Abstract
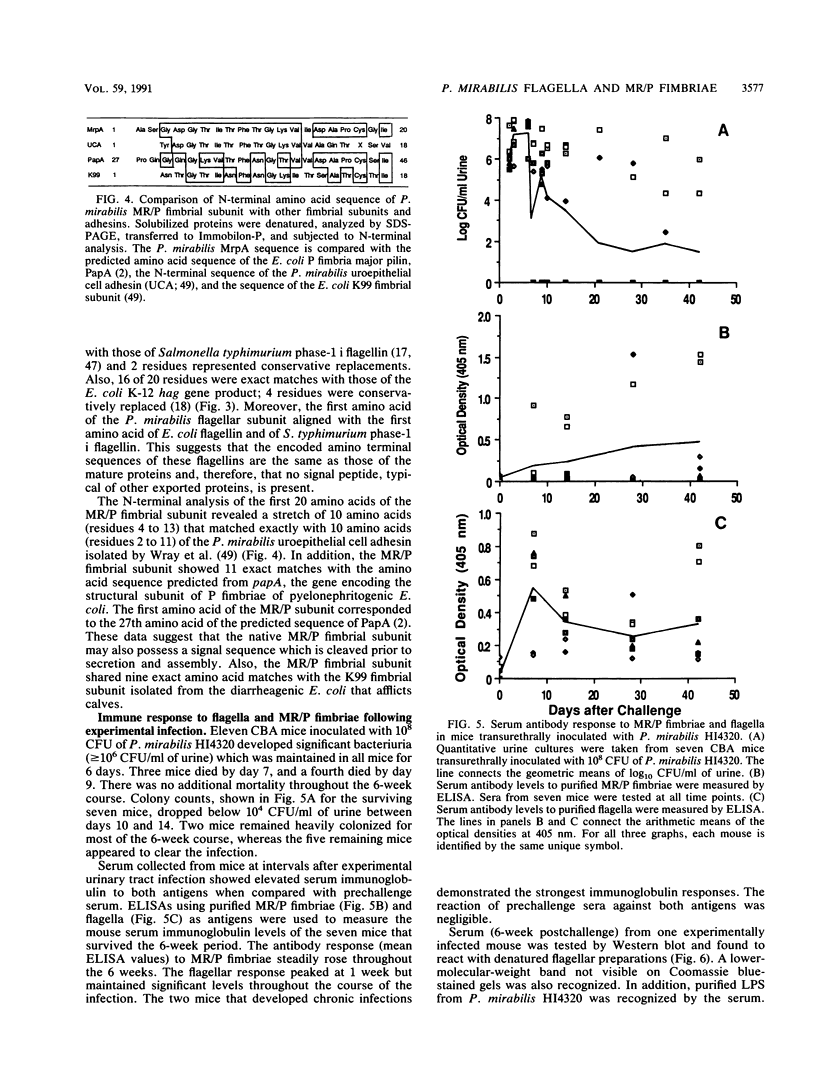
Urinary tract infection with Proteus mirabilis may lead to serious complications, including cystitis, acute pyelonephritis, fever, bacteremia, and death. In addition to the production of hemolysin and the enzyme urease, fimbriae and flagellum-mediated motility have been postulated as virulence factors for this species. We purified mannose-resistant/proteuslike (MR/P) fimbriae and flagella from strains CFT322 and HU2450, respectively. Electron microscopy revealed highly concentrated preparations of fimbriae and flagella. Fimbrial and flagellar structural subunits were estimated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis to be 18.5 and 41 kDa, respectively. N-terminal sequencing revealed that 10 of the first 20 amino acids of the major MR/P subunit matched the sequence of the P. mirabilis uroepithelial cell adhesin N terminus and 11 of 20 amino acids matched the predicted amino acid sequence of the Escherichia coli P fimbriae structural subunit, PapA. In addition, 90 and 80% homologies were found between the first 20 amino acids of P. mirabilis flagellin and those of Salmonella typhimurium phase-1 flagellin and the E. coli hag gene product, respectively. An enzyme-linked immunosorbent assay using purified antigens showed a strong reaction between the MR/P fimbriae or flagella and sera of CBA mice challenged transurethrally with P. mirabilis. A possible role for MR/P fimbriae in the pathogenesis of urinary tract infection is supported by (i) a strong immune response to the antigen in experimentally infected animals, (ii) amino acid sequence similarity to other enteric surface structure, and (iii) our previously reported observation that MR/P fimbriae are expressed preferentially as the sole fimbrial type in human pyelonephritis isolates.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adegbola R. A., Old D. C., Senior B. W. The adhesins and fimbriae of Proteus mirabilis strains associated with high and low affinity for the urinary tract. J Med Microbiol. 1983 Nov;16(4):427–431. doi: 10.1099/00222615-16-4-427. [DOI] [PubMed] [Google Scholar]
- BRAUDE A. I., SIEMIENSKI J. Role of bacterial urease in experimental pyelonephritis. J Bacteriol. 1960 Aug;80:171–179. doi: 10.1128/jb.80.2.171-179.1960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Batteiger B., Newhall W. J., 5th, Jones R. B. The use of Tween 20 as a blocking agent in the immunological detection of proteins transferred to nitrocellulose membranes. J Immunol Methods. 1982 Dec 30;55(3):297–307. doi: 10.1016/0022-1759(82)90089-8. [DOI] [PubMed] [Google Scholar]
- Bisset K. A., Douglas C. W. A continuous study of morphological phase in the swarm of Proteus. J Med Microbiol. 1976 May;9(2):229–231. doi: 10.1099/00222615-9-2-229. [DOI] [PubMed] [Google Scholar]
