Abstract
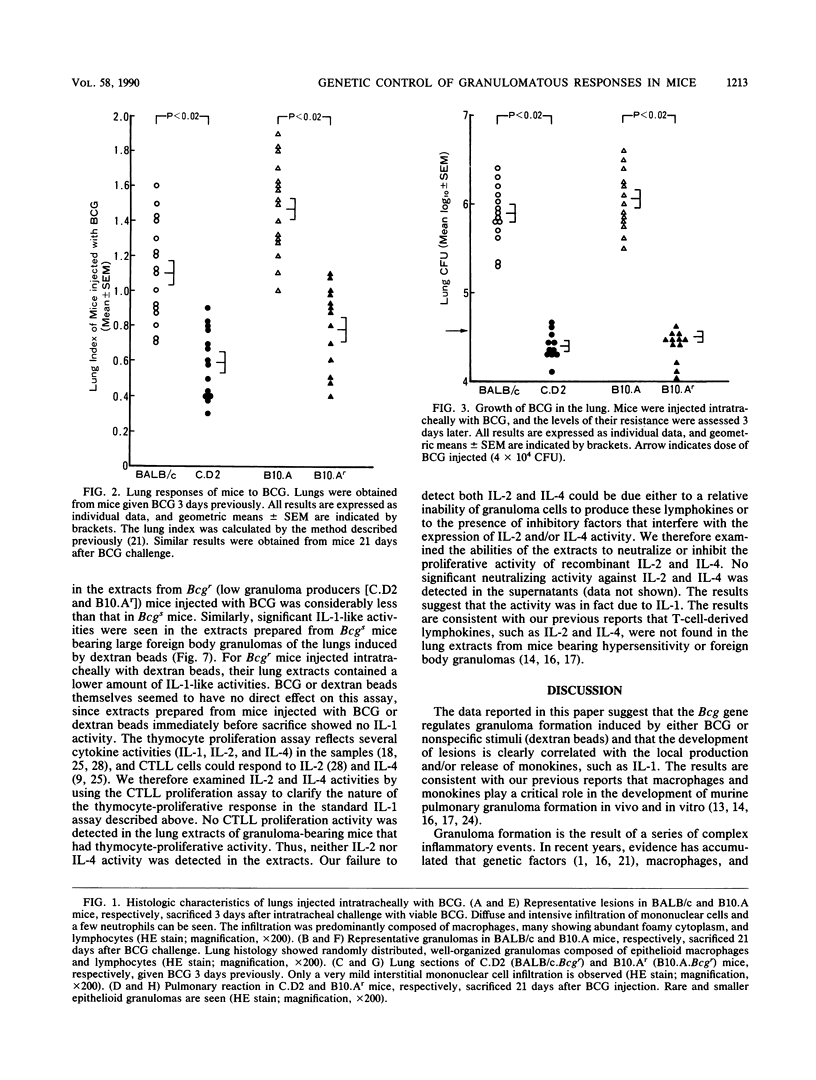
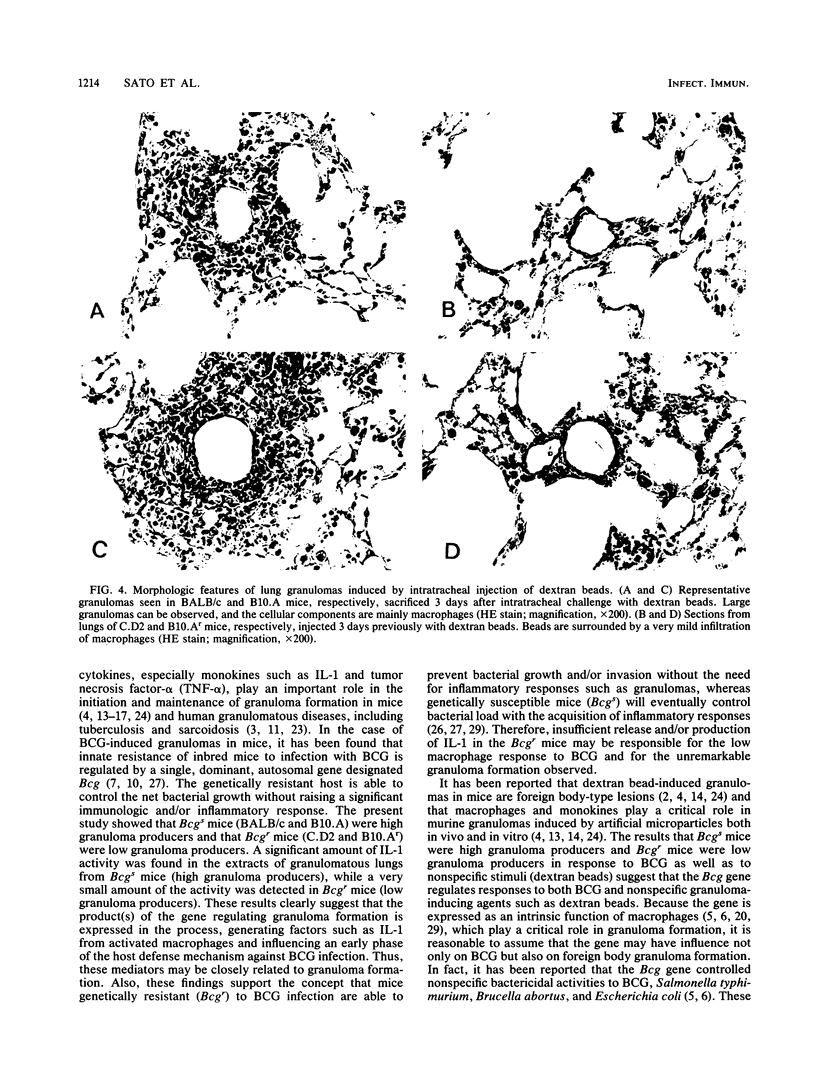
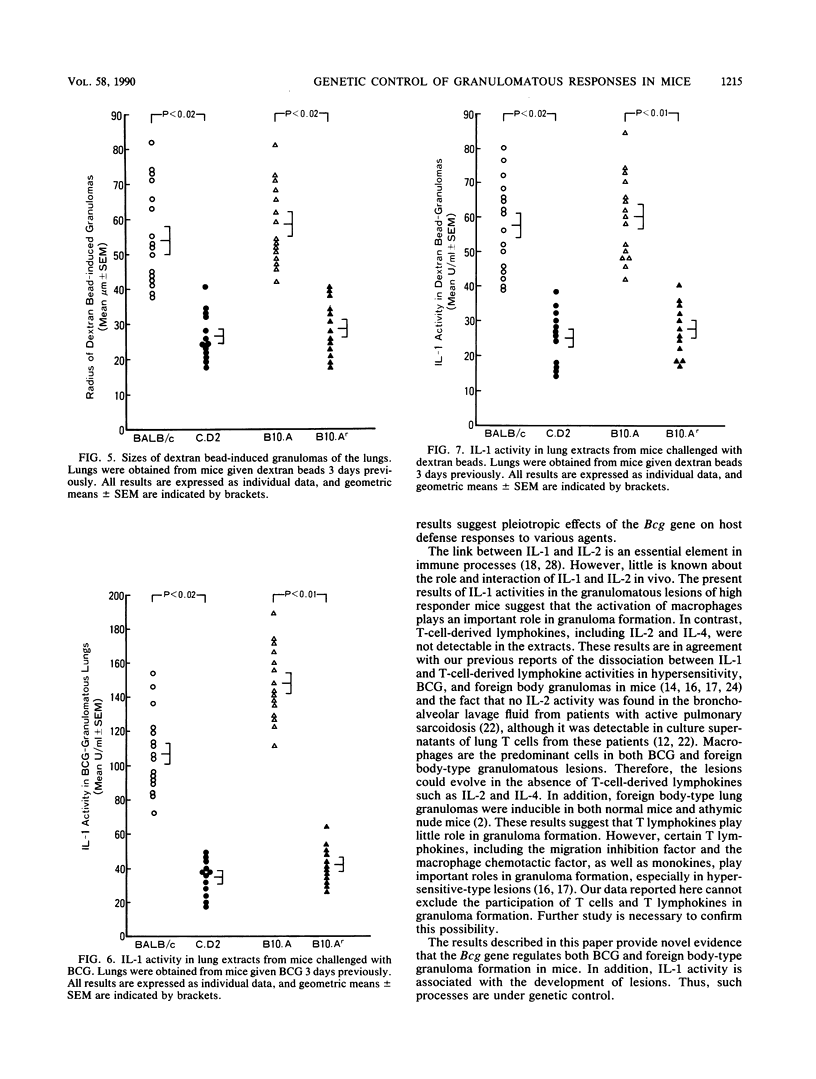
Natural bactericidal resistance to Mycobacterium bovis BCG is under the control of a single gene, designated Bcg. Lung granuloma formation in susceptible (Bcgs) and resistant (Bcgr) mice was studied in two sets of Bcg-congenic systems, namely, the BALB/c (Bcgs)-C.D2 (BALB/c.Bcgr) pair and the B10.A (Bcgs)-B10.Ar (Bcgr) pair, by using BCG as well as foreign body granuloma-inducing agents (dextran beads). Large granulomas of the lung induced by the intratracheal instillation of either BCG or dextran beads developed in Bcgs mice. In contrast, minimal inflammation was produced in Bcgr mice given BCG or dextran beads. Aqueous extracts prepared from pulmonary granuloma lesions induced by Bcgs mice by either BCG or dextran beads contained high levels of interleukin-1 (IL-1) activity but not interleukin-2 (IL-2) or interleukin-4 (IL-4) activity. Very low IL-1 activity was detected in extracts from Bcgr mice injected with BCG and dextran beads. The activity of IL-1 was correlated closely with the activity and size of the granulomatous inflammation in mice. These results suggest that pleiotropic effects of the Bcg gene are involved in the development of granulomas induced by either BCG or nonspecific foreign body agents (dextran beads) and that monokines participate in granuloma formation.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Allen E. M., Moore V. L., Stevens J. O. Strain variation in BCG-induced chronic pulmonary inflammation in mice. I. Basic model and possible genetic control by non-H-2 genes. J Immunol. 1977 Jul;119(1):343–347. [PubMed] [Google Scholar]
- Allred D. C., Kobayashi K., Yoshida T. Anergy-like immunosuppression in mice bearing pulmonary foreign-body granulomatous inflammation. Am J Pathol. 1985 Dec;121(3):466–473. [PMC free article] [PubMed] [Google Scholar]
- Bachwich P. R., Lynch J. P., 3rd, Larrick J., Spengler M., Kunkel S. L. Tumor necrosis factor production by human sarcoid alveolar macrophages. Am J Pathol. 1986 Dec;125(3):421–425. [PMC free article] [PubMed] [Google Scholar]
