Abstract
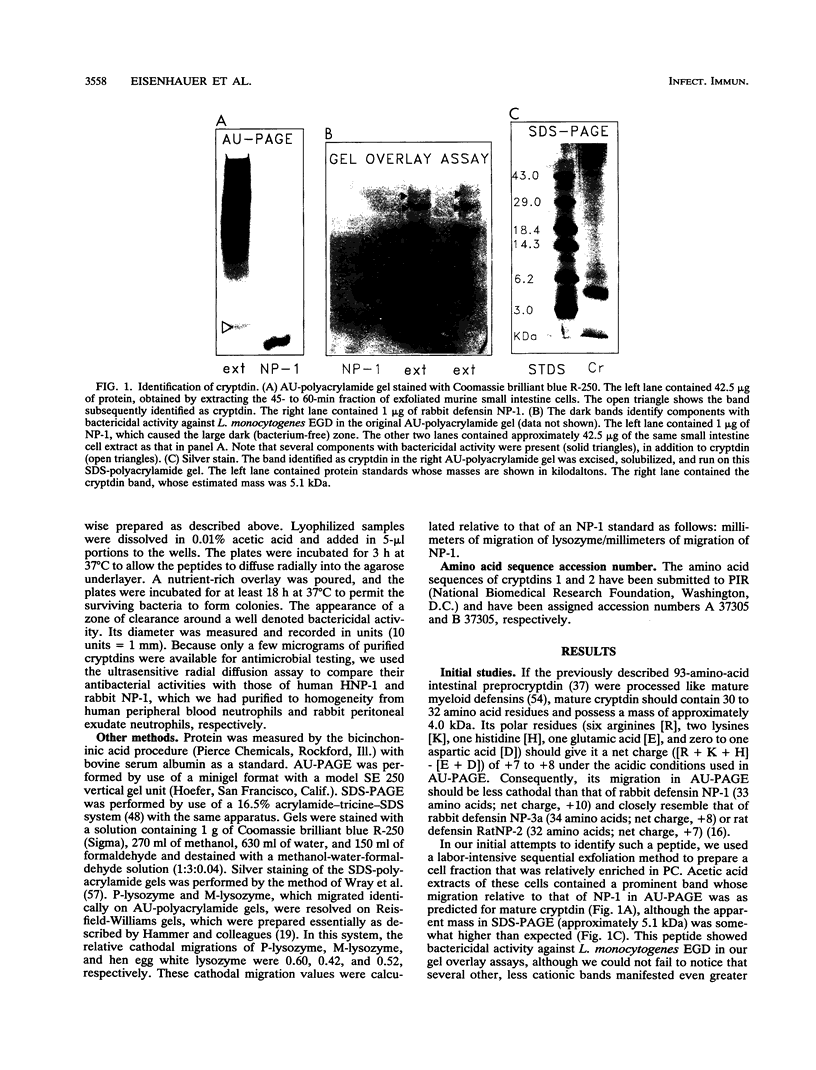
Paneth cells are specialized small intestine epithelial cells that contain lysozyme, possess phagocytic properties, and secrete cytoplasmic granules into the intestinal crypt lumen after the entry of bacteria. Recent studies by Ouellette and associates (A. J. Ouellette, R. M. Greco, M. James, D. Frederick, J. Naftilan, and J. T. Fallon, J. Cell Biol. 108:1687-1695, 1989) indicated that murine Paneth cells produce prodefensin mRNA, but the properties of its peptide product were not reported. We purified two closely related defensins, cryptdin 1 and cryptdin 2, from a subcellular fraction of murine small intestine cells that was enriched in Paneth cells. Both peptides contained 35 amino acid residues, including the characteristic defensin "signature" of six invariantly conserved cysteines. Cryptdins 1 and 2 were approximately 90 to 95% homologous to each other and to the carboxy-terminal domain of the 93-amino-acid defensin precursor, cryptdin A, described by Ouellette and associates (Ouellette et al., J. Cell Biol. 108:1687-1695, 1989). Both cryptdins exerted bactericidal activity against Listeria monocytogenes EGD and Escherichia coli ML-35p in vitro. Their potency exceeded that of human neutrophil defensin HNP-1 but was considerably lower than that of NP-1, a defensin produced by rabbit neutrophils and alveolar macrophages. Both cryptdins killed mouse-avirulent Salmonella typhimurium 7953S (phoP) much more effectively than its phoP+, mouse-virulent, isogenic counterpart, S. typhimurium 14028S. Our data indicate that mouse intestinal prodefensins are processed into 35-amino-acid mature defensins (cryptdins) with broad-spectrum antimicrobial properties. The production of defensins and lysozyme by Paneth cells may enable them to protect the small intestine from bacterial overgrowth by autochthonous flora and from invasion by potential pathogens that cause infection via the peroral route, such as L. monocytogenes and Salmonella species.
