Abstract
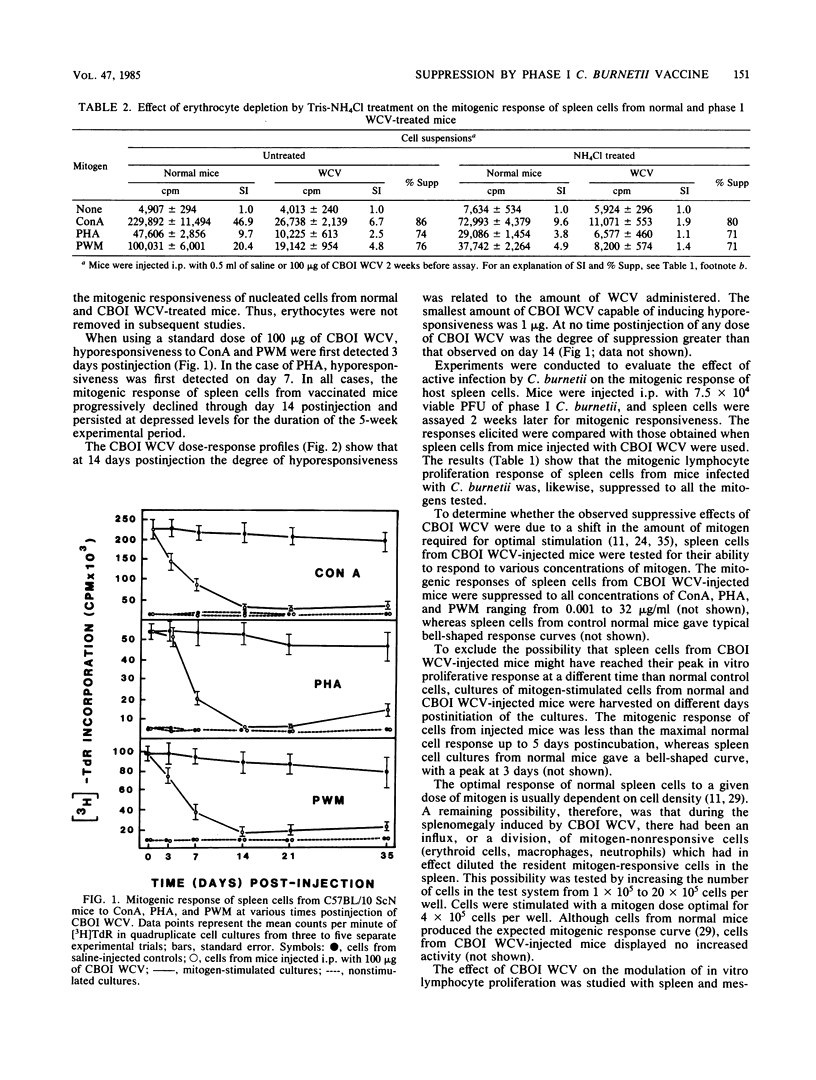
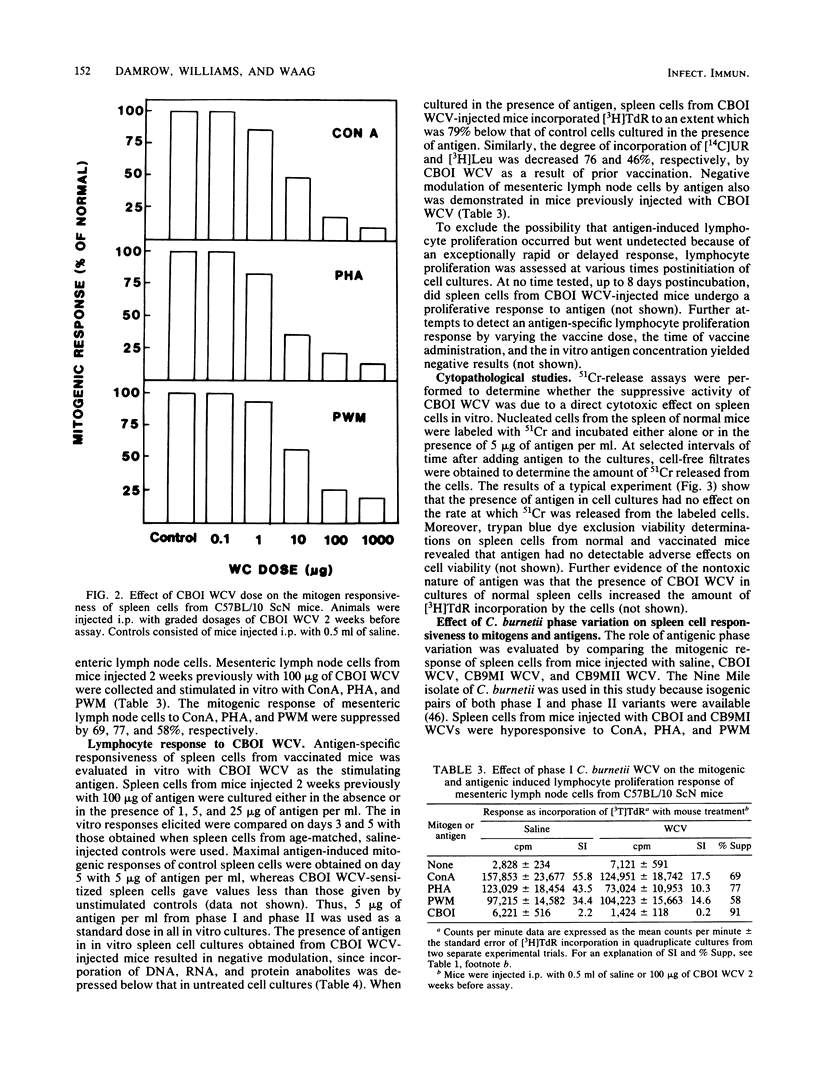
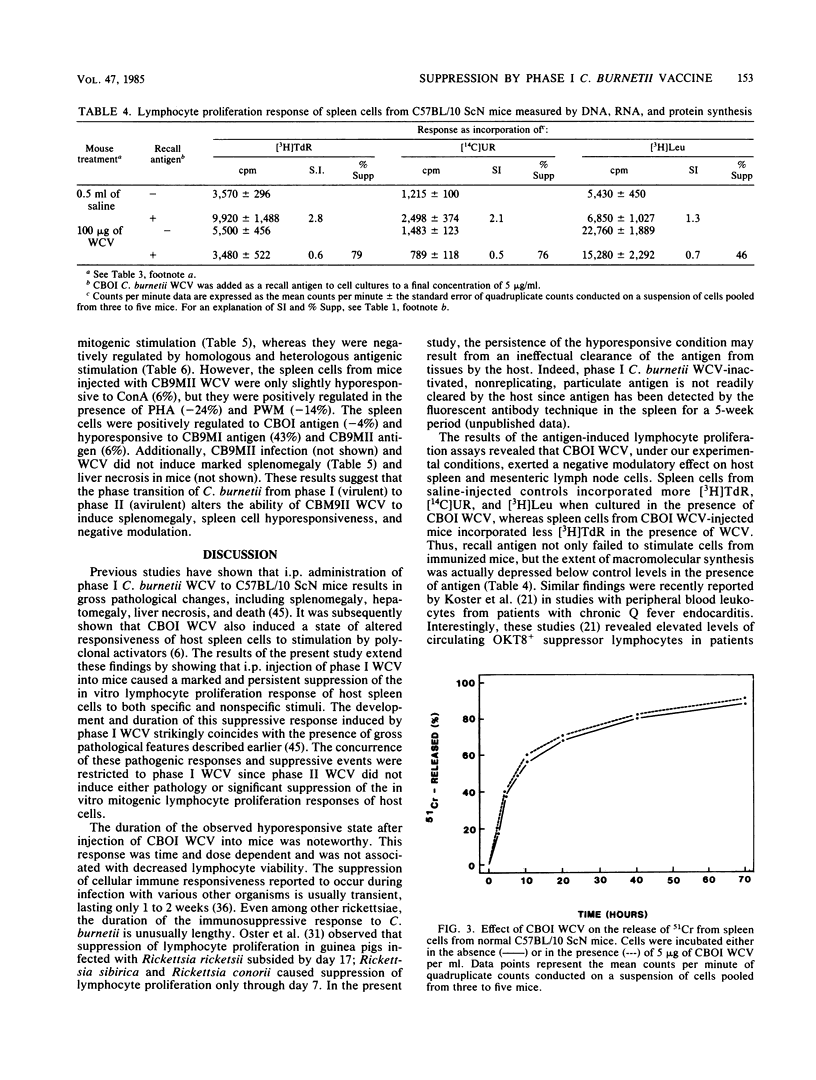
The effect of inactivated phase I and phase II Coxiella burnetii whole cell vaccine (WCV) on the response of murine spleen cells to mitogenic and antigenic stimuli was evaluated in C57BL/10 ScN endotoxin nonresponder mice with an in vitro lymphocyte proliferation assay. Intraperitoneal injection of phase I WCV into mice resulted in marked and persistent suppression of the proliferative response of spleen cells to concanavalin A, phytohemagglutinin, and pokeweed mitogen. This response was time and dose dependent and was not associated with decreased lymphocyte viability. By using a standard dose of 100 micrograms of phase I WCV, suppression of mitogenic responsiveness was first detected 3 days postinjection, attained maximum levels by day 14, and persisted for longer than 5 weeks. Suppression of mitogenic lymphocyte proliferation also was demonstrated after inoculation of animals with viable phase I organisms. The observed hyporesponsiveness of spleen cells from phase I WCV-injected animals was not either the result of a shift in the mitogenic dose optimum or due to a change in the day of in vitro peak response. Spleen cells from phase I WCV-injected mice were negatively regulated with homologous antigen. Investigation of the mechanism of action of phase I WCV, with a 51Cr-release assay, and trypan blue dye exclusion showed that phase I WCV was not directly cytolytic or cytotoxic to spleen cells from normal or vaccinated mice. Phase II WCV did not induce significant mitogenic hyporesponsiveness or negative modulation of spleen cells. These findings extend the observations of adverse host responses associated with the phase I WCV and underscore the need to develop a microbial fraction which possesses protective potency but which lacks the propensity to induce deleterious tissue reactions and immunosuppression.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BERMAN S., COLE G., LOWENTHAL J. P., GOCHENOUR R. B. Safety test for Q fever vaccine. J Bacteriol. 1960 May;79:747–751. doi: 10.1128/jb.79.5.747-751.1960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BOYSE E. A., OLD L. J., CHOUROULINKOV I. CYTOTOXIC TEST FOR DEMONSTRATION OF MOUSE ANTIBODY. Methods Med Res. 1964;10:39–47. [PubMed] [Google Scholar]