- Båga M., Normark S., Hardy J., O'Hanley P., Lark D., Olsson O., Schoolnik G., Falkow S. Nucleotide sequence of the papA gene encoding the Pap pilus subunit of human uropathogenic Escherichia coli. J Bacteriol. 1984 Jan;157(1):330–333. doi: 10.1128/jb.157.1.330-333.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eriksson S., Zbornik J., Dahnsjö H., Erlanson P., Kahlmeter O., Fritz H., Bauer C. A. The combination of pivampicillin and pivmecillinam versus pivampicillin alone in the treatment of acute pyelonephritis. Scand J Infect Dis. 1986;18(5):431–438. doi: 10.3109/00365548609032360. [DOI] [PubMed] [Google Scholar]
- File T. M., Jr, Tan J. S., Salstrom S. J., Johnson L. Timentin versus piperacillin in the therapy of serious urinary tract infections. Am J Med. 1985 Nov 29;79(5B):91–95. doi: 10.1016/0002-9343(85)90138-x. [DOI] [PubMed] [Google Scholar]
- Fowler J. E., Jr, Stamey T. A. Studies of introital colonization in women with recurrent urinary infections. X. Adhesive properties of Escherichia coli and Proteus mirabilis: lack of correlation with urinary pathogenicity. J Urol. 1978 Sep;120(3):315–318. doi: 10.1016/s0022-5347(17)57152-9. [DOI] [PubMed] [Google Scholar]
- Griffith D. P., Musher D. M., Itin C. Urease. The primary cause of infection-induced urinary stones. Invest Urol. 1976 Mar;13(5):346–350. [PubMed] [Google Scholar]
- Harmon R. C., Rutherford R. L., Wu H. M., Collins M. S. Monoclonal antibody-mediated protection and neutralization of motility in experimental Proteus mirabilis infection. Infect Immun. 1989 Jul;57(7):1936–1941. doi: 10.1128/iai.57.7.1936-1941.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Homma M., Fujita H., Yamaguchi S., Iino T. Regions of Salmonella typhimurium flagellin essential for its polymerization and excretion. J Bacteriol. 1987 Jan;169(1):291–296. doi: 10.1128/jb.169.1.291-296.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inzana T. J., Iritani B., Gogolewski R. P., Kania S. A., Corbeil L. B. Purification and characterization of lipooligosaccharides from four strains of "Haemophilus somnus". Infect Immun. 1988 Nov;56(11):2830–2837. doi: 10.1128/iai.56.11.2830-2837.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson D. E., Lockatell C. V., Hall-Craigs M., Mobley H. L., Warren J. W. Uropathogenicity in rats and mice of Providencia stuartii from long-term catheterized patients. J Urol. 1987 Sep;138(3):632–635. doi: 10.1016/s0022-5347(17)43287-3. [DOI] [PubMed] [Google Scholar]
- Johnson J. R. Virulence factors in Escherichia coli urinary tract infection. Clin Microbiol Rev. 1991 Jan;4(1):80–128. doi: 10.1128/cmr.4.1.80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones B. D., Lockatell C. V., Johnson D. E., Warren J. W., Mobley H. L. Construction of a urease-negative mutant of Proteus mirabilis: analysis of virulence in a mouse model of ascending urinary tract infection. Infect Immun. 1990 Apr;58(4):1120–1123. doi: 10.1128/iai.58.4.1120-1123.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones G. W., Isaacson R. E. Proteinaceous bacterial adhesins and their receptors. Crit Rev Microbiol. 1983;10(3):229–260. doi: 10.3109/10408418209113564. [DOI] [PubMed] [Google Scholar]
- Joys T. M. The covalent structure of the phase-1 flagellar filament protein of Salmonella typhimurium and its comparison with other flagellins. J Biol Chem. 1985 Dec 15;260(29):15758–15761. [PubMed] [Google Scholar]