- Chensue S. W., Otterness I. G., Higashi G. I., Forsch C. S., Kunkel S. L. Monokine production by hypersensitivity (Schistosoma mansoni egg) and foreign body (Sephadex bead)-type granuloma macrophages. Evidence for sequential production of IL-1 and tumor necrosis factor. J Immunol. 1989 Feb 15;142(4):1281–1286. [PubMed] [Google Scholar]
- Denis M., Buschman E., Forget A., Pelletier M., Skamene E. Pleiotropic effects of the Bcg gene. II. Genetic restriction of responses to mitogens and allogeneic targets. J Immunol. 1988 Dec 1;141(11):3988–3993. [PubMed] [Google Scholar]
- Denis M., Forget A., Pelletier M., Skamene E. Pleiotropic effects of the Bcg gene. I. Antigen presentation in genetically susceptible and resistant congenic mouse strains. J Immunol. 1988 Apr 1;140(7):2395–2400. [PubMed] [Google Scholar]
- Forget A., Skamene E., Gros P., Miailhe A. C., Turcotte R. Differences in response among inbred mouse strains to infection with small doses of Mycobacterium bovis BCG. Infect Immun. 1981 Apr;32(1):42–47. doi: 10.1128/iai.32.1.42-47.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gillis S., Ferm M. M., Ou W., Smith K. A. T cell growth factor: parameters of production and a quantitative microassay for activity. J Immunol. 1978 Jun;120(6):2027–2032. [PubMed] [Google Scholar]
- Grabstein K., Eisenman J., Mochizuki D., Shanebeck K., Conlon P., Hopp T., March C., Gillis S. Purification to homogeneity of B cell stimulating factor. A molecule that stimulates proliferation of multiple lymphokine-dependent cell lines. J Exp Med. 1986 Jun 1;163(6):1405–1414. doi: 10.1084/jem.163.6.1405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gros P., Skamene E., Forget A. Genetic control of natural resistance to Mycobacterium bovis (BCG) in mice. J Immunol. 1981 Dec;127(6):2417–2421. [PubMed] [Google Scholar]
- Hunninghake G. W., Bedell G. N., Zavala D. C., Monick M., Brady M. Role of interleukin-2 release by lung T-cells in active pulmonary sarcoidosis. Am Rev Respir Dis. 1983 Oct;128(4):634–638. doi: 10.1164/arrd.1983.128.4.634. [DOI] [PubMed] [Google Scholar]
