Abstract
Summary: Helicobacters other than Helicobacter pylori have been associated with gastritis, gastric ulcers, and gastric mucosa-associated lymphoid tissue lymphoma in humans. These very fastidious microorganisms with a typical large spiral-shaped morphology were provisionally designated “H. heilmannii,” but in fact they comprise at least five different Helicobacter species, all of which are known to colonize the gastric mucosa of animals. H. suis, which has been isolated from the stomachs of pigs, is the most prevalent gastric non-H. pylori Helicobacter species in humans. Other gastric non-H. pylori helicobacters colonizing the human stomach are H. felis, H. salomonis, H. bizzozeronii, and the still-uncultivable “Candidatus Helicobacter heilmannii.” These microorganisms are often detected in the stomachs of dogs and cats. “Candidatus Helicobacter bovis” is highly prevalent in the abomasums of cattle but has only occasionally been detected in the stomachs of humans. There are clear indications that gastric non-H. pylori Helicobacter infections in humans originate from animals, and it is likely that transmission to humans occurs through direct contact. Little is known about the virulence factors of these microorganisms. The recent successes with in vitro isolation of non-H. pylori helicobacters from domestic animals open new perspectives for studying these microorganisms and their interactions with the host.
INTRODUCTION
It was first reported in 1984 that gastric ulcer disease in humans is caused by a bacterial infection (141). The causative agent, Helicobacter pylori, has also been associated with gastritis, peptic ulcer disease, gastric adenocarcinoma, and mucosa-associated lymphoid tissue (MALT) lymphoma (132, 174, 215). This bacterium is very successful in the way that it colonizes the human stomach, since in developing countries, more than 80% of the population is infected with H. pylori, even at young age. In developed countries, the prevalence of H. pylori generally remains under 40% and is considerably lower in children and adolescents than in adults and elderly people (132, 181).
Various tests have been developed for the diagnosis of H. pylori infections (reviewed in reference 132). For routine diagnostic purposes, histology or culture of biopsies from patients who have undergone endoscopy and urea breath testing are most often used. On histology, H. pylori bacteria are identified on the basis of their typical localization and their characteristic, slightly curve-shaped morphology. In 0.2 to 6% (depending on the literature source and the geographical region) of these biopsies, however, bacteria with a different, typically long spiral-shaped morphology are found. These spiral-shaped non-H. pylori helicobacters were first described in 1987 (46). They were originally referred to as “Gastrospirillum hominis” (143). Analysis of the 16S rRNA gene of these uncultivated organisms resulted in their classification in the genus Helicobacter. They were provisionally named “H. heilmannii” after the German pathologist Konrad Heilmann, who first studied the pathology associated with these microorganisms (101). “H. heilmannii” has also been associated with gastritis (40), gastric ulcers (41), and gastric MALT lymphoma (153) but not with gastric adenocarcinoma. Further research on “H. heilmannii” has been seriously hampered by the very fastidious nature of these microorganisms. Even today, to our knowledge, only two “H. heilmannii” strains have been cultured from human tissue (2, 127). Long spiral-shaped helicobacters have also been demonstrated in the stomachs of different animal species. A summary of these helicobacters is given in Table 1.
TABLE 1.
Helicobacter species naturally colonizing the stomachs of animals and their pathogenic significance for humans
| Helicobacter species | Natural host (prevalence, %) | Associated with gastric disease in humans | Reference(s) |
|---|---|---|---|
| H. suis | Pig (60-80 in slaughter pigs), macaque (NAb), mandrill monkey (NA) | Yes | 14, 42, 45, 167, 227, 230 |
| H. felis | Dog (47), cat (63), rabbit (2-9), cheetah (NAc) | Yes | 45, 134, 166, 227, 230, 231, 234 |
| H. bizzozeronii | Dog (70), cat (35) | Yes | 45, 92, 227, 230 |
| H. salomonis | Dog (9), cat (2), rabbit (0-4) | Yes | 45, 115, 227, 230 |
| “Candidatus Helicobacter heilmannii” | Dog (20-100), cat (20-100), wild felidae (NAc), nonhuman primates (66) | Yes | 166, 227, 230 |
| H. baculiformis | Cat (NA) | No | 15 |
| H. cynogastricus | Dog (NA) | No | 233 |
| “Candidatus Helicobacter bovis” | Cattle (NA) | Yes | 44, 45 |
| H. mustelae | Ferret (0-100) | No | 66, 67, 68, 69, 70, 71, 85 |
| H. aurati | Syrian hamster (50-100) | No | 177 |
| H. nemestrinaea | Macaque (NA) | No | 24 |
| H. acinonychis | Cheetah (low), tiger (NA) | No | 60, 224 |
| H. cetorum | Whales (NA), dolphins (NA) | No | 97 |
| H. muridarum | Mice (0-62) | No | 86, 136, 183 |
Later heterotypic synonym of H. pylori (219).