- Kuwajima G. Construction of a minimum-size functional flagellin of Escherichia coli. J Bacteriol. 1988 Jul;170(7):3305–3309. doi: 10.1128/jb.170.7.3305-3309.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuwajima G., Kawagishi I., Homma M., Asaka J., Kondo E., Macnab R. M. Export of an N-terminal fragment of Escherichia coli flagellin by a flagellum-specific pathway. Proc Natl Acad Sci U S A. 1989 Jul;86(13):4953–4957. doi: 10.1073/pnas.86.13.4953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lomberg H., Larsson P., Leffler H., Svanborg-Edén C. Different binding specificities of P. mirabilis compared to E. coli. Scand J Infect Dis Suppl. 1982;33:37–42. [PubMed] [Google Scholar]
- MacLaren D. M. The significance of urease in proteus pyelonephritis: a bacteriological study. J Pathol Bacteriol. 1968 Jul;96(1):45–56. doi: 10.1002/path.1700960106. [DOI] [PubMed] [Google Scholar]
- McMichael J. C., Ou J. T. Structure of common pili from Escherichia coli. J Bacteriol. 1979 Jun;138(3):969–975. doi: 10.1128/jb.138.3.969-975.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mobley H. L., Chippendale G. R. Hemagglutinin, urease, and hemolysin production by Proteus mirabilis from clinical sources. J Infect Dis. 1990 Mar;161(3):525–530. doi: 10.1093/infdis/161.3.525. [DOI] [PubMed] [Google Scholar]
- Mobley H. L., Chippendale G. R., Swihart K. G., Welch R. A. Cytotoxicity of the HpmA hemolysin and urease of Proteus mirabilis and Proteus vulgaris against cultured human renal proximal tubular epithelial cells. Infect Immun. 1991 Jun;59(6):2036–2042. doi: 10.1128/iai.59.6.2036-2042.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mobley H. L., Hausinger R. P. Microbial ureases: significance, regulation, and molecular characterization. Microbiol Rev. 1989 Mar;53(1):85–108. doi: 10.1128/mr.53.1.85-108.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mobley H. L., Warren J. W. Urease-positive bacteriuria and obstruction of long-term urinary catheters. J Clin Microbiol. 1987 Nov;25(11):2216–2217. doi: 10.1128/jcm.25.11.2216-2217.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montie T. C., Doyle-Huntzinger D., Craven R. C., Holder I. A. Loss of virulence associated with absence of flagellum in an isogenic mutant of Pseudomonas aeruginosa in the burned-mouse model. Infect Immun. 1982 Dec;38(3):1296–1298. doi: 10.1128/iai.38.3.1296-1298.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moos M., Jr, Nguyen N. Y., Liu T. Y. Reproducible high yield sequencing of proteins electrophoretically separated and transferred to an inert support. J Biol Chem. 1988 May 5;263(13):6005–6008. [PubMed] [Google Scholar]
- Old D. C., Adegbola R. A. Antigenic relationships among type-3 fimbriae of Enterobacteriaceae revealed by immunoelectronmicroscopy. J Med Microbiol. 1985 Aug;20(1):113–121. doi: 10.1099/00222615-20-1-113. [DOI] [PubMed] [Google Scholar]
- Old D. C., Adegbola R. A. Haemagglutinins and fimbriae of Morganella, Proteus and Providencia. J Med Microbiol. 1982 Nov;15(4):551–564. doi: 10.1099/00222615-15-4-551. [DOI] [PubMed] [Google Scholar]
