Abstract
The present report is a review of data assuming an etiological relationship between pelvic inflammatory disease (PID) and Mycoplasma hominis. Thus the organism can be isolated from the vagina/cervix more frequently in PID patients than in any other clinical group, i.e., in half to three-fourths of all such cases. One-fourth of PID patients develop a significant antibody response to M. hominis during the course of the disease. The antibody response can be detected by indirect hemagglutination tests. Grivet monkeys infected experimentally with M. hominis develop PID, predominantly parametritis; the infection seems to spread via lymphatics to the parametria. These animals develop a significant antibody response. The animals, like naturally infected women, develop a marked increase in the serum level of IgM. In tissue cell cultures of human fallopian tubes experimentally infected with M. hominis, a decrease of the mucociliary wave activity occurs. So far, few clinical data support an etiological role for Ureaplasma urealyticum in PID. In grivet monkeys, the organism does not produce PID.
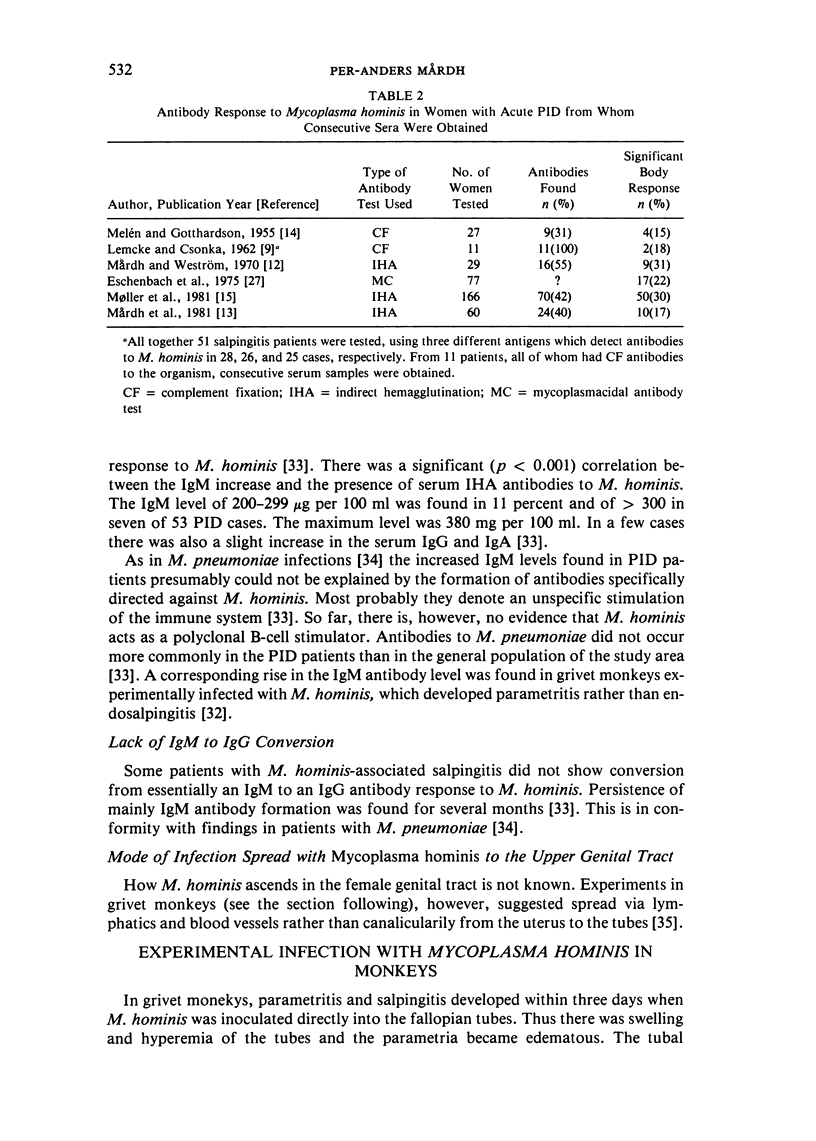
Full text
PDF






Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Braun P., Besdine R. Tuboovarian abscess with recovery of T-mycoplasma. Am J Obstet Gynecol. 1973 Nov 15;117(6):861–862. doi: 10.1016/0002-9378(73)90508-5. [DOI] [PubMed] [Google Scholar]
- DIENES L., ROPES M. W. The role of pleuropneumonia-like organisms in genitourinary and joint diseases. N Engl J Med. 1948 Apr 8;238(15):509–515. doi: 10.1056/NEJM194804082381505. [DOI] [PubMed] [Google Scholar]
- EDWARD D. G., FREUNDT E. A. The classification and nomenclature of organisms of the pleuropneumonia group. J Gen Microbiol. 1956 Feb;14(1):197–207. doi: 10.1099/00221287-14-1-197. [DOI] [PubMed] [Google Scholar]
- Eschenbach D. A., Buchanan T. M., Pollock H. M., Forsyth P. S., Alexander E. R., Lin J. S., Wang S. P., Wentworth B. B., MacCormack W. M., Holmes K. K. Polymicrobial etiology of acute pelvic inflammatory disease. N Engl J Med. 1975 Jul 24;293(4):166–171. doi: 10.1056/NEJM197507242930403. [DOI] [PubMed] [Google Scholar]
- FREUNDT E. A. The occurrence of micromyces (pleuropneumonia-like organisms) in the female genito-urinary tract; relation to the pH and general flora of the vagina. Acta Pathol Microbiol Scand. 1953;32(4):468–480. doi: 10.1111/j.1699-0463.1953.tb00275.x. [DOI] [PubMed] [Google Scholar]
- GOTTHARDSON A., MELEN B. Isolation of pleuropneumonia-like organisms from ovarian abscesses. Acta Pathol Microbiol Scand. 1953;33(3):291–293. doi: 10.1111/j.1699-0463.1953.tb01524.x. [DOI] [PubMed] [Google Scholar]