- Boyle W. An extension of the 51Cr-release assay for the estimation of mouse cytotoxins. Transplantation. 1968 Sep;6(6):761–764. doi: 10.1097/00007890-196809000-00002. [DOI] [PubMed] [Google Scholar]
- GOODMAN H. S. A general method for the quantitation of immune cyotysis. Nature. 1961 Apr 15;190:269–270. doi: 10.1038/190269a0. [DOI] [PubMed] [Google Scholar]
- Gershon R. K., Cohen P., Hencin R., Liebhaber S. A. Suppressor T cells. J Immunol. 1972 Mar;108(3):586–590. [PubMed] [Google Scholar]
- Goodwin J. S., Webb D. R. Regulation of the immune response by prostaglandins. Clin Immunol Immunopathol. 1980 Jan;15(1):106–122. doi: 10.1016/0090-1229(80)90024-0. [DOI] [PubMed] [Google Scholar]
- Hartmann K., Dutton R. W., McCarthy M. M., Mishell R. I. Cell components in the immune response. II. Cell attachment separation of immune cells. Cell Immunol. 1970 Jul;1(2):182–189. doi: 10.1016/0008-8749(70)90005-5. [DOI] [PubMed] [Google Scholar]
- Janossy G., Greaves M. F. Lymphocyte activation. I. Response of T and B lymphocytes to phytomitogens. Clin Exp Immunol. 1971 Oct;9(4):483–498. [PMC free article] [PubMed] [Google Scholar]
- Jerrells T. R., Mallavia L. P., Hinrichs D. J. Detection of long-term cellular immunity to Coxiella burneti as assayed by lymphocyte transformation. Infect Immun. 1975 Feb;11(2):280–286. doi: 10.1128/iai.11.2.280-286.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kazár J., Schramek S. Inhibition by Coxiella burnetii of ascites tumour formation in mice. Acta Virol. 1979 May;23(3):267–270. [PubMed] [Google Scholar]
- Kelly M. T. Activation of guinea pig macrophages by Q fever rickettsiae. Cell Immunol. 1977 Jan;28(1):198–205. doi: 10.1016/s0008-8749(77)80020-8. [DOI] [PubMed] [Google Scholar]
