Abstract
Our previous studies have shown that indigenous bacteria are able to block the in vitro attachment of uropathogenic bacteria to human uroepithelial cells. In the present study, we applied the concept of competitive exclusion to an animal model. A chronic urinary tract infection was established in female rats with bacteria incorporated into agar beads injected periurethrally into the urinary bladder via a no. 3 French ureteral catheter. Five strains of uropathogenic organisms were used in the first set of experiments, and their colonization of the bladder and kidneys of the animals was confirmed up to 2 months after injection. The uropathogens stimulated an immune response, detected by serum antibodies against the uropathogens, and an inflammatory response noted in sections of the kidneys stained with hematoxylin and eosin. Using this animal model, we established the persistent adherence of bacteria in the urinary tract without the need for creation of obstruction or implantation of a foreign body. In a second set of experiments, an isolate of Lactobacillus casei GR1 taken from the urethra of a healthy woman was incorporated into agar beads, instilled within the rat bladders on day 1, and then swabbed twice weekly for 21 days onto the introitus before challenge with uropathogens instilled into the urinary bladder. In 21 of 25 animals, no uropathogenic bacteria were recovered from the bladder and kidney tissues up to 60 days after challenge, and no immune response was detected. Our results show that L. casei prevented onset of urinary tract infection in 84% of the animals tested. The lactobacilli appeared to exclude the uropathogens from colonizing the urinary tract, within the first 48 hours after challenge, and the net effect was a complete eradication of bacteria from the uroepithelium. It is hoped that the demonstration of a protective role for indigenous bacteria in preventing urinary tract infection in an animal model will lead to the application of this technology to prevent recurrent urinary tract infection in female patients.
Full text
PDF

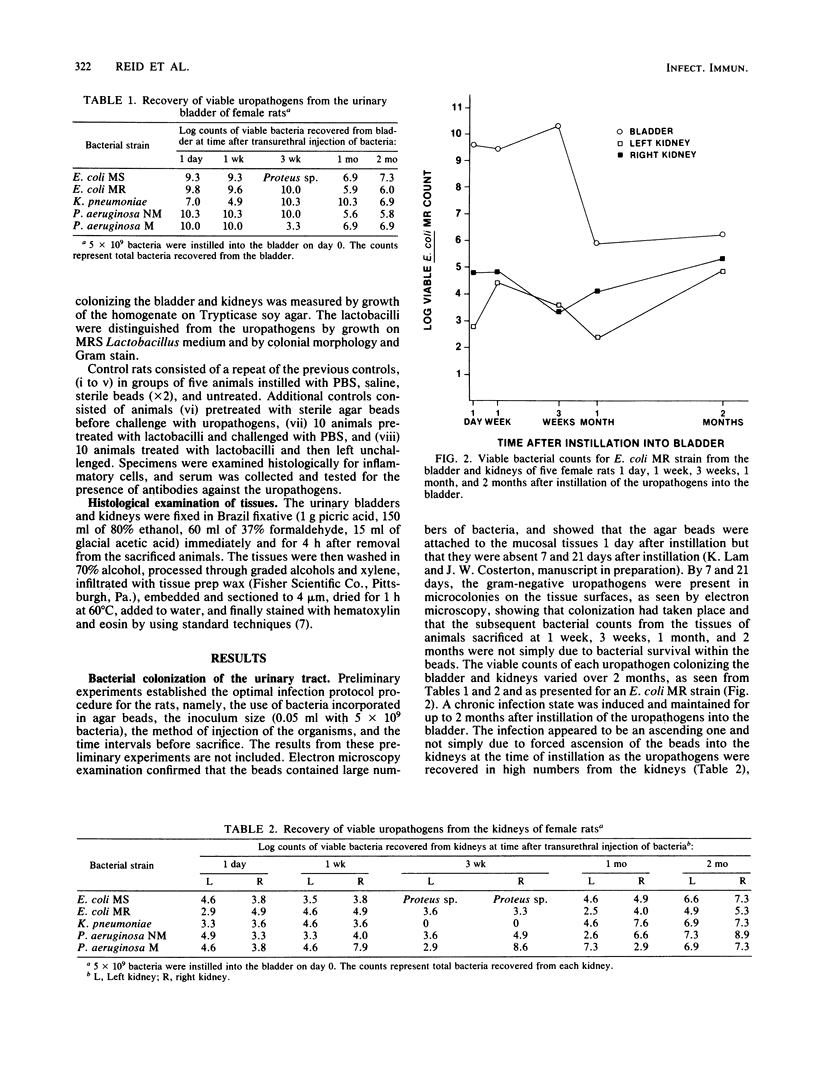


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BECK C., NECHELES H. Beneficial effects of administration of Lactobacillus acidophilus in diarrheal and other intestinal disorders. Am J Gastroenterol. 1961 May;35:522–530. [PubMed] [Google Scholar]
- Bruce A. W., Chan R. C., Pinkerton D., Morales A., Chadwick P. Adherence of gram-negative uropathogens to human uroepithelial cells. J Urol. 1983 Aug;130(2):293–298. doi: 10.1016/s0022-5347(17)51115-5. [DOI] [PubMed] [Google Scholar]
- Cash H. A., Woods D. E., McCullough B., Johanson W. G., Jr, Bass J. A. A rat model of chronic respiratory infection with Pseudomonas aeruginosa. Am Rev Respir Dis. 1979 Mar;119(3):453–459. doi: 10.1164/arrd.1979.119.3.453. [DOI] [PubMed] [Google Scholar]