NA, not available.
Terio et al. (224) and Mörner et al. (155) found the gastric mucosa of 75% of cheetahs and 68% of free-ranging lynx to be colonized with “pet carnivore-associated” helicobacters. Those studies do not allow differentiation between H. felis, H. bizzozeronii, H. salomonis, H. baculiformis, H. cynogastricus, and “Candidatus Helicobacter heilmannii”.
This article aims to provide an overview of Helicobacter species naturally colonizing the stomachs of food-producing animals, pet animals, and nonhuman primates. First, the very complex and confusing nomenclature used to designate non-H. pylori Helicobacter species colonizing the human stomach is considered. Thereafter, an overview of helicobacters colonizing the stomachs of domestic animals and nonhuman primates is presented, and their possible pathogenic significance for their animal hosts is discussed. The main aim of this article, however, is to have a closer look at the significance of these microorganisms for human health: should they be considered zoonotic agents, what are the disease signs in infected humans, how are they transmitted, and what is known about their virulence factors? The article ends with some thoughts on what the study of gastric Helicobacter infections in animals might teach us about these infections in humans.
GASTRIC NON-H. PYLORI HELICOBACTER NOMENCLATURE: THE NEED FOR CLARIFICATION
Since the description of H. pylori, the number of species in the genus Helicobacter has rapidly expanded. Today, a large number of non-H. pylori Helicobacter species in a wide variety of animals and humans have been described, and the genus Helicobacter contains at least 32 species with validly published names (http://www.bacterio.cict.fr/h/helicobacter.html). The frequent changes in nomenclature of non-H. pylori helicobacters colonizing the stomachs of humans have caused quite a lot of confusion, not only among clinicians but also among bacteriologists. Today, there is a serious problem in trying to reach international agreement on this complex and expanding group of microorganisms, all of which have in common their tightly coiled morphology and their difficulty to culture in vitro.
After the renaming of “Gastrospirillum hominis” as “H. heilmannii,” further genetic analysis of the 16S rRNA gene revealed two types that differed by more than 3% in their nucleotide sequence, which prompted the subclassification of the non-H. pylori helicobacters into “H. heilmannii” type 1 and “H. heilmannii” type 2. Sequencing of the 23S rRNA-encoding genes also makes it possible to distinguish between the two types (48).
“H. heilmannii” type 1 is both morphologically and genetically identical to a bacterium colonizing the stomachs of pigs (43, 166) that was first designated “Gastrospirillum suis” (146, 184). Almost 10 years later, sequencing of the 16S rRNA gene, fluorescent in situ hybridization (FISH), and electron microscopy showed that these organisms belong to the genus Helicobacter and are sufficiently different from all existing species to constitute a new taxon. Because at that time this species could not be thoroughly characterized due to the lack of pure in vitro isolates, the organism was described as “Candidatus Helicobacter suis” (43). Only recently have in vitro cultures been obtained, resulting in the description of H. suis as a species (14).
The situation with regard to “H. heilmannii” type 2 is even more complex. This type represents not a single Helicobacter species but rather a group of species, including three helicobacters that have been isolated from the stomachs of cats and dogs, namely, H. felis, H. bizzozeronii, and H. salomonis. To add to the confusion, one uncultivable species detected in the stomachs of humans, wild felids, dogs, and cats was named “Candidatus Helicobacter heilmannii” (166). Two other closely related species, one of which was isolated from a dog and the other from a cat, have been described as H. cynogastricus and H. baculiformis, respectively (15, 233). However, to date no information is available about the presence of these bacteria in humans.