- Kimbrough R. C., 3rd, Ormsbee R. A., Peacock M., Rogers W. R., Bennetts R. W., Raaf J., Krause A., Gardner C. Q fever endocarditis in the United States. Ann Intern Med. 1979 Sep;91(3):400–402. doi: 10.7326/0003-4819-91-3-400. [DOI] [PubMed] [Google Scholar]
- Kirchner H., Holden H. T., Herberman Splenic suppressor macrophages induced in mice by injection of Corynebacterium parvum. J Immunol. 1975 Nov;115(5):1212–1216. [PubMed] [Google Scholar]
- Kishimoto R. A., Johnson J. W., Kenyon R. H., Ascher M. S., Larson E. W., Pedersen C. E., Jr Cell-mediated immune responses of guinea pigs to an inactivated phase I Coxiella burnetii vaccine. Infect Immun. 1978 Jan;19(1):194–198. doi: 10.1128/iai.19.1.194-198.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kishimoto R. A., Walker J. S. Interaction between Coxiella burnetii and guinea pig peritoneal macrophages. Infect Immun. 1976 Aug;14(2):416–421. doi: 10.1128/iai.14.2.416-421.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LACKMAN D. B., BELL E. J., BELL J. F., PICKENS E. G. Intradermal sensitivity testing in man with a purified vaccine for Q fever. Am J Public Health Nations Health. 1962 Jan;52:87–93. doi: 10.2105/ajph.52.1.87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lake W. W., Bice D., Schwartz H. J., Salvaggio J. Suppression of in vitro antigen-induced lymphocyte transformation by carrageenan, a macrophage-toxic agent. J Immunol. 1971 Dec;107(6):1745–1751. [PubMed] [Google Scholar]
- Lee K. C., Shiozawa C., Shaw A., Diener E. Requirement for accessory cells in the antibody response to T cell-independent antigens in vitro. Eur J Immunol. 1976 Jan;6(1):63–68. doi: 10.1002/eji.1830060114. [DOI] [PubMed] [Google Scholar]
- Levy R., Kaplan H. S. Impaired lymphocyte function in untreated Hodgkin's disease. N Engl J Med. 1974 Jan 24;290(4):181–186. doi: 10.1056/NEJM197401242900402. [DOI] [PubMed] [Google Scholar]
- MEIKLEJOHN G., LENNETTE E. H. Q fever in California. I. Observations on vaccination of human beings. Am J Hyg. 1950 Jul;52(1):54–64. [PubMed] [Google Scholar]
- McAdam K. P., Ryan J. L. C57BL/10/CR mice: nonresponders to activation by the lipid a moiety of bacterial lipopolysaccharide. J Immunol. 1978 Jan;120(1):249–253. [PubMed] [Google Scholar]
