Abstract
Many researchers have suggested that the role of glucan-mediated interactions in the adherence of Streptococcus mutans is restricted to accumulation of this cariogenic bacterium following its sucrose (i.e., glucan)-independent binding to saliva-coated tooth surfaces. However, the presence of enzymatically active glucosyltransferase in salivary pellicle suggests that glucans could also promote the initial adherence of S. mutans to the teeth. In the present study, the commonly used hydroxyapatite adherence assay was modified to include the incorporation of glucosyltransferase and the synthesis of glucans in situ on saliva-coated hydroxyapatite beads. Several laboratory strains and clinical isolates of S. mutans were examined for their ability to adhere to experimental pellicles, either with or without the prior formation of glucans in situ. Results showed that most strains of S. mutans bound stereospecifically to glucans synthesized in pellicle. Inhibition studies with various polysaccharides and fungal dextranase indicated that alpha 1,6-linked glucose residues were of primary importance in the glucan binding observed. Scanning electron microscopic analysis showed direct binding of S. mutans to hydroxyapatite surface-associated polysaccharide and revealed no evidence of trapping or cell-to-cell binding. S. mutans strains also attached to host-derived structures in experimental pellicles, and the data suggest that the bacterial adhesins which recognize salivary binding sites were distinct from glucan-binding adhesins. Furthermore, glucans formed in experimental pellicles appeared to mask the host-derived components. These results support the concept that glucans synthesized in salivary pellicle can promote the selective adherence of the cariogenic streptococci which colonize human teeth.
Full text
PDF

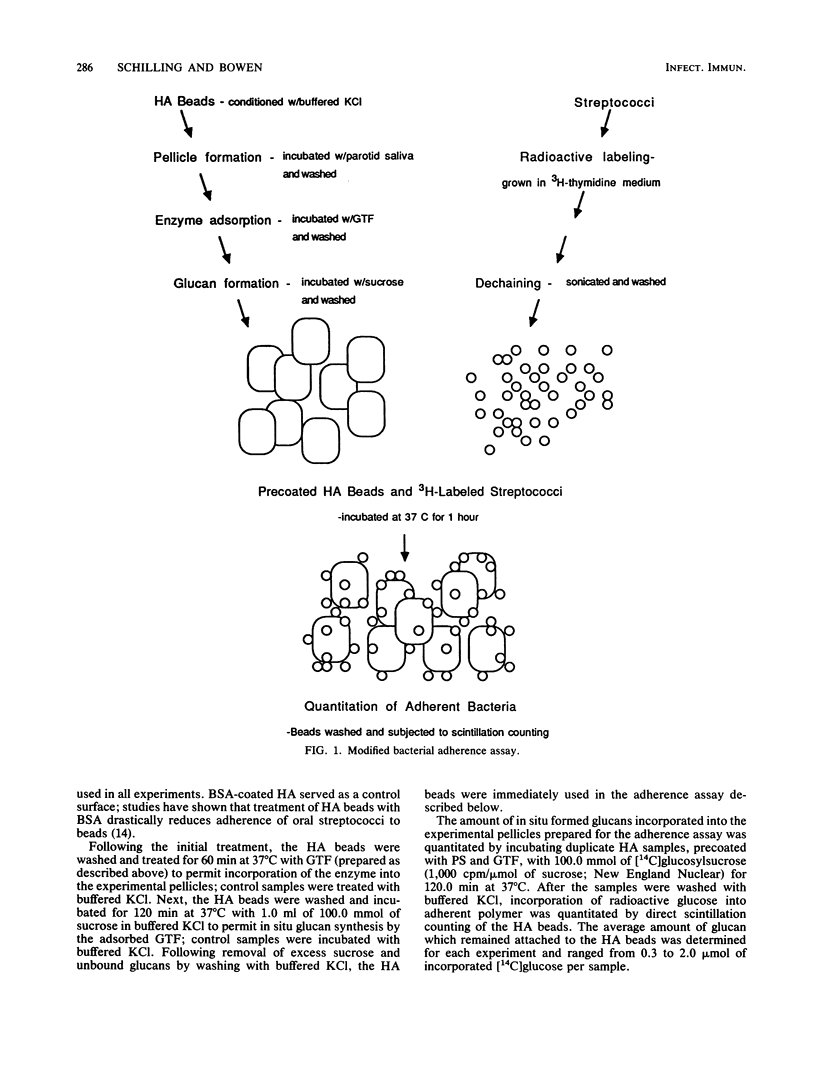
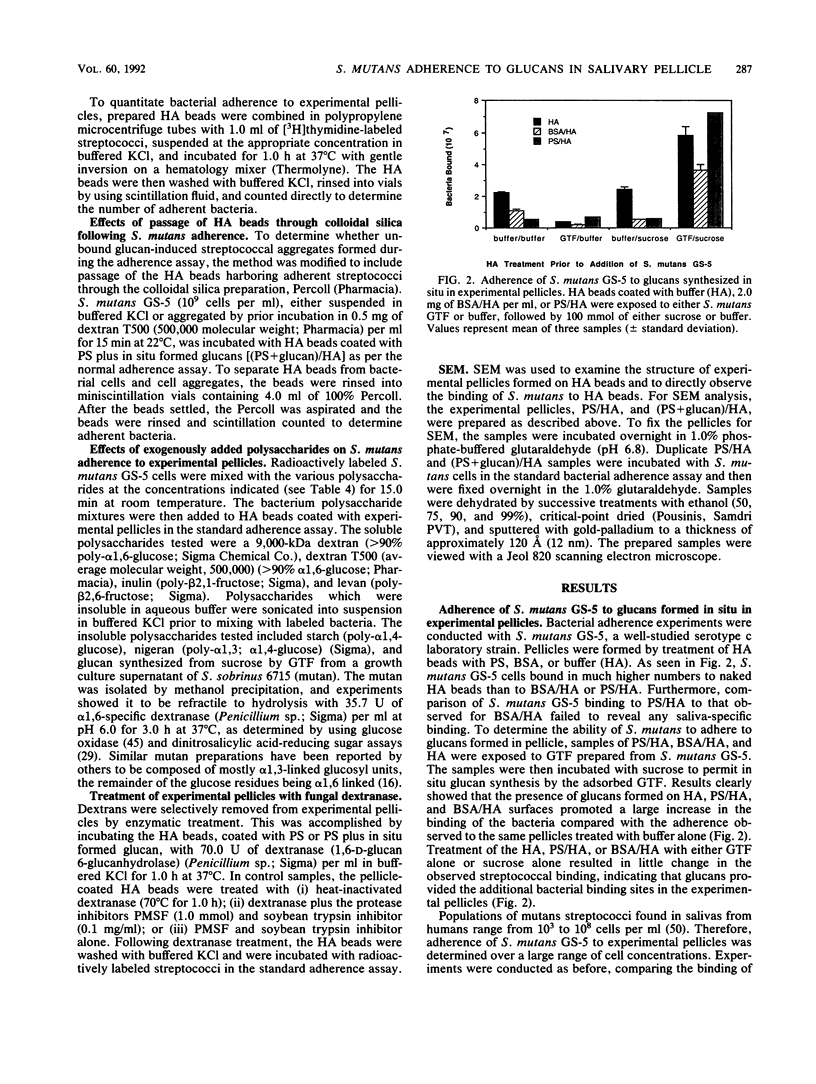

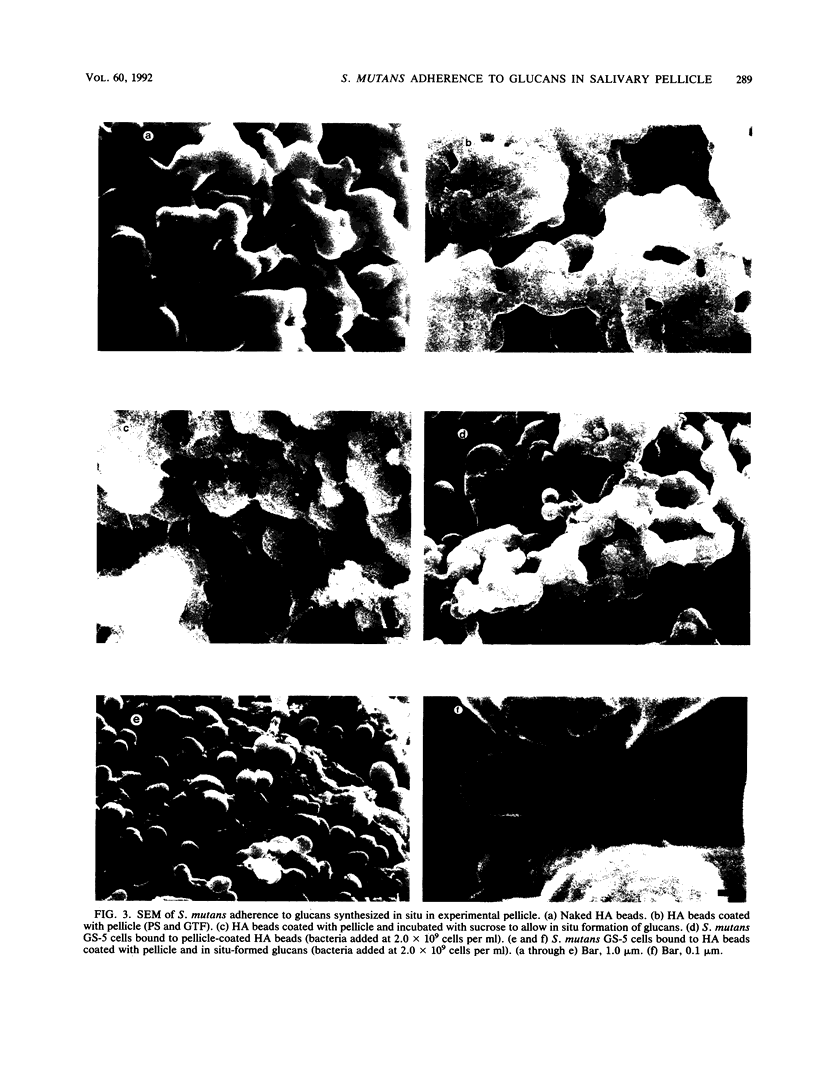

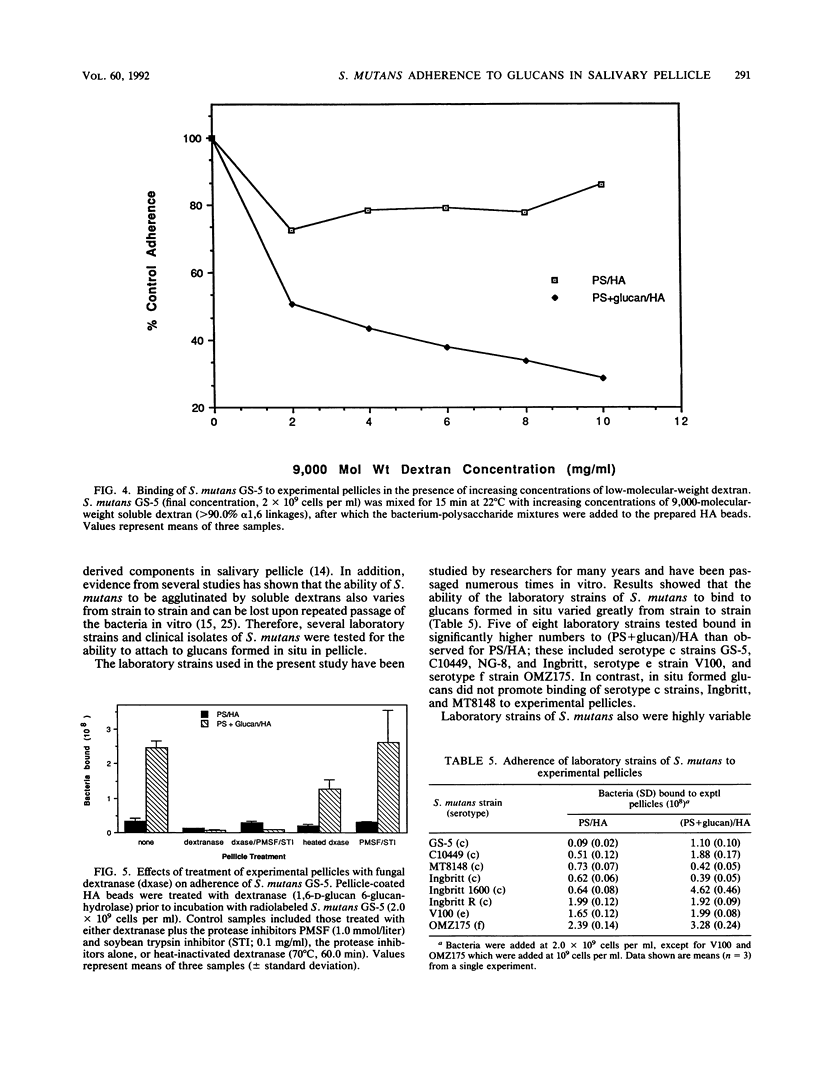