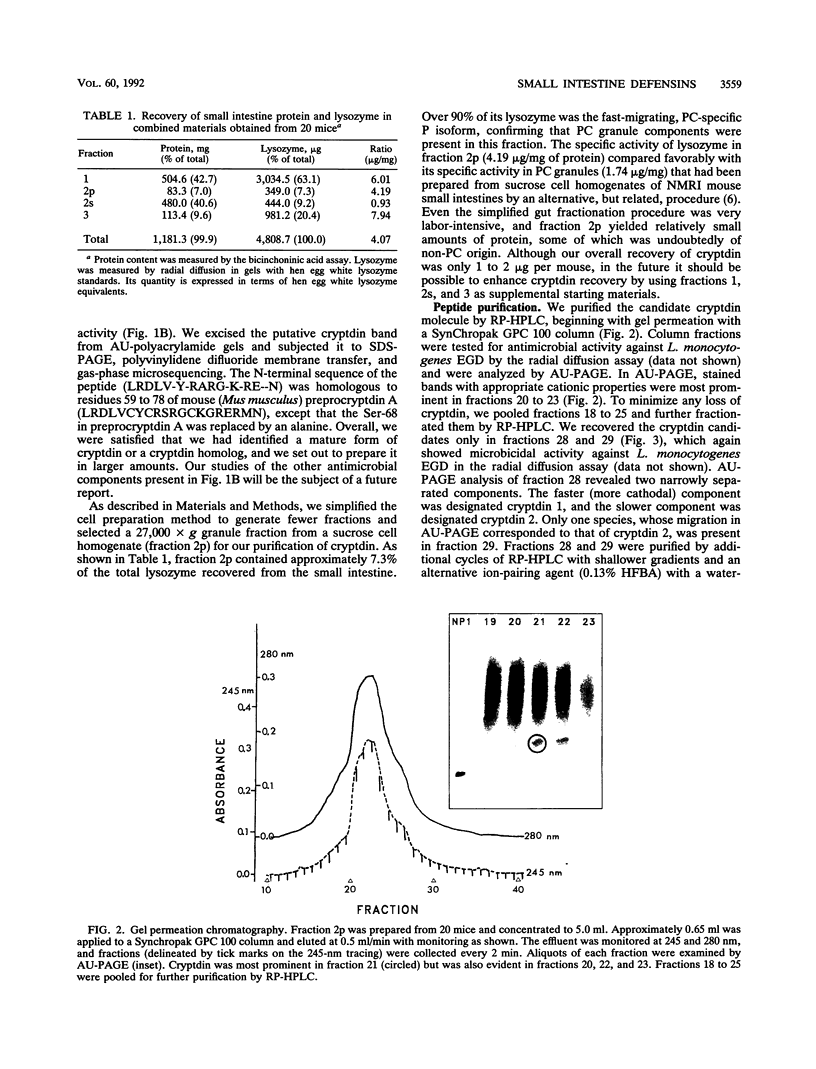
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ahonen A., Penttilä Effects of fasting and feeding and pilocarpine on paneth cells of the mouse. Scand J Gastroenterol. 1975;10(4):347–352. [PubMed] [Google Scholar]
- Boman H. G., Hultmark D. Cell-free immunity in insects. Annu Rev Microbiol. 1987;41:103–126. doi: 10.1146/annurev.mi.41.100187.000535. [DOI] [PubMed] [Google Scholar]
- Charp P. A., Rice W. G., Raynor R. L., Reimund E., Kinkade J. M., Jr, Ganz T., Selsted M. E., Lehrer R. I., Kuo J. F. Inhibition of protein kinase C by defensins, antibiotic peptides from human neutrophils. Biochem Pharmacol. 1988 Mar 1;37(5):951–956. doi: 10.1016/0006-2952(88)90187-6. [DOI] [PubMed] [Google Scholar]
- Cheng H. Origin, differentiation and renewal of the four main epithelial cell types in the mouse small intestine. IV. Paneth cells. Am J Anat. 1974 Dec;141(4):521–535. doi: 10.1002/aja.1001410406. [DOI] [PubMed] [Google Scholar]
- Deckx R. J., Vantrappen G. R., Parein M. M. Localization of lysozyme activity in a Paneth cell granule fraction. Biochim Biophys Acta. 1967 May 16;139(1):204–207. doi: 10.1016/0005-2744(67)90136-2. [DOI] [PubMed] [Google Scholar]
- Diamond G., Zasloff M., Eck H., Brasseur M., Maloy W. L., Bevins C. L. Tracheal antimicrobial peptide, a cysteine-rich peptide from mammalian tracheal mucosa: peptide isolation and cloning of a cDNA. Proc Natl Acad Sci U S A. 1991 May 1;88(9):3952–3956. doi: 10.1073/pnas.88.9.3952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dickman M. D., Chappelka A. R., Schaedler R. W. The Microbial ecology of the upper small bowel. Am J Gastroenterol. 1976 Jan;65(1):57–62. [PubMed] [Google Scholar]
- Dubos R. J., Savage D. C., Schaedler R. W. The indigenous flora of the gastrointestinal tract. Dis Colon Rectum. 1967 Jan-Feb;10(1):23–34. doi: 10.1007/BF02617382. [DOI] [PubMed] [Google Scholar]
- Eisenhauer P., Harwig S. S., Szklarek D., Ganz T., Lehrer R. I. Polymorphic expression of defensins in neutrophils from outbred rats. Infect Immun. 1990 Dec;58(12):3899–3902. doi: 10.1128/iai.58.12.3899-3902.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erlandsen S. L., Chase D. G. Paneth cell function: phagocytosis and intracellular digestion of intestinal microorganisms. I. Hexamita muris. J Ultrastruct Res. 1972 Nov;41(3):296–318. doi: 10.1016/s0022-5320(72)90071-8. [DOI] [PubMed] [Google Scholar]
- Erlandsen S. L., Chase D. G. Paneth cell function: phagocytosis and intracellular digestion of intestinal microorganisms. II. Spiral microorganism. J Ultrastruct Res. 1972 Nov;41(3):319–333. doi: 10.1016/s0022-5320(72)90072-x. [DOI] [PubMed] [Google Scholar]
- Fields P. I., Groisman E. A., Heffron F. A Salmonella locus that controls resistance to microbicidal proteins from phagocytic cells. Science. 1989 Feb 24;243(4894 Pt 1):1059–1062. doi: 10.1126/science.2646710. [DOI] [PubMed] [Google Scholar]
- Ganz T., Rayner J. R., Valore E. V., Tumolo A., Talmadge K., Fuller F. The structure of the rabbit macrophage defensin genes and their organ-specific expression. J Immunol. 1989 Aug 15;143(4):1358–1365. [PubMed] [Google Scholar]
