Abstract
Receptor recognition of pertussis toxin is mediated by the B oligomer consisting of subunits S2, S3, 2xS4, and S5. One possible way to interfere with toxin action would be the inhibition of recognition and binding of the cellular receptor(s) by preformed toxin-directed antipeptide antibodies. A prerequisite for this approach is the localization of linear antigenic determinants followed by the identification of inhibitory epitopes. Anti-S2 peptide antibodies have been shown to inhibit binding of the holotoxin to in vitro model receptor systems. For the elucidation of linear antigenic and immunogenic determinants harbored in the S3 subunit, synthetic peptides corresponding to selected linear amino acid sequences of S3 have been prepared and used to raise peptide-specific antibodies in rabbits. All peptides elicited a strong homologous response. Four synthetic peptides reacting with anti-pertussis toxin antibodies (R36-51, R87-95, R134-150, and R147-160) have been identified. Seven synthetic peptides (R1-12, R12-23, R14-29m, R36-51, R95-107, R134-150, and R164-178) induced antibodies recognizing pertussis toxin. Thus, these segments correspond to linear antigenic determinants. Analogous to the S2 subunit, the N terminus of S3 proved to be immunorecessive in the native toxin. The highly homologous S2 subunit was only bound strongly in Western blotting (immunoblotting) by antiserum directed at peptide R164-178, which is identical in the S2 and S3 subunits. A weak recognition of S2 in Western blotting was observed with anti-R95-107 antiserum. The ability of affinity-purified anti-S3 peptide antibodies to interfere with pertussis toxin binding was investigated by hemagglutination of goose erythrocytes as a model receptor system for S3-mediated receptor recognition. Antipeptide antibodies directed at R1-12, R12-23, R14-29m, and R36-51 inhibited hemagglutination of goose erythrocytes. This indicates that the corresponding antigenic regions in the S3 subunit are associated with the formation of the receptor binding domain. Inhibition of B-oligomer-mediated pertussis toxin binding to cellular receptors by preformed antipeptide antibodies of sufficient affinity should not only block the detrimental effects of the S1 subunits, but also interfere with the mitogenic effects attributed to the B oligomer.
Full text
PDF

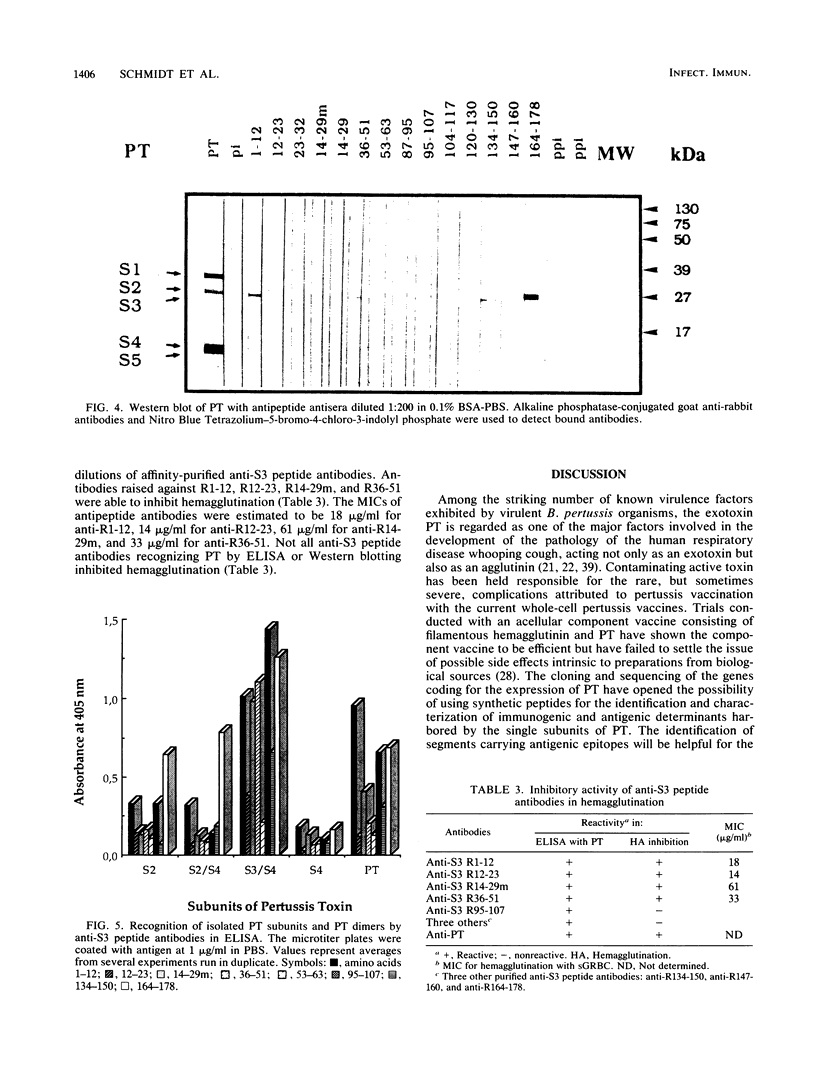


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arciniega J. L., Burns D. L., Garcia-Ortigoza E., Manclark C. R. Immune response to the B oligomer of pertussis toxin. Infect Immun. 1987 May;55(5):1132–1136. doi: 10.1128/iai.55.5.1132-1136.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Black W. J., Munoz J. J., Peacock M. G., Schad P. A., Cowell J. L., Burchall J. J., Lim M., Kent A., Steinman L., Falkow S. ADP-ribosyltransferase activity of pertussis toxin and immunomodulation by Bordetella pertussis. Science. 1988 Apr 29;240(4852):656–659. doi: 10.1126/science.2896387. [DOI] [PubMed] [Google Scholar]