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ayakawa G. Y., Boushell L. W., Crowley P. J., Erdos G. W., McArthur W. P., Bleiweis A. S. Isolation and characterization of monoclonal antibodies specific for antigen P1, a major surface protein of mutans streptococci. Infect Immun. 1987 Nov;55(11):2759–2767. doi: 10.1128/iai.55.11.2759-2767.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bowen W. H., Schilling K., Giertsen E., Pearson S., Lee S. F., Bleiweis A., Beeman D. Role of a cell surface-associated protein in adherence and dental caries. Infect Immun. 1991 Dec;59(12):4606–4609. doi: 10.1128/iai.59.12.4606-4609.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cisar J. O., Sandberg A. L., Mergenhagen S. E. The function and distribution of different fimbriae on strains of Actinomyces viscosus and Actinomyces naeslundii. J Dent Res. 1984 Mar;63(3):393–396. doi: 10.1177/00220345840630030701. [DOI] [PubMed] [Google Scholar]
- Clark W. B., Bammann L. L., Gibbons R. J. Comparative estimates of bacterial affinities and adsorption sites on hydroxyapatite surfaces. Infect Immun. 1978 Mar;19(3):846–853. doi: 10.1128/iai.19.3.846-853.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark W. B., Gibbons R. J. Influence of salivary components and extracellular polysaccharide synthesis from sucrose on the attachment of Streptococcus mutans 6715 to hydroxyapatite surfaces. Infect Immun. 1977 Nov;18(2):514–523. doi: 10.1128/iai.18.2.514-523.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Douglas C. W., Russell R. R. Effect of specific antisera on adherence properties of the oral bacterium Streptococcus mutans. Arch Oral Biol. 1982;27(12):1039–1045. doi: 10.1016/0003-9969(82)90009-7. [DOI] [PubMed] [Google Scholar]
- Drake D., Taylor K. G., Bleiweis A. S., Doyle R. J. Specificity of the glucan-binding lectin of Streptococcus cricetus. Infect Immun. 1988 Aug;56(8):1864–1872. doi: 10.1128/iai.56.8.1864-1872.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drake D., Taylor K. G., Doyle R. J. Expression of the glucan-binding lectin of Streptococcus cricetus requires manganous ion. Infect Immun. 1988 Aug;56(8):2205–2207. doi: 10.1128/iai.56.8.2205-2207.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freedman M., Birked D., Granath K. Analyses of glucans from cariogenic and mutant Streptococcus mutans. Infect Immun. 