- Hirth R. S., Nielsen S. W., Plastridge W. N. Bovine salpingo-oophoritis produced with semen containing a Mycoplasma. Pathol Vet. 1966;3(6):616–632. doi: 10.1177/030098586600300604. [DOI] [PubMed] [Google Scholar]
- Hjelm E., Jonsell G., Linglöf T., Mårdh P. A., Møller B., Sedin G. Meningitis in a newborn infant caused by Mycoplasma hominis. Acta Paediatr Scand. 1980 May;69(3):415–418. doi: 10.1111/j.1651-2227.1980.tb07103.x. [DOI] [PubMed] [Google Scholar]
- Hollingdale M. R., Lemcke R. M. Antigenic differences within the species Mycoplasma hominis. J Hyg (Lond) 1970 Sep;68(3):469–477. doi: 10.1017/s0022172400042376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoogendijk J. L., de Bruijne J. I., Herderscheê D. Infectie door Mycoplasma hominis bij een pasgeborene. Ned Tijdschr Geneeskd. 1965 Jul 31;109(31):1433–1434. [PubMed] [Google Scholar]
- Jacobson L., Weström L. Objectivized diagnosis of acute pelvic inflammatory disease. Diagnostic and prognostic value of routine laparoscopy. Am J Obstet Gynecol. 1969 Dec 1;105(7):1088–1098. doi: 10.1016/0002-9378(69)90132-x. [DOI] [PubMed] [Google Scholar]
- Jones D. M., Tobin B. Neonatal eye infections due to Mycoplasma hominis. Br Med J. 1968 Aug 24;3(5616):467–468. doi: 10.1136/bmj.3.5616.467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krogsgaard-Jensen A. Indirect hemagglutination with Mycoplasma antigens: effects of pH on antigen sensitization of tanned fresh and formalinized sheep erythrocytes. Appl Microbiol. 1971 Nov;22(5):756–759. doi: 10.1128/am.22.5.756-759.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LEMCKE R., CSONKA G. W. Antibodies against pleuropneumonia-like organisms in patients with salpingitis. Br J Vener Dis. 1962 Dec;38:212–217. doi: 10.1136/sti.38.4.212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MELEN B., GOTTHARDSON A. Complement fixation with human pleuropneumonia-like organisms. Acta Pathol Microbiol Scand. 1955;37(2):196–200. doi: 10.1111/j.1699-0463.1955.tb00938.x. [DOI] [PubMed] [Google Scholar]
- MUFSON M. A., LUDWIG W. M., PURCELL R. H., CATE T. R., TAYLOR-ROBINSON D., CHANOCK R. M. EXUDATIVE PHARYNGITIS FOLLOWING EXPERIMENTAL MYCOPLASMA HOMINIS: TYPE 1 INFECTION. JAMA. 1965 Jun 28;192:1146–1152. doi: 10.1001/jama.1965.03080260034010. [DOI] [PubMed] [Google Scholar]
- McCormack W. M., Almeida P. C., Bailey P. E., Grady E. M., Lee Y. H. Sexual activity and vaginal colonization with genital mycoplasmas. JAMA. 1972 Sep 18;221(12):1375–1377. [PubMed] [Google Scholar]
