Abstract
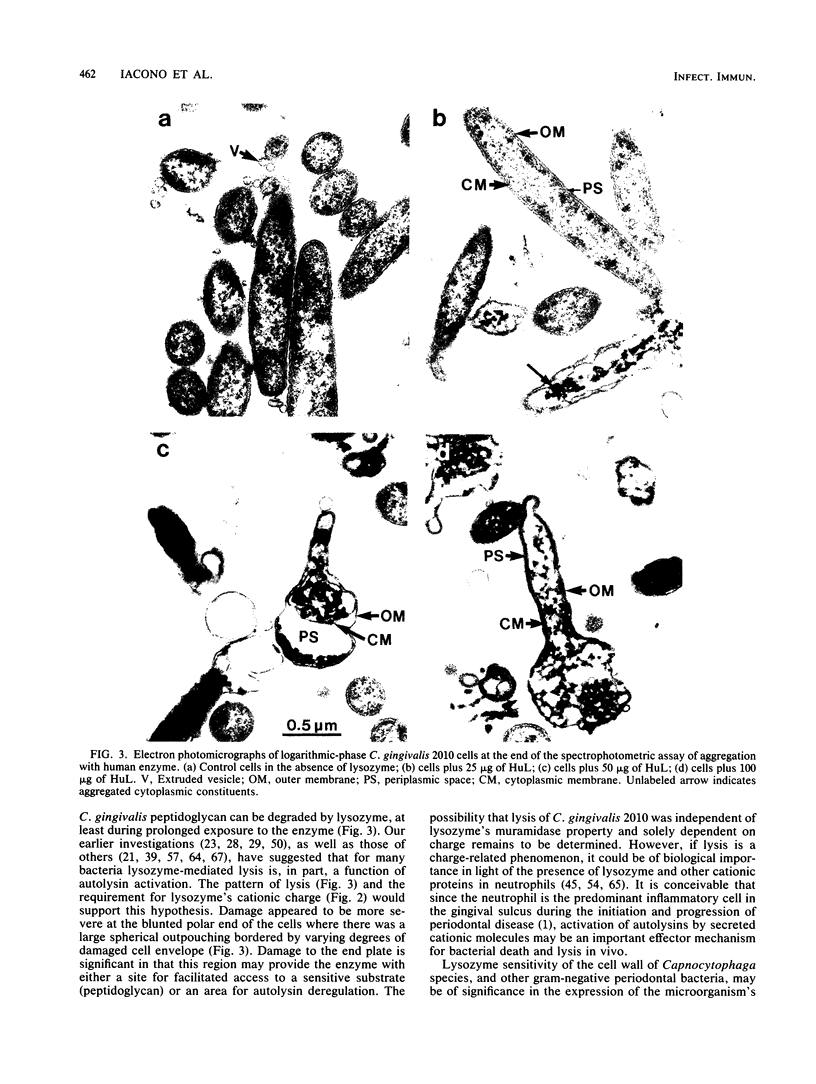
The ability of lysozyme to aggregate and lyse the gram-negative capnophilic periodontal microorganism Capnocytophaga gingivalis 2010 was monitored optically at 540 nm. Both hen egg white and chromatographically purified human lysozymes had significant but similar aggregation potentials for both logarithmic- and stationary-phase bacteria. In general, an increase in enzyme concentration resulted in a graded increase in both the initial and maximum changes in turbidity which occurred during the reaction period. The greatest change in turbidity occurred within the initial minutes of interaction of lysozyme and the cells, and the extent of aggregation paralleled a rapid depletion of lysozyme by the suspensions during the first minute of its incubation with the bacteria. Interestingly, the muramidase inhibitors N-acetyl-D-glucosamine and histamine did not block aggregation, whereas maleylation of lysozyme completely inhibited its aggregating ability. Demaleylation, however, restored aggregation activity comparable to the native enzyme, indicating that maleylated lysozyme retained its integrity and that aggregation was primarily dependent on charge. The addition of up to physiological concentrations of NaHCO3 and NaCl to cell aggregates resulted in varying degrees of deaggregation and lysis. Surprisingly, ultrastructural analysis of lysozyme-treated cells revealed morphological changes with or without the addition of salt. Damage appeared to occur at the blunted polar end of the cells where there was a large spherical outpouching bordered by a damaged cell envelope. Damaged cells uniformly contained dense granular cytoplasmic debris. In effect, the cationic enzyme lysed C. gingivalis 2010, which was not apparent in the spectrophotometric assay. The paradoxical finding that during bacterial aggregation there was lysis may be of significance to the further elucidation of lysozyme's antibacterial role in the gingival sulcus.