- Brennan M. J., David J. L., Kenimer J. G., Manclark C. R. Lectin-like binding of pertussis toxin to a 165-kilodalton Chinese hamster ovary cell glycoprotein. J Biol Chem. 1988 Apr 5;263(10):4895–4899. [PubMed] [Google Scholar]
- Burnette W. N. "Western blotting": electrophoretic transfer of proteins from sodium dodecyl sulfate--polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem. 1981 Apr;112(2):195–203. doi: 10.1016/0003-2697(81)90281-5. [DOI] [PubMed] [Google Scholar]
- Chou P. Y., Fasman G. D. Empirical predictions of protein conformation. Annu Rev Biochem. 1978;47:251–276. doi: 10.1146/annurev.bi.47.070178.001343. [DOI] [PubMed] [Google Scholar]
- Gilman A. G. G proteins and dual control of adenylate cyclase. Cell. 1984 Mar;36(3):577–579. doi: 10.1016/0092-8674(84)90336-2. [DOI] [PubMed] [Google Scholar]
- Gross R., Aricò B., Rappuoli R. Genetics of pertussis toxin. Mol Microbiol. 1989 Jan;3(1):119–124. doi: 10.1111/j.1365-2958.1989.tb00111.x. [DOI] [PubMed] [Google Scholar]
- Hewlett E. L., Cowell J. L. Evaluation of the mouse model for study of encephalopathy in pertussis vaccine recipients. Infect Immun. 1989 Mar;57(3):661–663. doi: 10.1128/iai.57.3.661-663.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hopp T. P., Woods K. R. Prediction of protein antigenic determinants from amino acid sequences. Proc Natl Acad Sci U S A. 1981 Jun;78(6):3824–3828. doi: 10.1073/pnas.78.6.3824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaiser E., Colescott R. L., Bossinger C. D., Cook P. I. Color test for detection of free terminal amino groups in the solid-phase synthesis of peptides. Anal Biochem. 1970 Apr;34(2):595–598. doi: 10.1016/0003-2697(70)90146-6. [DOI] [PubMed] [Google Scholar]
- Kyte J., Doolittle R. F. A simple method for displaying the hydropathic character of a protein. J Mol Biol. 1982 May 5;157(1):105–132. doi: 10.1016/0022-2836(82)90515-0. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Locht C., Barstad P. A., Coligan J. E., Mayer L., Munoz J. J., Smith S. G., Keith J. M. Molecular cloning of pertussis toxin genes. Nucleic Acids Res. 1986 Apr 25;14(8):3251–3261. doi: 10.1093/nar/14.8.3251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Locht C., Keith J. M. Pertussis toxin gene: nucleotide sequence and genetic organization. Science. 1986 Jun 6;232(4755):1258–1264. doi: 10.1126/science.3704651. [DOI] [PubMed] [Google Scholar]
- Melton A. R., Weiss A. A. Environmental regulation of expression of virulence determinants in Bordetella pertussis. J Bacteriol. 1989 Nov;171(11):6206–6212. doi: 10.1128/jb.171.11.6206-6212.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monack D., Munoz J. J., Peacock M. G., Black W. J., Falkow S. Expression of pertussis toxin correlates with pathogenesis in Bordetella species. J Infect Dis. 1989 Feb;159(2):205–210. doi: 10.1093/infdis/159.2.205. [DOI] [PubMed] [Google Scholar]