- Hunninghake G. W. Release of interleukin-1 by alveolar macrophages of patients with active pulmonary sarcoidosis. Am Rev Respir Dis. 1984 Apr;129(4):569–572. [PubMed] [Google Scholar]
- Kasahara K., Kobayashi K., Shikama Y., Yoneya I., Kaga S., Hashimoto M., Odagiri T., Soejima K., Ide H., Takahashi T. The role of monokines in granuloma formation in mice: the ability of interleukin 1 and tumor necrosis factor-alpha to induce lung granulomas. Clin Immunol Immunopathol. 1989 Jun;51(3):419–425. doi: 10.1016/0090-1229(89)90040-8. [DOI] [PubMed] [Google Scholar]
- Kasahara K., Kobayashi K., Shikama Y., Yoneya I., Soezima K., Ide H., Takahashi T. Direct evidence for granuloma-inducing activity of interleukin-1. Induction of experimental pulmonary granuloma formation in mice by interleukin-1-coupled beads. Am J Pathol. 1988 Mar;130(3):629–638. [PMC free article] [PubMed] [Google Scholar]
- Kindler V., Sappino A. P., Grau G. E., Piguet P. F., Vassalli P. The inducing role of tumor necrosis factor in the development of bactericidal granulomas during BCG infection. Cell. 1989 Mar 10;56(5):731–740. doi: 10.1016/0092-8674(89)90676-4. [DOI] [PubMed] [Google Scholar]
- Kobayashi K., Allred C., Castriotta R., Yoshida T. Strain variation of bacillus Calmette-Guerin-induced pulmonary granuloma formation is correlated with anergy and the local production of migration inhibition factor and interleukin 1. Am J Pathol. 1985 May;119(2):223–235. [PMC free article] [PubMed] [Google Scholar]
- Kobayashi K., Allred C., Cohen S., Yoshida T. Role of interleukin 1 in experimental pulmonary granuloma in mice. J Immunol. 1985 Jan;134(1):358–364. [PubMed] [Google Scholar]
- Oppenheim J. J., Shneyour A., Kook A. I. Enhancement of DNA synthesis and cAMP content of mouse thymocytes by mediator(s) derived from adherent cells. J Immunol. 1976 May;116(5):1466–1472. [PubMed] [Google Scholar]
- Orme I. M., Collins F. M. Resistance of various strains of mycobacteria to killing by activated macrophages in vivo. J Immunol. 1983 Sep;131(3):1452–1454. [PubMed] [Google Scholar]