1978 Jul;21(1):17–27. doi: 10.1128/iai.21.1.17-27.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frostell G., Keyes P. H., Larson R. H. Effect of various sugars and sugar substitutes on dental caries in hamsters and rats. J Nutr. 1967 Sep;93(1):65–76. doi: 10.1093/jn/93.1.65. [DOI] [PubMed] [Google Scholar]
- Gibbons R. J. Adherent interactions which may affect microbial ecology in the mouth. J Dent Res. 1984 Mar;63(3):378–385. doi: 10.1177/00220345840630030401. [DOI] [PubMed] [Google Scholar]
- Gibbons R. J., Cohen L., Hay D. I. Strains of Streptococcus mutans and Streptococcus sobrinus attach to different pellicle receptors. Infect Immun. 1986 May;52(2):555–561. doi: 10.1128/iai.52.2.555-561.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibbons R. J., Fitzgerald R. J. Dextran-induced agglutination of Streptococcus mutans, and its potential role in the formation of microbial dental plaques. J Bacteriol. 1969 May;98(2):341–346. doi: 10.1128/jb.98.2.341-346.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guggenheim B. Enzymatic hydrolysis and structure of water-insoluble glucan produced by glucosyltransferases from a strain of streptococcus mutans. Helv Odontol Acta. 1970 Nov;14(Suppl):89+–89+. [PubMed] [Google Scholar]
- Hamada S., Slade H. D. Biology, immunology, and cariogenicity of Streptococcus mutans. Microbiol Rev. 1980 Jun;44(2):331–384. doi: 10.1128/mr.44.2.331-384.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamada S., Torii M. Effect of sucrose in culture media on the location of glucosyltransferase of Streptococcus mutans and cell adherence to glass surfaces. Infect Immun. 1978 Jun;20(3):592–599. doi: 10.1128/iai.20.3.592-599.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inoue M., Shibata H., Morioka T. Serotype specificity for some biochemical characteristics of Streptococcus mutans. Microbios. 1982;33(131):7–14. [PubMed] [Google Scholar]
- KRASSE B. THE EFFECT OF CARIES-INDUCING STREPTOCOCCI IN HAMSTERS FED DIETS WITH SUCROSE OR GLUCOSE. Arch Oral Biol. 1965 Mar-Apr;10:223–226. doi: 10.1016/0003-9969(65)90023-3. [DOI] [PubMed] [Google Scholar]