Full text
PDF

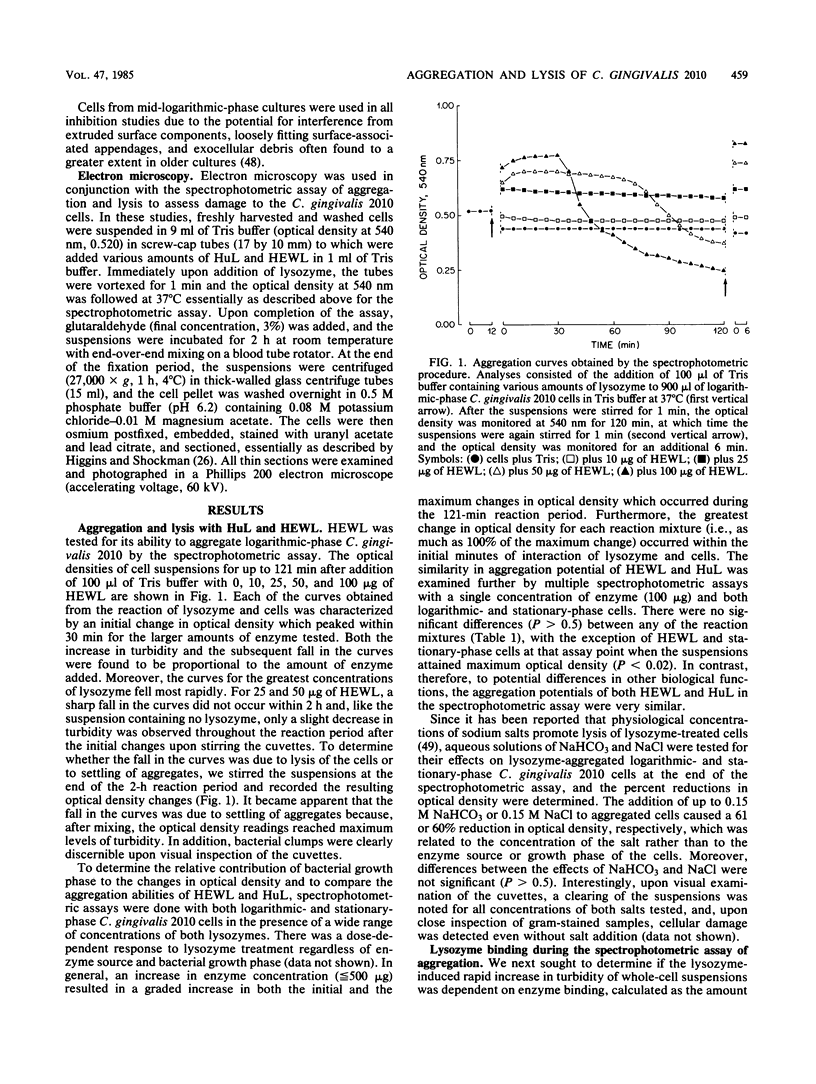


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Attström R. Studies on neutrophil polymorphonuclear leukocytes at the dento-gingival junction in gingival health and disease. J Periodontal Res Suppl. 1971;8:1–15. [PubMed] [Google Scholar]
- BLADEN H. A., MERGENHAGEN S. E. ULTRASTRUCTURE OF VEILLONELLA AND MORPHOLOGICAL CORRELATION OF AN OUTER MEMBRANE WITH PARTICLES ASSOCIATED WITH ENDOTOXIC ACTIVITY. J Bacteriol. 1964 Nov;88:1482–1492. doi: 10.1128/jb.88.5.1482-1492.1964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BRANDTZAEG P., MANN W. V., Jr A COMPARATIVE STUDY OF THE LYSOZYME ACTIVITY OF HUMAN GINGIVAL POCKET FLUID, SERUM, AND SALIVA. Acta Odontol Scand. 1964 Oct;22:441–455. doi: 10.3109/00016356409028217. [DOI] [PubMed] [Google Scholar]
- Babu U. M., Zeiger A. R. Soluble peptidoglycan from Staphylococcus aureus is a murine B-lymphocyte mitogen. Infect Immun. 1983 Dec;42(3):1013–1016. doi: 10.1128/iai.42.3.1013-1016.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bolton R. W., Dyer J. K., Reinhardt R. A., Okano D. K. Modulation of in vitro human lymphocyte responses by an exopolysaccharide from Capnocytophaga ochracea. J Dent Res. 1983 Dec;62(12):1186–1189. doi: 10.1177/00220345830620120201. [DOI] [PubMed] [Google Scholar]
- Bolton R. W., Dyer J. K. Suppression of murine lymphocyte mitogen responses by exopolysaccharide from Capnocytophaga ochracea. Infect Immun. 1983 Jan;39(1):476–479. doi: 10.1128/iai.39.1.476-479.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bretz U., Baggiolini M. Biochemical and morphological characterization of azurophil and specific granules of human neutrophilic polymorphonuclear leukocytes. J Cell Biol. 1974 Oct;63(1):251–269. doi: 10.1083/jcb.63.1.251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Butler P. J., Harris J. I., Hartley B. S., Lebeman R. The use of maleic anhydride for the reversible blocking of amino groups in polypeptide chains. Biochem J. 1969 May;112(5):679–689. doi: 10.1042/bj1120679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carranza F. A., Jr, Saglie R., Newman M. G., Valentin P. L. Scanning and transmission electron microscopic study of tissue-invading microorganisms in localized juvenile periodontitis. J Periodontol. 1983 Oct;54(10):598–617. doi: 10.1902/jop.1983.54.10.598. [DOI] [PubMed] [Google Scholar]
- Davies P., Bonney R. J., Humes J. L., Kuehl F. A. The role of macrophage secretory products in chronic inflammatory processes. J Invest Dermatol. 1980 May;74(5):292–296. doi: 10.1111/1523-1747.ep12543476. [DOI] [PubMed] [Google Scholar]
- Dziarski R. Polyclonal activation of immunoglobulin secretion in B lymphocytes induced by staphylococcal peptidoglycan. J Immunol. 1980 Dec;125(6):2478–2483. [PubMed] [Google Scholar]