Differences in morphology between different gastric non-H. pylori Helicobacter species have been described (Table 2), but this is not an accurate method for species identification. It has been stated that periplasmic fibrils wrapped around the cell body are a typical feature of H. felis (134). However, H. cynogastricus also possesses a periplasmic fibril running along the external side of the helix, and both species are tightly coiled organisms (233). H. salomonis is less tightly coiled and does not have periplasmic fibrils (115). H. baculiformis is a large, slender to slightly spiral rod with periplasmic fibrils (15). “Candidatus Helicobacter heilmannii,” H. bizzozeronii, and H. suis are morphologically very similar. These microorganisms do not possess periplasmic fibrils and show very tight coils (14, 92, 166).
TABLE 2.
Differential characteristics of gastric Helicobacter species associated with domestic animals and nonhuman primatesa
| Characteristic | H. baculiformis | H. cynogastricus | H. bizzozeronii | H. felis | H. salomonis | H. pylori | H. suis | “Candidatus Helicobacter heilmannii” | H. mustelae | H. nemestrinaeb | “Candidatus Helicobacter bovis” | H. aurati |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Length (μm) | 10 | 10-18 | 5-10 | 5-7.5 | 5-7 | 2.5-5 | 2.3-6.7 | 5-10 | 2 | ND | 1.5-2.5 | 4-8 |
| Cell width (μm) | 1 | 0.8-1.0 | 0.3 | 0.4 | 0.8-1.2 | 0.5-1.0 | 0.9-1.2 | 0.5-0.6 | 0.5 | ND | 0.3 | 0.6 |
| Nitrate reduction | + | + | + | + | + | − | − | ND | + | − | ND | − |
| Urease | + | + | (+) | (+) | + | + | + | + | + | + | + | + |
| Alkaline phosphate hydrolysis | + | + | V | V | V | + | + | ND | + | + | ND | − |
| γ-Glutamyl transpeptidase | + | + | + | + | + | + | + | ND | + | ND | ND | + |
| Indoxyl acetate hydrolysis | − | − | (−) | (−) | (−) | (−) | − | ND | + | − | ND | + |
| Growth at 42°C | − | − | V | V | − | (−) | − | ND | V | + | ND | + |
| Growth on 1% glycine | − | − | (−) | − | − | − | − | ND | − | − | ND | − |
| Periplasmic fibril | + | + | − | + | − | − | − | − | − | − | − | + |
| No. of flagella/cell | 11 | 6-12 | 10-20 | 14-20 | 10-23 | 4-8 | 4-10 | 10-20 | 4-8 | 4-8 | ≥4 | 7-10 |
| Distribution of flagella | BP | BP | BP | BP | BP | MP | BP | BP | LP | BP | ND | BP |
Data were obtained from references 14, 15, 44, 47, 68, 92, 93, 115, 134, 166, 177, and 233. All taxa are positive for catalase production and possess sheathed flagella. +, 100% of strains positive; −, 0% of strains positive; (+), 80 to 94% of strains positive; −, 7 to 33% of strains positive; V, 42 to 66% of strains positive; ND, not determined; BP, bipolar; MP, monopolar; LP, lateral polar.
Later heterotypic synonym of H. pylori (219).
Sequencing of the 16S and 23S rRNA-encoding genes allows differentiation of H. suis from the other gastric non-H. pylori Helicobacter species mentioned above, but it cannot distinguish between H. felis, H. bizzozeronii, H. salomonis, H. cynogastricus, H. baculiformis, and “Candidatus Helicobacter heilmannii” (15, 48, 233). For differentiation between these species, sequencing of the Hsp60 gene (149), the urease A and B genes (161, 166), and the gyrB gene (95) is useful, as is whole-cell protein profiling (228) if pure in vitro cultures are available.