- Kelstrup J., Funder-Nielsen T. D. Adhesion of dextran to Streptococcus mutans. J Gen Microbiol. 1974 Apr;81(2):485–489. doi: 10.1099/00221287-81-2-485. [DOI] [PubMed] [Google Scholar]
- Knox K. W., Hardy L. N., Wicken A. J. Comparative studies on the protein profiles and hydrophobicity of strains of Streptococcus mutans serotype c. J Gen Microbiol. 1986 Sep;132(9):2541–2548. doi: 10.1099/00221287-132-9-2541. [DOI] [PubMed] [Google Scholar]
- Koga T., Asakawa H., Okahashi N., Hamada S. Sucrose-dependent cell adherence and cariogenicity of serotype c Streptococcus mutans. J Gen Microbiol. 1986 Oct;132(10):2873–2883. doi: 10.1099/00221287-132-10-2873. [DOI] [PubMed] [Google Scholar]
- Kuramitsu H. K. Adherence of Streptococcus mutans to dextran synthesized in the presence of extracellular dextransucrase. Infect Immun. 1974 Apr;9(4):764–765. doi: 10.1128/iai.9.4.764-765.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuramitsu H. K. Properties of a mutant of Streptococcus mutans altered in glucosyltransferase activity. Infect Immun. 1976 Feb;13(2):345–353. doi: 10.1128/iai.13.2.345-353.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LUCHSINGER W. W., CORNESKY R. A. Reducing power by the dinitrosalicylic acid method. Anal Biochem. 1962 Oct;4:346–347. doi: 10.1016/0003-2697(62)90098-2. [DOI] [PubMed] [Google Scholar]
- Landale E. C., McCabe M. M. Characterization by affinity electrophoresis of an alpha-1,6-glucan-binding protein from Streptococcus sobrinus. Infect Immun. 1987 Dec;55(12):3011–3016. doi: 10.1128/iai.55.12.3011-3016.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee S. F., Progulske-Fox A., Erdos G. W., Piacentini D. A., Ayakawa G. Y., Crowley P. J., Bleiweis A. S. Construction and characterization of isogenic mutants of Streptococcus mutans deficient in major surface protein antigen P1 (I/II). Infect Immun. 1989 Nov;57(11):3306–3313. doi: 10.1128/iai.57.11.3306-3313.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCabe M. M., Hamelik R. M. Multiple forms of dextran-binding proteins from Streptococcus mutans. Adv Exp Med Biol. 1978;107:749–759. doi: 10.1007/978-1-4684-3369-2_84. [DOI] [PubMed] [Google Scholar]