- Forlenza S. W., Newman M. G., Lipsey A. I., Siegel S. E., Blachman U. Capnocytophaga sepsis: a newly recognised clinical entity in granulocytopenic patients. Lancet. 1980 Mar 15;1(8168 Pt 1):567–568. doi: 10.1016/s0140-6736(80)91057-0. [DOI] [PubMed] [Google Scholar]
- Friedman S. A., Mandel I. D., Herrera M. S. Lysozyme and lactoferrin quantitation in the crevicular fluid. J Periodontol. 1983 Jun;54(6):347–350. doi: 10.1902/jop.1983.54.6.347. [DOI] [PubMed] [Google Scholar]
- Gandola C., Butler T., Badger S., Cheng E., Beard S. Septicemia caused by Capnocytophaga in a granulocytopenic patient with glossitis. Arch Intern Med. 1980 Jun;140(6):851–852. [PubMed] [Google Scholar]
- Genco R. J., Slots J. Host responses in periodontal diseases. J Dent Res. 1984 Mar;63(3):441–451. doi: 10.1177/00220345840630031601. [DOI] [PubMed] [Google Scholar]
- Gibbons R. J. Adherent interactions which may affect microbial ecology in the mouth. J Dent Res. 1984 Mar;63(3):378–385. doi: 10.1177/00220345840630030401. [DOI] [PubMed] [Google Scholar]
- Ginsburg I. The role of lysosomal factors of leukocytes in the biodegradation and storage of microbial constituents in infectious granulomas. Front Biol. 1979;48:327–406. [PubMed] [Google Scholar]
- Glazer A. N., Barel A. O., Howard J. B., Brown D. M. Isolation and characterization of fig lysozyme. J Biol Chem. 1969 Jul 10;244(13):3583–3589. [PubMed] [Google Scholar]
- Greenblatt J., Boackle R. J., Schwab J. H. Activation of the alternate complement pathway by peptidoglycan from streptococcal cell wall. Infect Immun. 1978 Jan;19(1):296–303. doi: 10.1128/iai.19.1.296-303.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gregory E. M., Moore W. E., Holdeman L. V. Superoxide dismutase in anaerobes: survey. Appl Environ Microbiol. 1978 May;35(5):988–991. doi: 10.1128/aem.35.5.988-991.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Higgins M. L., Shockman G. D. Early changes in the ultrastructure of Streptococcus faecalis after amino acid starvation. J Bacteriol. 1970 Jul;103(1):244–253. doi: 10.1128/jb.103.1.244-253.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iacono V. J., Boldt P. R., MacKay B. J., Cho M. I., Pollock J. J. Lytic sensitivity of Actinobacillus actinomycetemcomitans Y4 to lysozyme. Infect Immun. 1983 May;40(2):773–784. doi: 10.1128/iai.40.2.773-784.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iacono V. J., MacKay B. J., DiRienzo S., Pollock J. J. Selective antibacterial properties of lysozyme for oral microorganisms. Infect Immun. 1980 Aug;29(2):623–632. doi: 10.1128/iai.29.2.623-632.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iacono V. J., Taubman M. A., Smith D. J., Moreno E. C. A spectrophotometric procedure for quantitation of antibody directed to bacterial antigens. Immunochemistry. 1976 Mar;13(3):235–243. doi: 10.1016/0019-2791(76)90221-4. [DOI] [PubMed] [Google Scholar]