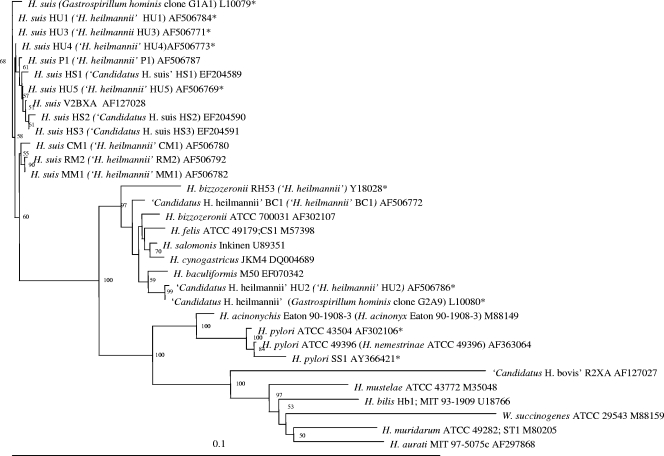
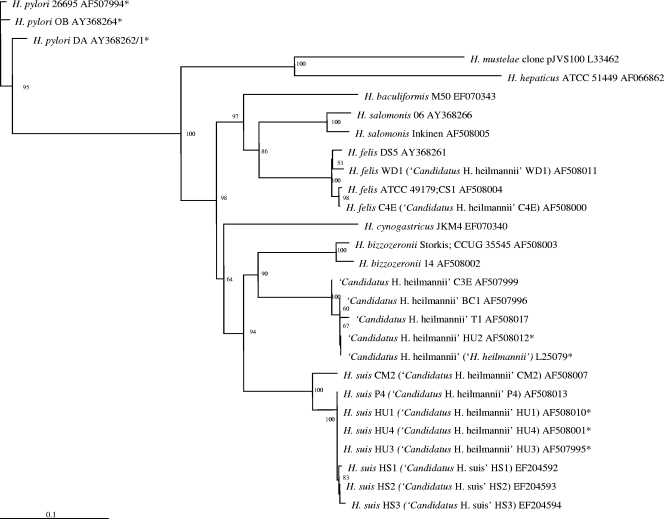
Phylogenetic trees for the gastric helicobacters discussed in this review are shown in Fig. 1 and 2. Figure 1 is based on 16S rRNA gene sequence similarity data and Fig. 2 on the partial ureA and ureB gene sequences. The sequences that have been detected in human stomachs are also indicated.
FIG. 1.
Phylogenetic tree based on the near-complete 16S rRNA gene sequences from gastric Helicobacter species and other closely related bacteria. The sequences were aligned using the CLUSTAL W program (225), and a phylogenetic tree was constructed using the neighbor-joining method (191) via the PHYLIP package (64). DNADIST was used for distance analysis (126). Bootstrap values (for branches present in more than 50 out of 100 resamplings of the data) are indicated at the nodes. Original names found in the Entrez Nucleotide database (NCBI) are shown in parentheses. Sequences marked with an asterisk are derived from bacteria demonstrated to be present in the stomachs of humans.
FIG. 2.
Phylogenetic tree based on the partial ureA and ureB gene sequences from gastric Helicobacter species and other closely related bacteria. The sequences were aligned using the CLUSTAL W program (225), and a phylogenetic tree was constructed using the neighbor-joining method (191) via the PHYLIP package (64). DNADIST was used for distance analysis (126). Bootstrap values (for branches present in more than 50 out of 100 resamplings of the data) are indicated at the nodes. Original names found in the Entrez Nucleotide database (NCBI) are shown in parentheses. Sequences marked with an asterisk are derived from bacteria demonstrated in the stomach of humans.
In the literature, gastric infections with spiral-shaped bacteria in humans are often referred to as “H. heilmanni” or “H. heilmannii-like organism” infections. However, at present, the name “H. heilmannii” cannot be used as a species name, according to taxonomical rules. To avoid confusion, we propose to use the term “gastric non-H. pylori helicobacters” to designate these spiral-shaped bacteria when only results of histopathology or crude taxonomic data are available and to reserve true species designations for those situations in which the species is defined.
To nonbacteriologists, the changes in “H. heilmannii” nomenclature may appear unwieldy and unnecessary. However, it should be kept in mind that several important traits, including pathogenicity and antimicrobial susceptibility, may vary depending on the bacterial species. At present it is not known whether certain non-H. pylori Helicobacter species are more often associated with a certain disease outcome in humans than others.
GASTRIC HELICOBACTERS IN DOMESTIC ANIMALS AND NONHUMAN PRIMATES: AN OVERVIEW
A summary of gastric lesions described in domestic animals and nonhuman primates naturally or experimentally infected with helicobacters is presented in Table 3. Below, infections with gastric helicobacters in pigs, dogs, cats, rabbits, ferrets, hamsters, ruminants, horses, and nonhuman primates are considered. Guinea pigs and Mongolian gerbils are also often kept as pets, but natural infections with gastric helicobacters have not been described in these animal species.