- Peerbooms P. G., Verweij A. M., MacLaren D. M. Investigation of the haemolytic activity of Proteus mirabilis strains. Antonie Van Leeuwenhoek. 1983 Apr;49(1):1–11. doi: 10.1007/BF00457874. [DOI] [PubMed] [Google Scholar]
- Peerbooms P. G., Verweij A. M., MacLaren D. M. Vero cell invasiveness of Proteus mirabilis. Infect Immun. 1984 Mar;43(3):1068–1071. doi: 10.1128/iai.43.3.1068-1071.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pere A., Selander R. K., Korhonen T. K. Characterization of P fimbriae on O1, O7, O75, rough, and nontypable strains of Escherichia coli. Infect Immun. 1988 May;56(5):1288–1294. doi: 10.1128/iai.56.5.1288-1294.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sareneva T., Holthöfer H., Korhonen T. K. Tissue-binding affinity of Proteus mirabilis fimbriae in the human urinary tract. Infect Immun. 1990 Oct;58(10):3330–3336. doi: 10.1128/iai.58.10.3330-3336.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Senior B. W., Albrechtsen M., Kerr M. A. Proteus mirabilis strains of diverse type have IgA protease activity. J Med Microbiol. 1987 Sep;24(2):175–180. doi: 10.1099/00222615-24-2-175. [DOI] [PubMed] [Google Scholar]
- Setia U., Serventi I., Lorenz P. Bacteremia in a long-term care facility. Spectrum and mortality. Arch Intern Med. 1984 Aug;144(8):1633–1635. [PubMed] [Google Scholar]
- Silverblatt F. J. Host-parasite interaction in the rat renal pelvis: a possible role for pili in the pathogenesis of pyelonephritis. J Exp Med. 1974 Dec 1;140(6):1696–1711. doi: 10.1084/jem.140.6.1696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silverblatt F. J., Ofek I. Influence of pili on the virulence of Proteus mirabilis in experimental hematogenous pyelonephritis. J Infect Dis. 1978 Nov;138(5):664–667. doi: 10.1093/infdis/138.5.664. [DOI] [PubMed] [Google Scholar]
- Tacket C. O., Maneval D. R., Levine M. M. Purification, morphology, and genetics of a new fimbrial putative colonization factor of enterotoxigenic Escherichia coli O159:H4. Infect Immun. 1987 May;55(5):1063–1069. doi: 10.1128/iai.55.5.1063-1069.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voller A., Bidwell D., Huldt G., Engvall E. A microplate method of enzyme-linked immunosorbent assay and its application to malaria. Bull World Health Organ. 1974;51(2):209–211. [PMC free article] [PubMed] [Google Scholar]
- Warren J. W., Tenney J. H., Hoopes J. M., Muncie H. L., Anthony W. C. A prospective microbiologic study of bacteriuria in patients with chronic indwelling urethral catheters. J Infect Dis. 1982 Dec;146(6):719–723. doi: 10.1093/infdis/146.6.719. [DOI] [PubMed] [Google Scholar]
- Wei L. N., Joys T. M. Covalent structure of three phase-1 flagellar filament proteins of Salmonella. J Mol Biol. 1985 Dec 20;186(4):791–803. doi: 10.1016/0022-2836(85)90397-3. [DOI] [PubMed] [Google Scholar]
- Wray S. K., Hull S. I., Cook R. G., Barrish J., Hull R. A. Identification and characterization of a uroepithelial cell adhesin from a uropathogenic isolate of Proteus mirabilis. Infect Immun. 1986 Oct;54(1):43–49. doi: 10.1128/iai.54.1.43-49.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]