- Märdh P. A., Lind I., Svensson L., Weström L., Møller B. R. Antibodies to Chlamydia trachomatis, Mycoplasma hominis, and Neisseria gonorrhoeae in sera from patients with acute salpingitis. Br J Vener Dis. 1981 Apr;57(2):125–129. doi: 10.1136/sti.57.2.125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mårdh P. A. Human respiratory tract infections with mycoplasmas and their in vitro susceptibility to tetracyclines and some other antibiotics. Chemotherapy. 1975;21 (Suppl 1):47–57. doi: 10.1159/000221891. [DOI] [PubMed] [Google Scholar]
- Mårdh P. A. Increased serum levels of IgM in acute salpingitis related to the occurrence of Mycoplasma hominis. Acta Pathol Microbiol Scand B Microbiol Immunol. 1970;78(6):726–732. doi: 10.1111/j.1699-0463.1970.tb04363.x. [DOI] [PubMed] [Google Scholar]
- Mårdh P. A., Stormby N., Weström L. Mycoplasma and vaginal cytology. Acta Cytol. 1971 May-Jun;15(3):310–315. [PubMed] [Google Scholar]
- Mårdh P. A., Weström L. Antibodies to Mycoplasma hominis in patients with genital infections and in healthy controls. Br J Vener Dis. 1970 Oct;46(5):390–397. [PMC free article] [PubMed] [Google Scholar]
- Mårdh P. A., Weström L. T-mycoplasmas in the genito-urinary tract of the female. Acta Pathol Microbiol Scand B Microbiol Immunol. 1970;78(3):367–374. doi: 10.1111/j.1699-0463.1970.tb04316.x. [DOI] [PubMed] [Google Scholar]
- Mårdh P. A., Weström L. Tubal and cervical cultures in acute salpingitis with special reference to Mycoplasma hominis and T-strain mycoplasmas. Br J Vener Dis. 1970 Jun;46(3):179–186. [PMC free article] [PubMed] [Google Scholar]
- Mårdh P. A., Weström L., von Mecklenburg C., Hammar E. Studies on ciliated epithelia of the human genital tract. I. Swelling of the cilia of Fallopian tube epithelium in organ cultures infected with Mycoplasma hominis. Br J Vener Dis. 1976 Feb;52(1):52–57. doi: 10.1136/sti.52.1.52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Møller B. R. A modification of the mycoplasma serum-drop growth inhibition test. J Appl Bacteriol. 1979 Aug;47(1):97–104. doi: 10.1111/j.1365-2672.1979.tb01173.x. [DOI] [PubMed] [Google Scholar]
- Møller B. R., Black F. T., Freundt E. A. Attempts to produce gynaecological disease in grivet monkeys with Ureaplasma urealyticum. J Med Microbiol. 1981 Nov;14(4):475–478. doi: 10.1099/00222615-14-4-475. [DOI] [PubMed] [Google Scholar]
- Møller B. R. Comparison of serological tests for detection of Mycoplasma hominis antibodies in female Grivet monkeys with experimentally induced salpingitis. Acta Pathol Microbiol Scand B. 1981 Feb;89(1):7–11. [PubMed] [Google Scholar]
- Møller B. R., Freundt E. A., Black F. T., Frederiksen P. Experimental infection of the genital tract of female grivet monkeys by Mycoplasma hominis. Infect Immun. 1978 Apr;20(1):248–257. doi: 10.1128/iai.20.1.248-257.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Møller B. R., Mårdh P. A., Ahrons S., Nüssler E. Infection with Chlamydia trachomatis, Mycoplasma hominis and Neisseria gonorrhoeae in patients with acute pelvic inflammatory disease. Sex Transm Dis. 1981 Jul-Sep;8(3):198–202. [PubMed] [Google Scholar]