TABLE 3.
Summary of gastric lesions in different animal species naturally or experimentally infected with helicobacters
| Animal species | Helicobacter | Infection | Gastric gross lesion(s) | Gastric histological lesion(s) | Localization of Helicobacter sp. | Reference(s) |
|---|---|---|---|---|---|---|
| Pig | H. suis | Natural | Surface redness | NDa | Antrum, fundus | 87 |
| None | ND | Antrum, fundus | 104 | |||
| Mucosa redness and edema, occasional erosions and hemorrhage | Antrum, fundus: diffuse mononuclear cell infiltration with occasional neutrophilic infiltrate and lymphoid follicles | Antrum, fundus: in mucus, in lumen of the pits, in mucosal surface; positive correlation between presence of bacteria and pyloric gastritis | 147 | |||
| Fundus: proliferation of gastric folds with occasional necrosis, severe mucosal congestion; pars esophagea: hyperkeratosis | Diffuse lymphocytic infiltration and lymphoid follicles in lamina propria | Antrum: in mucus, gastric pits, and lumen of gastric glands | 173 | |||
| Pars esophagea: hyperkeratosis with yellow discoloration or chronic ulcers | Pars esophagea: increased thickness of epithelium, elongation of papillae, parakeratosis, balloon cells or chronic peptic ulcers (layers of necrosis, numerous inflammatory cells, and granulation tissue and fibrosis); antrum: mild diffuse mononuclear cell infiltration in lamina propria with multiple lymphocytic aggregates or lymphoid follicles | Antrum, fundus, cardia; positive correlation between presence of bacteria and lesions in pars esophagea | 185 | |||
| Pars esophagea ulcerative gastritis | Pars esophagea: erosion of surface epithelium, necrosis, mixed inflammatory infiltrate, and granulation tissue | Antrum, fundus: in mucous layer and foveolae, occasionally in lumen of gastric glands; positive correlation between presence of bacteria and lesions in pars esophagea | 18 | |||
| Pars esophagea ulcerative gastritis | ND | Pars esophagea: positive correlation between presence of bacteria and lesions in pars esophagea | 28 | |||
| Pars esophagea ulcerative gastritis | Diffuse mononuclear cell infiltration in propria mucosa | Antrum: positive correlation between presence of bacteria and lesions in pars esophagea | 190 | |||
| Experimental | Pars esophagea: no, preulcerative, and ulcerative lesions | Antrum: mild diffuse mononuclear infiltration in lamina propria with multiple lymphocytic aggregates or lymphoid follicles | Antrum: in mucus overlying the surface epithelium and in the surface foveola; fundus: in glandular foveola extending halfway down the gastric pits and often in close association with mucus-producing cells and parietal cells | 103 | ||
| Curve-shaped bacteria morphologically similar to H. pylori | Natural | None | Antrum, fundus, cardia: diffuse mononuclear cell infiltration in lamina propria with multiple lymphoid follicles, and occasional neutrophilic infiltrates and exudation into the glandular lumens | Antrum, fundus, cardia; in close apposition to the gastric epithelia and in the gastric mucus | 129 | |
| Experimental | Gastroesophageal ulceration, glandular mucosal ulcers, lymphoid follicles, excess luminal mucus, and mucosal edema | Pars esophagea: peptic ulceration; antrum, cardia, fundus: diffuse mononuclear cell infiltration in lamina propria with multiple lymphoid follicles | Cardia, antrum: extracellularly | 130 | ||
| Dog | H. felis | Natural | ND | Chronic active gastritis: diffuse lymphoplasmic infiltration, lymphocytic aggregates, and occasional neutrophilic infiltration | Fundus, corpus, antrum: in mucus adjacent to surface epithelium, glandular lamina adjacent to parietal cells, and gastric pits | 51 |
| Experimental | No lesions | Fundus, antrum: diffuse mononuclear cell infiltration in lamina propria with multiple variable-sized lymphoid follicles | Fundus, corpus, antrum: in mucous layer on mucosal surface within gastric pits and glandular lumen, occasionally intracellular in glandular epithelial cells | 137 | ||
| No lesions | Antrum: mild diffuse lymphoplasmacytic inflammation of lamina propria | Fundus, corpus, antrum: in superficial gastric mucous layer, in gastric glands and parietal cells | 204 | |||