- McCabe M. M., Smith E. E. Relationship between cell-bound dextransucrase and the agglutination of Streptococcus mutans. Infect Immun. 1975 Sep;12(3):512–520. doi: 10.1128/iai.12.3.512-520.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mooser G., Wong C. Isolation of a glucan-binding domain of glucosyltransferase (1,6-alpha-glucan synthase) from Streptococcus sobrinus. Infect Immun. 1988 Apr;56(4):880–884. doi: 10.1128/iai.56.4.880-884.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morris E. J., McBride B. C. Adherence of Streptococcus sanguis to saliva-coated hydroxyapatite: evidence for two binding sites. Infect Immun. 1984 Feb;43(2):656–663. doi: 10.1128/iai.43.2.656-663.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohta H., Kato H., Okahashi N., Takahashi I., Hamada S., Koga T. Characterization of a cell-surface protein antigen of hydrophilic Streptococcus mutans strain GS-5. J Gen Microbiol. 1989 Apr;135(4):981–988. doi: 10.1099/00221287-135-4-981. [DOI] [PubMed] [Google Scholar]
- Okahashi N., Sasakawa C., Yoshikawa M., Hamada S., Koga T. Molecular characterization of a surface protein antigen gene from serotype c Streptococcus mutans, implicated in dental caries. Mol Microbiol. 1989 May;3(5):673–678. doi: 10.1111/j.1365-2958.1989.tb00215.x. [DOI] [PubMed] [Google Scholar]
- Perry D., Kuramitsu H. K. Linkage of sucrose-metabolizing genes in Streptococcus mutans. Infect Immun. 1990 Oct;58(10):3462–3464. doi: 10.1128/iai.58.10.3462-3464.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russell R. R., Coleman D., Dougan G. Expression of a gene for glucan-binding protein from Streptococcus mutans in Escherichia coli. J Gen Microbiol. 1985 Feb;131(2):295–299. doi: 10.1099/00221287-131-2-295. [DOI] [PubMed] [Google Scholar]
- Russell R. R. Glucan-binding proteins of Streptococcus mutans serotype c. J Gen Microbiol. 1979 May;112(1):197–201. doi: 10.1099/00221287-112-1-197. [DOI] [PubMed] [Google Scholar]
- Scheie A. A., Eggen K. H., Rölla G. Glucosyltransferase activity in human in vivo formed enamel pellicle and in whole saliva. Scand J Dent Res. 1987 Jun;95(3):212–215. doi: 10.1111/j.1600-0722.1987.tb01833.x. [DOI] [PubMed] [Google Scholar]