- Møller B. R., Mårdh P. A. Experimental salpingitis in grivet monkeys by Chlamydia trachomatis. Modes of spread of infection to the Fallopian tubes. Acta Pathol Microbiol Scand B. 1980 Apr;88(2):107–114. [PubMed] [Google Scholar]
- Purcell R. H., Wong D., Chanock R. M., Taylor-Robinson D., Canchola J., Valdesuso J. Significance of antibody to mycoplasma as measured by metabolic-inhibition techniques. Ann N Y Acad Sci. 1967 Jul 28;143(1):664–675. doi: 10.1111/j.1749-6632.1967.tb27712.x. [DOI] [PubMed] [Google Scholar]
- STOKES E. J. Human infection with pleuropneumonia-like organisms. Lancet. 1955 Feb 5;268(6858):276–279. doi: 10.1016/s0140-6736(55)90107-0. [DOI] [PubMed] [Google Scholar]
- Sacker I., Walker M., Brunell P. A. Abscess in newborn infants caused by Mycoplasma. Pediatrics. 1970 Aug;46(2):303–304. [PubMed] [Google Scholar]
- Sweet R. L., Mills J., Hadley K. W., Blumenstock E., Schachter J., Robbie M. O., Draper D. L. Use of laparoscopy to determine the microbiologic etiology of acute salpingitis. Am J Obstet Gynecol. 1979 May 1;134(1):68–74. doi: 10.1016/0002-9378(79)90798-1. [DOI] [PubMed] [Google Scholar]
- TAYLOR-ROBINSON D., LUDWIG W. M., PURCELL R. H., MUFSON M. A., CHANOCK R. M. SIGNIFICANCE OF ANTIBODY TO MYCOPLASMA HOMINIS TYPE 1 AS MEASURED BY INDIRECT HEMAGGLUTINATION. Proc Soc Exp Biol Med. 1965 Apr;118:1073–1083. doi: 10.3181/00379727-118-30049. [DOI] [PubMed] [Google Scholar]
- Weström L., Bengtsson L. P., Mårdh P. A. Incidence, trends, and risks of ectopic pregnancy in a population of women. Br Med J (Clin Res Ed) 1981 Jan 3;282(6257):15–18. doi: 10.1136/bmj.282.6257.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weström L., Bengtsson L. P., Mårdh P. A. The risk of pelvic inflammatory disease in women using intrauterine contraceptive devices as compared to non-users. Lancet. 1976 Jul 31;2(7979):221–224. doi: 10.1016/s0140-6736(76)91025-4. [DOI] [PubMed] [Google Scholar]
- Weström L. Effect of acute pelvic inflammatory disease on fertility. Am J Obstet Gynecol. 1975 Mar 1;121(5):707–713. doi: 10.1016/0002-9378(75)90477-9. [DOI] [PubMed] [Google Scholar]
- Weström L., Mårdh P. A. The effect of antibiotic therapy on mycoplasma in the female genital tract. In vitro and in vivo studies on the sensitivity of Mycoplasma hominis and T-Mycoplasmas to tetracyclines and other antibiotics. Acta Obstet Gynecol Scand. 1971;50(1):25–31. doi: 10.3109/00016347109157281. [DOI] [PubMed] [Google Scholar]