| NHPHb | Natural | Mucosal reddening, edema, erosions, and ulcerations | Fundus: glandular degeneration with accumulation of lymphocytes and neutrophilic granulocytes, edema, fibrosis, diffuse lymphoplasmacytic infiltrates, and lymphoid follicles in lamina propria | Fundus: in mucus covering the surface epithelium, the gastric pits, the glandular lumina, and the parietal cells; presence of very high numbers of bacteria was directly related to the no. of lymphoid follicles | 105 | |
| Cat | NHPH | Natural | Mucosal reddening, edema, erosions, and ulcerations | Fundus: glandular degeneration with accumulation of lymphocytes and neutrophilic granulocytes, edema, fibrosis, diffuse lymphoplasmacytic infiltrates, and lymphoid follicles in lamina propria | Fundus: in mucus covering the surface epithelium, the gastric pits, the glandular lumina, and the parietal cells; increased bacterial colonization was directly related to the no. of lymphoid follicles, fibrosis, lesions of surface epithelium, and glandular degeneration | 105 |
| ND | Antrum: diffuse, mixed subglandular leukocytic infiltrates and multiple lymphoid nodules in the lamina propria | Antrum: in the canaliculi or cytoplasm of viable parietal cells | 171 | |||
| ND | Antrum: moderate lymphoid follicles in lamina propria; antrum and fundus: moderate mononuclear infiltrates in lamina propria | Antrum, fundus: at the mucosal surface, in the lumina of gastric glands, and in cytoplasm of parietal cells of the fundus | 193 | |||
| H. felis | Experimental | ND | Antrum: mild diffuse lymphoplasmacytic and eosinophilic infiltration, lymphoid follicular hyperplasia, and mild fibrosis of lamina propria | Antrum, fundus: at the mucosal surface, in the lumina of gastric glands, and in cytoplasm of parietal cells of the fundus | 193 | |
| H. pylori | Experimental | ND | Antrum: mild diffuse lymphoplasmacytic and granulocytic infiltration, severe lymphoid follicular hyperplasia, and mild fibrosis of lamina propria | Antrum, fundus: at the mucosal surface, in the lumina of gastric glands, and in cytoplasm of parietal cells of the fundus | 193 | |
| Ferret | H. mustelae | Natural | Gastritis, peptic ulcer | Proximal antrum: mononuclear cell infiltrates in superficial layer of lamina propria, mucus depletion and occasional neutrophilic infiltrate, gland necrosis and regeneration; distal antrum: diffuse mononuclear cell infiltration and mucus depletion; fundus: mononuclear cell infiltrates in superficial layer of lamina propria, mucus depletion, and occasional neutrophilic infiltrate | Proximal antrum: at surface and lumen of foveola and occasionally in deep glandular lumen; distal antrum: at surface and lumen of foveola and occasionally in superficial glandular lumen; fundus: at surface and lumen of foveola adjacent to the inflammation | 70, 140 |
| Pyloric adenocarcinoma | Antrum: multifocal segmental glandular proliferation and surface erosion, multifocal mucosal lymphoid aggregates, mixed inflammatory cell infiltration, and multifocal fibrosis | Antrum: in the lumen of gastric pits, adherent to apical surface of the mucous epithelium | 75 | |||
| Gastric lymphoma | High- and low-grade B-cell lymphoma | Antrum, fundus: within the mucosal glands | 63 | |||
| Experimental | ND | Antrum: focal minimal lymphocytic infiltrates with moderate numbers of eosinophils and neutrophils; fundus: superficial gastritis consisting of lymphocytes and occasional neutrophils | Antrum, fundus: on the surface of the gastric epithelium within the mucous layer and within gastric pits | 72 | ||
| H. mustelae + MMNGc | Experimental | Pyloric adenocarcinoma | ND | Antrum: epithelial surfaces of the neck glands | 75 | |
| Hamster | H. aurati | Natural | ND | Distal antrum: diffuse lymphoplasmacytic inflammation, scattered heterophils and eosinophils, and goblet cell hyperplasia | Antrum: within gastric pits or glands | 176 |
| Pyloric adenocarcinoma | Distal antrum: locally extensive chronic gastritis with intestinal metaplasia and occasional well-differentiated and moderately pleomorphic tubular to tortuous gastric glands | Antrum: within gastric glands | 159 | |||