- McCaul T. F., Williams J. C. Developmental cycle of Coxiella burnetii: structure and morphogenesis of vegetative and sporogenic differentiations. J Bacteriol. 1981 Sep;147(3):1063–1076. doi: 10.1128/jb.147.3.1063-1076.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moorhead J. F., Connolly J. J., McFarland W. Factors affecting the reactivity of human lymphocytes in vitro. I. Cell number, duration of culture and surface area. J Immunol. 1967 Aug;99(2):413–419. [PubMed] [Google Scholar]
- Nydegger U. E., Anner R. M., Gerebtzoff A., Lambert P. H., Miescher P. A. Polymorphonuclear leukocytes stimulation by immune complexes. Assessment by nitroblue tetrazolium dye reduction. Eur J Immunol. 1973 Aug;3(8):465–470. doi: 10.1002/eji.1830030803. [DOI] [PubMed] [Google Scholar]
- ORMSBEE R. A., BELL E. J., LACKMAN D. B., TALLENT G. THE INFLUENCE OF PHASE ON THE PROTECTIVE POTENCY OF Q FEVER VACCINE. J Immunol. 1964 Mar;92:404–412. [PubMed] [Google Scholar]
- Oster C. N., Kenyon R. H., Pedersen C. E., Jr Suppression of cellular immune responses in guinea pigs infected with spotted fever group rickettsiae. Infect Immun. 1978 Nov;22(2):411–417. doi: 10.1128/iai.22.2.411-417.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pelus L. M., Strausser H. R. Indomethacin enhancement of spleen-cell responsiveness to mitogen stimulation in tumorous mice. Int J Cancer. 1976 Nov 15;18(5):653–660. doi: 10.1002/ijc.2910180514. [DOI] [PubMed] [Google Scholar]
- Pierce C. W., Kapp J. A., Wood D. D., Benacerraf B. Immune responses in vitro. X. Functions of macrophages. J Immunol. 1974 Mar;112(3):1181–1189. [PubMed] [Google Scholar]
- REED C. F., WENTWORTH B. B. Q fever studies in Ohio. J Am Vet Med Assoc. 1957 May 15;130(10):458–461. [PubMed] [Google Scholar]
- Riglar C., Cheers C. Macrophage activation during experimental murine brucellosis. II. Inhibition of in vitro lymphocyte proliferation by brucella-activated macrophages. Cell Immunol. 1980 Jan;49(1):154–167. doi: 10.1016/0008-8749(80)90065-9. [DOI] [PubMed] [Google Scholar]