- Montecucco C., Tomasi M., Schiavo G., Rappuoli R. Hydrophobic photolabelling of pertussis toxin subunits interacting with lipids. FEBS Lett. 1986 Jan 6;194(2):301–304. doi: 10.1016/0014-5793(86)80105-3. [DOI] [PubMed] [Google Scholar]
- Munoz J. J., Peacock M. G., Hadlow W. J. Anaphylaxis or so-called encephalopathy in mice sensitized to an antigen with the aid of pertussigen (pertussis toxin). Infect Immun. 1987 Apr;55(4):1004–1008. doi: 10.1128/iai.55.4.1004-1008.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munoz J. J., Peacock M. G. Role of pertussigen (pertussis toxin) on the mouse protective activity of vaccines made from Bordetella species. Microbiol Immunol. 1989;33(4):341–355. doi: 10.1111/j.1348-0421.1989.tb01982.x. [DOI] [PubMed] [Google Scholar]
- Nencioni L., Pizza M., Bugnoli M., De Magistris T., Di Tommaso A., Giovannoni F., Manetti R., Marsili I., Matteucci G., Nucci D. Characterization of genetically inactivated pertussis toxin mutants: candidates for a new vaccine against whooping cough. Infect Immun. 1990 May;58(5):1308–1315. doi: 10.1128/iai.58.5.1308-1315.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nicosia A., Bartoloni A., Perugini M., Rappuoli R. Expression and immunological properties of the five subunits of pertussis toxin. Infect Immun. 1987 Apr;55(4):963–967. doi: 10.1128/iai.55.4.963-967.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nicosia A., Perugini M., Franzini C., Casagli M. C., Borri M. G., Antoni G., Almoni M., Neri P., Ratti G., Rappuoli R. Cloning and sequencing of the pertussis toxin genes: operon structure and gene duplication. Proc Natl Acad Sci U S A. 1986 Jul;83(13):4631–4635. doi: 10.1073/pnas.83.13.4631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oda M., Cowell J. L., Burstyn D. G., Manclark C. R. Protective activities of the filamentous hemagglutinin and the lymphocytosis-promoting factor of Bordetella pertussis in mice. J Infect Dis. 1984 Dec;150(6):823–833. doi: 10.1093/infdis/150.6.823. [DOI] [PubMed] [Google Scholar]
- Pittman M., Furman B. L., Wardlaw A. C. Bordetella pertussis respiratory tract infection in the mouse: pathophysiological responses. J Infect Dis. 1980 Jul;142(1):56–66. doi: 10.1093/infdis/142.1.56. [DOI] [PubMed] [Google Scholar]
- Pittman M. The concept of pertussis as a toxin-mediated disease. Pediatr Infect Dis. 1984 Sep-Oct;3(5):467–486. doi: 10.1097/00006454-198409000-00019. [DOI] [PubMed] [Google Scholar]
- Pizza M., Covacci A., Bartoloni A., Perugini M., Nencioni L., De Magistris M. T., Villa L., Nucci D., Manetti R., Bugnoli M. Mutants of pertussis toxin suitable for vaccine development. Science. 1989 Oct 27;246(4929):497–500. doi: 10.1126/science.2683073. [DOI] [PubMed] [Google Scholar]