- Schilling K. M., Bowen W. H. The activity of glucosyltransferase adsorbed onto saliva-coated hydroxyapatite. J Dent Res. 1988 Jan;67(1):2–8. doi: 10.1177/00220345880670010201. [DOI] [PubMed] [Google Scholar]
- Staat R. H., Langley S. D., Doyle R. J. Streptococcus mutans adherence: presumptive evidence for protein-mediated attachment followed by glucan-dependent cellular accumulation. Infect Immun. 1980 Feb;27(2):675–681. doi: 10.1128/iai.27.2.675-681.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stein D. C., Kopec L. K., Yasbin R. E., Young F. E. Characterization of Bacillus subtilis DSM704 and its production of 1-deoxynojirimycin. Appl Environ Microbiol. 1984 Aug;48(2):280–284. doi: 10.1128/aem.48.2.280-284.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Svanberg M., Westergren G., Olsson J. Oral implantation in humans of Streptococcus mutans strains with different degrees of hydrophobicity. Infect Immun. 1984 Mar;43(3):817–821. doi: 10.1128/iai.43.3.817-821.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Houte J., Green D. B. Relationship between the concentration of bacteria in saliva and the colonization of teeth in humans. Infect Immun. 1974 Apr;9(4):624–630. doi: 10.1128/iai.9.4.624-630.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu-Yuan C. D., Tai S., Slade H. D. Properties of Streptococcus mutans grown in a synthetic medium: binding of glucosyltransferase and in vitro adherence, and binding of dextran/glucan and glycoprotein and agglutination. Infect Immun. 1979 Mar;23(3):600–608. doi: 10.1128/iai.23.3.600-608.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Houte J., Burgess R. C., Onose H. Oral implantation of human strains of Streptococcus mutans in rats fed sucrose or glucose diets. Arch Oral Biol. 1976;21(9):561–564. doi: 10.1016/0003-9969(76)90023-6. [DOI] [PubMed] [Google Scholar]
- van Houte J., Duchin S. Streptococcus mutans in the mouths of children with congenital sucrase deficiency. Arch Oral Biol. 1975 Nov;20(11):771–773. doi: 10.1016/0003-9969(75)90050-3. [DOI] [PubMed] [Google Scholar]