- Chan R. C., Bruce A. W., Reid G. Adherence of cervical, vaginal and distal urethral normal microbial flora to human uroepithelial cells and the inhibition of adherence of gram-negative uropathogens by competitive exclusion. J Urol. 1984 Mar;131(3):596–601. doi: 10.1016/s0022-5347(17)50512-1. [DOI] [PubMed] [Google Scholar]
- Chan R. C., Bruce A. W. The influence of growth media on the morphology and in vitro adherence characteristics of gram-negative urinary pathogens. J Urol. 1983 Feb;129(2):411–417. doi: 10.1016/s0022-5347(17)52128-x. [DOI] [PubMed] [Google Scholar]
- Chan R. C., Reid G., Irvin R. T., Bruce A. W., Costerton J. W. Competitive exclusion of uropathogens from human uroepithelial cells by Lactobacillus whole cells and cell wall fragments. Infect Immun. 1985 Jan;47(1):84–89. doi: 10.1128/iai.47.1.84-89.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edén C. S., Hanson L. A., Jodal U., Lindberg U., Akerlund A. S. Variable adherence to normal human urinary-tract epithelial cells of Escherichia coli strains associated with various forms of urinary-tract infection. Lancet. 1976 Sep 4;1(7984):490–492. [PubMed] [Google Scholar]
- Fader R. C., Davis C. P. Effect of piliation on Klebsiella pneumoniae infection in rat bladders. Infect Immun. 1980 Nov;30(2):554–561. doi: 10.1128/iai.30.2.554-561.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fader R. C., Davis C. P. Klebsiella pneumoniae-induced experimental pyelitis: the effect of piliation on infectivity. J Urol. 1982 Jul;128(1):197–201. doi: 10.1016/s0022-5347(17)52817-7. [DOI] [PubMed] [Google Scholar]
- Gorbach S. L., Menda K. B., Thadepalli H., Keith L. Anaerobic microflora of the cervix in healthy women. Am J Obstet Gynecol. 1973 Dec 15;117(8):1053–1055. doi: 10.1016/0002-9378(73)90753-9. [DOI] [PubMed] [Google Scholar]
- Hagberg L., Engberg I., Freter R., Lam J., Olling S., Svanborg Edén C. Ascending, unobstructed urinary tract infection in mice caused by pyelonephritogenic Escherichia coli of human origin. Infect Immun. 1983 Apr;40(1):273–283. doi: 10.1128/iai.40.1.273-283.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagberg L., Hull R., Hull S., Falkow S., Freter R., Svanborg Edén C. Contribution of adhesion to bacterial persistence in the mouse urinary tract. Infect Immun. 1983 Apr;40(1):265–272. doi: 10.1128/iai.40.1.265-272.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iwahi T., Abe Y., Nakao M., Imada A., Tsuchiya K. Role of type 1 fimbriae in the pathogenesis of ascending urinary tract infection induced by escherichia coli in mice. Infect Immun. 1983 Mar;39(3):1307–1315. doi: 10.1128/iai.39.3.1307-1315.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klinger J. D., Cash H. A., Wood R. E., Miler J. J. Protective immunization against chronic Pseudomonas aeruginosa pulmonary infection in rats. Infect Immun. 1983 Mar;39(3):1377–1384. doi: 10.1128/iai.39.3.1377-1384.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Källenius G., Svenson S., Möllby R., Cedergren B., Hultberg H., Winberg J. Structure of carbohydrate part of receptor on human uroepithelial cells for pyelonephritogenic Escherichia coli. Lancet. 1981 Sep 19;2(8247):604–606. doi: 10.1016/s0140-6736(81)92743-4. [DOI] [PubMed] [Google Scholar]