- Relman D. A., Domenighini M., Tuomanen E., Rappuoli R., Falkow S. Filamentous hemagglutinin of Bordetella pertussis: nucleotide sequence and crucial role in adherence. Proc Natl Acad Sci U S A. 1989 Apr;86(8):2637–2641. doi: 10.1073/pnas.86.8.2637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robson B., Suzuki E. Conformational properties of amino acid residues in globular proteins. J Mol Biol. 1976 Nov 5;107(3):327–356. doi: 10.1016/s0022-2836(76)80008-3. [DOI] [PubMed] [Google Scholar]
- Roy C. R., Miller J. F., Falkow S. Autogenous regulation of the Bordetella pertussis bvgABC operon. Proc Natl Acad Sci U S A. 1990 May;87(10):3763–3767. doi: 10.1073/pnas.87.10.3763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sato H., Sato Y. Bordetella pertussis infection in mice: correlation of specific antibodies against two antigens, pertussis toxin, and filamentous hemagglutinin with mouse protectivity in an intracerebral or aerosol challenge system. Infect Immun. 1984 Nov;46(2):415–421. doi: 10.1128/iai.46.2.415-421.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmidt M. A., Schmidt W. Inhibition of pertussis toxin binding to model receptors by antipeptide antibodies directed at an antigenic domain of the S2 subunit. Infect Immun. 1989 Dec;57(12):3828–3833. doi: 10.1128/iai.57.12.3828-3833.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmidt W., Schmidt M. A. Mapping of linear B-cell epitopes of the S2 subunit of pertussis toxin. Infect Immun. 1989 Feb;57(2):438–445. doi: 10.1128/iai.57.2.438-445.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinman L., Weiss A., Adelman N., Lim M., Zuniga R., Oehlert J., Hewlett E., Falkow S. Pertussis toxin is required for pertussis vaccine encephalopathy. Proc Natl Acad Sci U S A. 1985 Dec;82(24):8733–8736. doi: 10.1073/pnas.82.24.8733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tamura M., Nogimori K., Murai S., Yajima M., Ito K., Katada T., Ui M., Ishii S. Subunit structure of islet-activating protein, pertussis toxin, in conformity with the A-B model. Biochemistry. 1982 Oct 26;21(22):5516–5522. doi: 10.1021/bi00265a021. [DOI] [PubMed] [Google Scholar]
- Tamura M., Nogimori K., Yajima M., Ase K., Ui M. A role of the B-oligomer moiety of islet-activating protein, pertussis toxin, in development of the biological effects on intact cells. J Biol Chem. 1983 Jun 10;258(11):6756–6761. [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiss A. A., Hewlett E. L., Myers G. A., Falkow S. Pertussis toxin and extracytoplasmic adenylate cyclase as virulence factors of Bordetella pertussis. J Infect Dis. 1984 Aug;150(2):219–222. doi: 10.1093/infdis/150.2.219. [DOI] [PubMed] [Google Scholar]
- Weiss A. A., Hewlett E. L. Virulence factors of Bordetella pertussis. Annu Rev Microbiol. 1986;40:661–686. doi: 10.1146/annurev.mi.40.100186.003305. [DOI] [PubMed] [Google Scholar]