- Ganz T., Selsted M. E., Lehrer R. I. Defensins. Eur J Haematol. 1990 Jan;44(1):1–8. doi: 10.1111/j.1600-0609.1990.tb00339.x. [DOI] [PubMed] [Google Scholar]
- Gorbach S. L. Intestinal microflora. Gastroenterology. 1971 Jun;60(6):1110–1129. [PubMed] [Google Scholar]
- Groisman E. A., Chiao E., Lipps C. J., Heffron F. Salmonella typhimurium phoP virulence gene is a transcriptional regulator. Proc Natl Acad Sci U S A. 1989 Sep;86(18):7077–7081. doi: 10.1073/pnas.86.18.7077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hammer M. F., Schilling J. W., Prager E. M., Wilson A. C. Recruitment of lysozyme as a major enzyme in the mouse gut: duplication, divergence, and regulatory evolution. J Mol Evol. 1987;24(3):272–279. doi: 10.1007/BF02111240. [DOI] [PubMed] [Google Scholar]
- Hammer M. F., Wilson A. C. Regulatory and structural genes for lysozymes of mice. Genetics. 1987 Mar;115(3):521–533. doi: 10.1093/genetics/115.3.521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jollès P., Jollès J. What's new in lysozyme research? Always a model system, today as yesterday. Mol Cell Biochem. 1984 Sep;63(2):165–189. doi: 10.1007/BF00285225. [DOI] [PubMed] [Google Scholar]
- Keshav S., Lawson L., Chung L. P., Stein M., Perry V. H., Gordon S. Tumor necrosis factor mRNA localized to Paneth cells of normal murine intestinal epithelium by in situ hybridization. J Exp Med. 1990 Jan 1;171(1):327–332. doi: 10.1084/jem.171.1.327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawson A. J., Smit R. A., Jeffers N. A., Osborne J. W. Isolation of rat intestinal crypt cells. Cell Tissue Kinet. 1982 Jan;15(1):69–80. doi: 10.1111/j.1365-2184.1982.tb01025.x. [DOI] [PubMed] [Google Scholar]
- Lee J. Y., Boman A., Sun C. X., Andersson M., Jörnvall H., Mutt V., Boman H. G. Antibacterial peptides from pig intestine: isolation of a mammalian cecropin. Proc Natl Acad Sci U S A. 1989 Dec;86(23):9159–9162. doi: 10.1073/pnas.86.23.9159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lehrer R. I., Barton A., Ganz T. Concurrent assessment of inner and outer membrane permeabilization and bacteriolysis in E. coli by multiple-wavelength spectrophotometry. J Immunol Methods. 1988 Apr 6;108(1-2):153–158. doi: 10.1016/0022-1759(88)90414-0. [DOI] [PubMed] [Google Scholar]
- Lehrer R. I., Ganz T. Antimicrobial polypeptides of human neutrophils. Blood. 1990 Dec 1;76(11):2169–2181. [PubMed] [Google Scholar]
- Lehrer R. I., Rosenman M., Harwig S. S., Jackson R., Eisenhauer P. Ultrasensitive assays for endogenous antimicrobial polypeptides. J Immunol Methods. 1991 Mar 21;137(2):167–173. doi: 10.1016/0022-1759(91)90021-7. [DOI] [PubMed] [Google Scholar]
- Lichtenstein A. K., Ganz T., Nguyen T. M., Selsted M. E., Lehrer R. I. Mechanism of target cytolysis by peptide defensins. Target cell metabolic activities, possibly involving endocytosis, are crucial for expression of cytotoxicity. J Immunol. 1988 Apr 15;140(8):2686–2694. [PubMed] [Google Scholar]