- Pelletier M., Forget A., Bourassa D., Gros P., Skamene E. Immunopathology of BCG infection in genetically resistant and susceptible mouse strains. J Immunol. 1982 Nov;129(5):2179–2185. [PubMed] [Google Scholar]
- Pinkston P., Bitterman P. B., Crystal R. G. Spontaneous release of interleukin-2 by lung T lymphocytes in active pulmonary sarcoidosis. N Engl J Med. 1983 Apr 7;308(14):793–800. doi: 10.1056/NEJM198304073081401. [DOI] [PubMed] [Google Scholar]
- Rook G. A., Taverne J., Leveton C., Steele J. The role of gamma-interferon, vitamin D3 metabolites and tumour necrosis factor in the pathogenesis of tuberculosis. Immunology. 1987 Oct;62(2):229–234. [PMC free article] [PubMed] [Google Scholar]
- Shikama Y., Kobayashi K., Kasahara K., Kaga S., Hashimoto M., Yoneya I., Hosoda S., Soejima K., Ide H., Takahashi T. Granuloma formation by artificial microparticles in vitro. Macrophages and monokines play a critical role in granuloma formation. Am J Pathol. 1989 Jun;134(6):1189–1199. [PMC free article] [PubMed] [Google Scholar]
- Sideras P., Noma T., Honjo T. Structure and function of interleukin 4 and 5. Immunol Rev. 1988 Feb;102:189–212. doi: 10.1111/j.1600-065x.1988.tb00745.x. [DOI] [PubMed] [Google Scholar]
- Skamene E. Genetic control of susceptibility to mycobacterial infections. Rev Infect Dis. 1989 Mar-Apr;11 (Suppl 2):S394–S399. doi: 10.1093/clinids/11.supplement_2.s394. [DOI] [PubMed] [Google Scholar]
- Skamene E., Gros P., Forget A., Kongshavn P. A., St Charles C., Taylor B. A. Genetic regulation of resistance to intracellular pathogens. Nature. 1982 Jun 10;297(5866):506–509. doi: 10.1038/297506a0. [DOI] [PubMed] [Google Scholar]
- Smith K. A., Ruscetti F. W. T-cell growth factor and the culture of cloned functional T cells. Adv Immunol. 1981;31:137–175. doi: 10.1016/s0065-2776(08)60920-7. [DOI] [PubMed] [Google Scholar]
- Stach J. L., Gros P., Forget A., Skamene E. Phenotypic expression of genetically-controlled natural resistance to Mycobacterium bovis (BCG). J Immunol. 1984 Feb;132(2):888–892. [PubMed] [Google Scholar]