- Kilian M. Degradation of immunoglobulins A2, A2, and G by suspected principal periodontal pathogens. Infect Immun. 1981 Dec;34(3):757–765. doi: 10.1128/iai.34.3.757-765.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laible N. J., Germaine G. R. Adsorption of lysozyme from human whole saliva by Streptococcus sanguis 903 and other oral microorganisms. Infect Immun. 1982 Apr;36(1):148–159. doi: 10.1128/iai.36.1.148-159.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leadbetter E. R., Holt S. C., Socransky S. S. Capnocytophaga: new genus of gram-negative gliding bacteria. I. General characteristics, taxonomic considerations and significance. Arch Microbiol. 1979 Jul;122(1):9–16. doi: 10.1007/BF00408040. [DOI] [PubMed] [Google Scholar]
- Leffell M. S., Spitznagel J. K. Association of lactoferrin with lysozyme in granules of human polymorphonuclear leukocytes. Infect Immun. 1972 Nov;6(5):761–765. doi: 10.1128/iai.6.5.761-765.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lehrer S. S., Fasman G. D. The fluorescence of lysozyme and lysozyme substrate complexes. Biochem Biophys Res Commun. 1966 Apr 19;23(2):133–138. doi: 10.1016/0006-291x(66)90517-1. [DOI] [PubMed] [Google Scholar]
- Liljemark W. F., Bloomquist C. G., Germaine G. R. Effect of bacterial aggregation on the adherence of oral streptococci to hydroxyapatite. Infect Immun. 1981 Mar;31(3):935–941. doi: 10.1128/iai.31.3.935-941.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Logardt I. M., Neujahr H. Y. Lysis of modified walls from Lactobacillus fermentum. J Bacteriol. 1975 Oct;124(1):73–77. doi: 10.1128/jb.124.1.73-77.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacKay B. J., Iacono V. J., Zuckerman J. M., Osserman E. F., Pollock J. J. Quantitative recovery, selective removal and one-step purification of human parotid and leukemic lysozymes by immunoadsorption. Eur J Biochem. 1982 Dec;129(1):93–98. doi: 10.1111/j.1432-1033.1982.tb07025.x. [DOI] [PubMed] [Google Scholar]
- Mashimo P. A., Yamamoto Y., Nakamura M., Slots J. Selective recovery of oral Capnocytophaga spp. with sheep blood agar containing bacitracin and polymyxin B. J Clin Microbiol. 1983 Feb;17(2):187–191. doi: 10.1128/jcm.17.2.187-191.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mashimo P. A., Yamamoto Y., Slots J., Park B. H., Genco R. J. The periodontal microflora of juvenile diabetics. Culture, immunofluorescence, and serum antibody studies. J Periodontol. 1983 Jul;54(7):420–430. doi: 10.1902/jop.1983.54.7.420. [DOI] [PubMed] [Google Scholar]