- Lopez-Lewellyn J., Erlandsen S. L. Cytodifferentiation of the rat Paneth cell: an immunocytochemical investigation in suckling and weanling animals. Am J Anat. 1980 Jul;158(3):285–297. doi: 10.1002/aja.1001580305. [DOI] [PubMed] [Google Scholar]
- Matsudaira P. Sequence from picomole quantities of proteins electroblotted onto polyvinylidene difluoride membranes. J Biol Chem. 1987 Jul 25;262(21):10035–10038. [PubMed] [Google Scholar]
- Merzel J. Some histophysiological aspects of Paneth's cells of mice as shown by histochemical and radioautographical studies. Acta Anat (Basel) 1967;66(4):603–630. doi: 10.1159/000142964. [DOI] [PubMed] [Google Scholar]
- Miller S. I., Kukral A. M., Mekalanos J. J. A two-component regulatory system (phoP phoQ) controls Salmonella typhimurium virulence. Proc Natl Acad Sci U S A. 1989 Jul;86(13):5054–5058. doi: 10.1073/pnas.86.13.5054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller S. I., Pulkkinen W. S., Selsted M. E., Mekalanos J. J. Characterization of defensin resistance phenotypes associated with mutations in the phoP virulence regulon of Salmonella typhimurium. Infect Immun. 1990 Nov;58(11):3706–3710. doi: 10.1128/iai.58.11.3706-3710.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mushin R., Dubos R. Colonization of the mouse intestine with Escherichia coli. J Exp Med. 1965 Oct 1;122(4):745–757. doi: 10.1084/jem.122.4.745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ouellette A. J., Greco R. M., James M., Frederick D., Naftilan J., Fallon J. T. Developmental regulation of cryptdin, a corticostatin/defensin precursor mRNA in mouse small intestinal crypt epithelium. J Cell Biol. 1989 May;108(5):1687–1695. doi: 10.1083/jcb.108.5.1687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poulsen S. S., Nexø E., Olsen P. S., Hess J., Kirkegaard P. Immunohistochemical localization of epidermal growth factor in rat and man. Histochemistry. 1986;85(5):389–394. doi: 10.1007/BF00982668. [DOI] [PubMed] [Google Scholar]
- Pulkkinen W. S., Miller S. I. A Salmonella typhimurium virulence protein is similar to a Yersinia enterocolitica invasion protein and a bacteriophage lambda outer membrane protein. J Bacteriol. 1991 Jan;173(1):86–93. doi: 10.1128/jb.173.1.86-93.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Satoh Y. Atropine inhibits the degranulation of Paneth cells in ex-germ-free mice. Cell Tissue Res. 1988 Aug;253(2):397–402. doi: 10.1007/BF00222296. [DOI] [PubMed] [Google Scholar]
- Satoh Y. Effect of live and heat-killed bacteria on the secretory activity of Paneth cells in germ-free mice. Cell Tissue Res. 1988 Jan;251(1):87–93. doi: 10.1007/BF00215451. [DOI] [PubMed] [Google Scholar]
- Satoh Y., Ishikawa K., Tanaka H., Ono K. Immunohistochemical observations of immunoglobulin A in the Paneth cells of germ-free and formerly-germ-free rats. Histochemistry. 1986;85(3):197–201. doi: 10.1007/BF00494804. [DOI] [PubMed] [Google Scholar]
- Satoh Y., Ishikawa K., Tanaka H., Oomori Y., Ono K. Immunohistochemical observations of lysozyme in the Paneth cells of specific-pathogen-free and germ-free mice. Acta Histochem. 1988;83(2):185–188. doi: 10.1016/S0065-1281(88)80055-2. [DOI] [PubMed] [Google Scholar]