- Schwab J. H. Suppression of the immune response by microorganisms. Bacteriol Rev. 1975 Jun;39(2):121–143. doi: 10.1128/br.39.2.121-143.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scott P. A., Farrell J. P. Experimental cutaneous leishmaniasis. I. Nonspecific immunodepression in BALB/c mice infected with Leishmania tropica. J Immunol. 1981 Dec;127(6):2395–2400. [PubMed] [Google Scholar]
- Sjöberg O., Andersson J., Möller G. Requirement for adherent cells in the primary and secondary immune response in vitro. Eur J Immunol. 1972 Apr;2(2):123–126. doi: 10.1002/eji.1830020206. [DOI] [PubMed] [Google Scholar]
- Strong D. M., Ahmed A. A., Thurman G. B., Sell K. W. In vitro stimulation of murine spleen cells using a microculture system and a multiple automated sample harvester. J Immunol Methods. 1973 Apr;2(3):279–291. doi: 10.1016/0022-1759(73)90054-9. [DOI] [PubMed] [Google Scholar]
- Sumner J. B., Howell S. F. Identification of Hemagglutinin of Jack Bean with Concanavalin A. J Bacteriol. 1936 Aug;32(2):227–237. doi: 10.1128/jb.32.2.227-237.1936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Svejgaard A., Platz P., Ryder L. P. HLA and disease 1982--a survey. Immunol Rev. 1983;70:193–218. doi: 10.1111/j.1600-065x.1983.tb00715.x. [DOI] [PubMed] [Google Scholar]
- Wellhausen S. R., Mansfield J. M. Characteristics of the splenic suppressor cell--target cell interaction in experimental African trypanosomiasis. Cell Immunol. 1980 Sep 1;54(2):414–424. doi: 10.1016/0008-8749(80)90221-x. [DOI] [PubMed] [Google Scholar]
- Williams J. C., Cantrell J. L. Biological and immunological properties of Coxiella burnetii vaccines in C57BL/10ScN endotoxin-nonresponder mice. Infect Immun. 1982 Mar;35(3):1091–1102. doi: 10.1128/iai.35.3.1091-1102.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams J. C., Peacock M. G., McCaul T. F. Immunological and biological characterization of Coxiella burnetii, phases I and II, separated from host components. Infect Immun. 1981 May;32(2):840–851. doi: 10.1128/iai.32.2.840-851.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]