- Metcalf R. H., Deibel R. H. Differential lytic response of enterococci associated with addition order of lysozyme and anions. J Bacteriol. 1969 Sep;99(3):674–680. doi: 10.1128/jb.99.3.674-680.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Modrzakowski M. C., Spitznagel J. K. Bactericidal activity of fractionated granule contents from human polymorphonuclear leukocytes: antagonism of granule cationic proteins by lipopolysaccharide. Infect Immun. 1979 Aug;25(2):597–602. doi: 10.1128/iai.25.2.597-602.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oken M. M., Peterson P. K., Wilkinson B. J. Endogenous pyrogen production by human blood monocytes stimulated by staphylococcal cell wall components. Infect Immun. 1981 Jan;31(1):208–213. doi: 10.1128/iai.31.1.208-213.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Osserman E. F., Lawlor D. P. Serum and urinary lysozyme (muramidase) in monocytic and monomyelocytic leukemia. J Exp Med. 1966 Nov 1;124(5):921–952. doi: 10.1084/jem.124.5.921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poirier T. P., Tonelli S. J., Holt S. C. Ultrastructure of gliding bacteria: scanning electron microscopy of Capnocytophaga sputigena, Capnocytophaga gingivalis, and Capnocytophaga ochracea. Infect Immun. 1979 Dec;26(3):1146–1158. doi: 10.1128/iai.26.3.1146-1158.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pollock J. J., Goodman H., Elsey P. K., Iacono V. J. Synergism of lysozyme, proteases and inorganic monovalent anions in the bacteriolysis of oral Streptococcus mutans GS5. Arch Oral Biol. 1983;28(9):865–871. doi: 10.1016/0003-9969(83)90045-6. [DOI] [PubMed] [Google Scholar]
- Pollock J. J., Sharon N. Studies on the acceptor specificity of the lysozyme-catalyzed transglycosylation reaction. Biochemistry. 1970 Sep 29;9(20):3913–3925. doi: 10.1021/bi00822a009. [DOI] [PubMed] [Google Scholar]
- Rateitschak-Plüss E. M., Schroeder H. E. History of periodontitis in a child with Papillon-Lefèvre syndrome. A case report. J Periodontol. 1984 Jan;55(1):35–46. doi: 10.1902/jop.1984.55.1.35. [DOI] [PubMed] [Google Scholar]
- Reitamo S., Klockars M., Adinolfi M., Osserman E. F. Human lysozyme (origin and distribution in health and disease). Ric Clin Lab. 1978 Oct-Dec;8(4):211–231. [PubMed] [Google Scholar]
- Rest R. F., Cooney M. H., Spitznagel J. K. Bactericidal activity of specific and azurophil granules from human neutrophils: studies with outer-membrane mutants of Salmonella typhimurium LT-2. Infect Immun. 1978 Jan;19(1):131–137. doi: 10.1128/iai.19.1.131-137.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SALTON M. R. The properties of lysozyme and its action on microorganisms. Bacteriol Rev. 1957 Jun;21(2):82–100. doi: 10.1128/br.21.2.82-100.1957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SOPHIANOPOULOS A. J., RHODES C. K., HOLCOMB D. N., VAN HOLDE K. E. Physical studies of lysozyme. I. Characterization. J Biol Chem. 1962 Apr;237:1107–1112. [PubMed] [Google Scholar]
