. [DOI] [PubMed] [Google Scholar]
- Raju R., Kolakofsky D. Translational requirement of La Crosse virus S-mRNA synthesis: in vivo studies. J Virol. 1987 Jan;61(1):96–103. doi: 10.1128/jvi.61.1.96-103.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rossier C., Patterson J., Kolakofsky D. La Crosse virus small genome mRNA is made in the cytoplasm. J Virol. 1986 May;58(2):647–650. doi: 10.1128/jvi.58.2.647-650.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanofsky C. Attenuation in the control of expression of bacterial operons. Nature. 1981 Feb 26;289(5800):751–758. doi: 10.1038/289751a0. [DOI] [PubMed] [Google Scholar]
- von Hippel P. H., Bear D. G., Morgan W. D., McSwiggen J. A. Protein-nucleic acid interactions in transcription: a molecular analysis. Annu Rev Biochem. 1984;53:389–446. doi: 10.1146/annurev.bi.53.070184.002133. [DOI] [PubMed] [Google Scholar]