- Marrie T. J., Swantee C. A., Hartlen M. Aerobic and anaerobic urethral flora of healthy females in various physiological age groups and of females with urinary tract infections. J Clin Microbiol. 1980 Jun;11(6):654–659. doi: 10.1128/jcm.11.6.654-659.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parsons C. L., Greenspan C., Mulholland S. G. The primary antibacterial defense mechanism of the bladder. Invest Urol. 1975 Jul;13(1):72–78. [PubMed] [Google Scholar]
- Read A. E., McCarthy C. F., Heaton K. W., Laidlaw J. Lactobacillus acidophilus (enpac) in treatment of hepatic encephalopathy. Br Med J. 1966 May 21;1(5498):1267–1269. doi: 10.1136/bmj.1.5498.1267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reid G., Brooks H. J., Bacon D. F. In vitro attachment of Escherichia coli to human uroepithelial cells: variation in receptivity during the menstrual cycle and pregnancy. J Infect Dis. 1983 Sep;148(3):412–421. doi: 10.1093/infdis/148.3.412. [DOI] [PubMed] [Google Scholar]
- Roberts J. A., Hardaway K., Kaack B., Fussell E. N., Baskin G. Prevention of pyelonephritis by immunization with P-fimbriae. J Urol. 1984 Mar;131(3):602–607. doi: 10.1016/s0022-5347(17)50513-3. [DOI] [PubMed] [Google Scholar]
- Roberts J. A. Pathogenesis of pyelonephritis. J Urol. 1983 Jun;129(6):1102–1106. doi: 10.1016/s0022-5347(17)52592-6. [DOI] [PubMed] [Google Scholar]
- SHINEFIELD H. R., RIBBLE J. C., EICHENWALD H. F., BORIS M., SUTHERLAND J. M. Bacterial interference: its effect on nursery-acquired infection with Staphylococcus aureus. V. An analysis and interpretation. Am J Dis Child. 1963 Jun;105:683–688. [PubMed] [Google Scholar]
- Sautter R. L., Brown W. J. Sequential vaginal cultures from normal young women. J Clin Microbiol. 1980 May;11(5):479–484. doi: 10.1128/jcm.11.5.479-484.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schaeffer A. J., Chmiel J. S., Duncan J. L., Falkowski W. S. Mannose-sensitive adherence of Escherichia coli to epithelial cells from women with recurrent urinary tract infections. J Urol. 1984 May;131(5):906–910. doi: 10.1016/s0022-5347(17)50706-5. [DOI] [PubMed] [Google Scholar]
- Sobel J. D., Myers P. G., Kaye D., Levison M. E. Adherence of Candida albicans to human vaginal and buccal epithelial cells. J Infect Dis. 1981 Jan;143(1):76–82. doi: 10.1093/infdis/143.1.76. [DOI] [PubMed] [Google Scholar]
- Sprunt K., Redman W. Evidence suggesting importance of role of interbacterial inhibition in maintaining balance of normal flora. Ann Intern Med. 1968 Mar;68(3):579–590. doi: 10.7326/0003-4819-68-3-579. [DOI] [PubMed] [Google Scholar]
- Uehling D. T., Mizutani K., Balish E. Effect of immunization of bacterial adherence to urothelium. Invest Urol. 1978 Sep;16(2):145–147. [PubMed] [Google Scholar]