- Shinitzky M., Katchalski E., Grisaro V., Sharon N. Inhibition of lysozyme by imidazole and indole derivatives. Arch Biochem Biophys. 1966 Sep 26;116(1):332–343. doi: 10.1016/0003-9861(66)90039-7. [DOI] [PubMed] [Google Scholar]
- Shurin S. B., Socransky S. S., Sweeney E., Stossel T. P. A neutrophil disorder induced by capnocytophaga, a dental micro-organism. N Engl J Med. 1979 Oct 18;301(16):849–854. doi: 10.1056/NEJM197910183011601. [DOI] [PubMed] [Google Scholar]
- Slots J. Subgingival microflora and periodontal disease. J Clin Periodontol. 1979 Oct;6(5):351–382. doi: 10.1111/j.1600-051x.1979.tb01935.x. [DOI] [PubMed] [Google Scholar]
- Stevens R. H., Hammond B. F., Lai C. H. Characterization of an inducible bacteriophage from a leukotoxic strain of Actinobacillus actinomycetemcomitans. Infect Immun. 1982 Jan;35(1):343–349. doi: 10.1128/iai.35.1.343-349.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanner A. C., Haffer C., Bratthall G. T., Visconti R. A., Socransky S. S. A study of the bacteria associated with advancing periodontitis in man. J Clin Periodontol. 1979 Oct;6(5):278–307. doi: 10.1111/j.1600-051x.1979.tb01931.x. [DOI] [PubMed] [Google Scholar]
- Tortosa M., Cho M. I., Wilkens T. J., Iacono V. J., Pollock J. J. Bacteriolysis of Veillonella alcalescens by lysozyme and inorganic anions present in saliva. Infect Immun. 1981 Jun;32(3):1261–1273. doi: 10.1128/iai.32.3.1261-1273.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wecke J., Lahav M., Ginsburg I., Giesbrecht P. Cell wall degradation of Staphylococcus aureus by lysozyme. Arch Microbiol. 1982 Mar;131(2):116–123. doi: 10.1007/BF01053992. [DOI] [PubMed] [Google Scholar]
- Weiss J., Elsbach P., Olsson I., Odeberg H. Purification and characterization of a potent bactericidal and membrane active protein from the granules of human polymorphonuclear leukocytes. J Biol Chem. 1978 Apr 25;253(8):2664–2672. [PubMed] [Google Scholar]
- Weissmann G., Smolen J. E., Korchak H. M. Release of inflammatory mediators from stimulated neutrophils. N Engl J Med. 1980 Jul 3;303(1):27–34. doi: 10.1056/NEJM198007033030109. [DOI] [PubMed] [Google Scholar]
- Westmacott D., Perkins H. R. Effects of lysozyme on Bacillus cereus 569: rupture of chains of bacteria and enhancement of sensitivity to autolysins. J Gen Microbiol. 1979 Nov;115(1):1–11. doi: 10.1099/00221287-115-1-1. [DOI] [PubMed] [Google Scholar]