- Satoh Y., Yamano M., Matsuda M., Ono K. Ultrastructure of Paneth cells in the intestine of various mammals. J Electron Microsc Tech. 1990 Sep;16(1):69–80. doi: 10.1002/jemt.1060160109. [DOI] [PubMed] [Google Scholar]
- Savage D. C., Dubos R., Schaedler R. W. The gastrointestinal epithelium and its autochthonous bacterial flora. J Exp Med. 1968 Jan 1;127(1):67–76. doi: 10.1084/jem.127.1.67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schaedler R. W. The relationshp between the host and its intestinal microflora. Proc Nutr Soc. 1973 Sep;32(2):41–47. doi: 10.1079/pns19730013. [DOI] [PubMed] [Google Scholar]
- Schägger H., von Jagow G. Tricine-sodium dodecyl sulfate-polyacrylamide gel electrophoresis for the separation of proteins in the range from 1 to 100 kDa. Anal Biochem. 1987 Nov 1;166(2):368–379. doi: 10.1016/0003-2697(87)90587-2. [DOI] [PubMed] [Google Scholar]
- Senegas-Balas F., Balas D., Verger R., de Caro A., Figarella C., Ferrato F., Lechene P., Bertrand C., Ribet A. Immunohistochemical localization of intestinal phospholipase A2 in rat paneth cells. Histochemistry. 1984;81(6):581–584. doi: 10.1007/BF00489538. [DOI] [PubMed] [Google Scholar]
- Sheu M. J., Baldwin W. W., Brunson K. W. Cytotoxicity of rabbit macrophage peptides MCP-1 and MCP-2 for mouse tumor cells. Antimicrob Agents Chemother. 1985 Nov;28(5):626–629. doi: 10.1128/aac.28.5.626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spicer S. S., Staley M. W., Wetzel M. G., Wetzel B. K. Acid mucosubstance and basic protein in mouse Paneth cells. J Histochem Cytochem. 1967 Apr;15(4):225–242. doi: 10.1177/15.4.225. [DOI] [PubMed] [Google Scholar]
- Territo M. C., Ganz T., Selsted M. E., Lehrer R. Monocyte-chemotactic activity of defensins from human neutrophils. J Clin Invest. 1989 Dec;84(6):2017–2020. doi: 10.1172/JCI114394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiser M. M. Intestinal epithelial cell surface membrane glycoprotein synthesis. I. An indicator of cellular differentiation. J Biol Chem. 1973 Apr 10;248(7):2536–2541. [PubMed] [Google Scholar]
- Wray W., Boulikas T., Wray V. P., Hancock R. Silver staining of proteins in polyacrylamide gels. Anal Biochem. 1981 Nov 15;118(1):197–203. doi: 10.1016/0003-2697(81)90179-2. [DOI] [PubMed] [Google Scholar]
- Zasloff M. Magainins, a class of antimicrobial peptides from Xenopus skin: isolation, characterization of two active forms, and partial cDNA sequence of a precursor. Proc Natl Acad Sci U S A. 1987 Aug;84(15):5449–5453. doi: 10.1073/pnas.84.15.5449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu Q. Z., Hu J., Mulay S., Esch F., Shimasaki S., Solomon S. Isolation and structure of corticostatin peptides from rabbit fetal and adult lung. Proc Natl Acad Sci U S A. 1988 Jan;85(2):592–596. doi: 10.1073/pnas.85.2.592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu Q. Z., Singh A. V., Bateman A., Esch F., Solomon S. The corticostatic (anti-ACTH) and cytotoxic activity of peptides isolated from fetal, adult and tumor-bearing lung. J Steroid Biochem. 1987;27(4-6):1017–1022. doi: 10.1016/0022-4731(87)90184-1. [DOI] [PubMed] [Google Scholar]