| Cattle | “Candidatus Helicobacter bovis” | Natural | ND | ND | Distal antrum: in mucus layer and proximal gastric crypts | 44 |
| Horse | NHPH | Natural | No lesions, gastritis or gastric ulcers | Gastric mucosa: loss of continuity with submucosa exposure and edema; parakeratotic hyperkeratosis; lymphoplasmocytic mononuclear infiltrate | Glandular and nonglandular stomach near margo plicatus | 31 |
| Nonhuman primates | H. pylori | Natural | No lesions, superficial gastritis | Distal fundus, antrum: superficial erosions, marked mononuclear and polynuclear infiltration | Antrum: proximity to the mucosal epithelial cells or in the lumen of the gastric pits | 57 |
| None | No lesions or diffuse lymphoplasmacytic infiltration of the lamina propria, occasional neutrophilic infiltrates and lymphoid follicles | Antrum: superficial mucosa | 55 | |||
| ND | Antrum: diffuse lymphoplasmacytic infiltration in lamina propria, prominent lymphoid follicles, and occasional glandular epithelial hyperplasia, patchy necrosis, attenuation of glandular epithelium, and neutrophilic infiltrates | Antrum: gastric pits and the upper portions of gastric glands, often in intimate association with the epithelial cell surface | 138 | |||
| Localized to multifocal reddening of mucosa | Antrum, cardia: lymphoplasmacytic infiltrates, gastric gland epithelial hyperplasia, reduction in mucin content of surface and gland epithelia, increased lymphoid follicles, minor infiltrates of neutrophils in superficial lamina propria and gastric glands, and occasional erosions | Antrum, cardia: mucosal regions of inflammation; located in gastric pits and upper portions of gastric glands, often associated with epithelial cell surface | 187 | |||
| Experimental | ND | Antrum: chronic-active gastritis, marked atrophy of the mucosa, microerosions and loss of mucus from superficial epithelial cells | Antrum | 56 | ||
| NHPH | Natural | No lesions, superficial gastritis | No lesions to occasional mononuclear and polynuclear infiltration | Fundus: mucus covering the surface of epithelial cells, in the lumina of the gastric glands, and overlying parietal cells | 57 | |
| None | No lesions or diffuse lymphoplasmacytic infiltration of the lamina propria, occasional neutrophilic infiltrates and lymphoid follicles | Fundus: gastric pits, superficial glands or on the surface epithelium, | 55 | |||
| ND | Fundus: minor scattered aggregates of lymphocytes, low numbers of plasma cells, and occasional lymphoid follicles | Fundus: in the gland lumens, sometimes attached to the epithelial cell surface, and within the cytoplasm of parietal cells | 138 | |||
| Localized to multifocal reddening of mucosa | Fundus: no lesions | Fundus: in gland lumens, parietal cells, and surface mucus | 187 |
ND, not described.
NHPH, non-H. pylori Helicobacter species.
MNNG, N-methyl-N-nitro-N′-nitrosoguanidine.
Gastric Helicobacters Associated with Pigs
The main Helicobacter species colonizing the stomachs of pigs is H. suis. Its prevalence at slaughter age in most reports is 60% or more. H. suis causes gastritis in experimentally and naturally infected pigs (87, 104, 147, 173, 185). It has also been associated with ulcers of the nonglandular part of the stomach (18, 28, 185, 190), although the exact role of H. suis in porcine gastric pathology remains to be elucidated. Indeed, Grasso et al. (87), Melnichouk et al. (145), Park et al. (173), and Szeredi et al. (222) did not find this association. These discrepancies might be due to differences in laboratory techniques for demonstration of Helicobacter, different sampling practices, or differences in virulence between different H. suis strains. In any case, in a recent study carried out by our research group, gastric ulcers were induced in pigs experimentally infected with H. suis (T. Meyns, R. Ducatelle, B. Flahou, K. Chiers, F. Pasmans, and F. Haesebrouck, submitted for publication). In this study, 6-week-old piglets that were free of H. suis were used. Nine piglets were intragastrically inoculated with a pure culture of H. suis, while five sham-inoculated piglets were used as controls. All piglets were fed a finely ground diet. Hyperkeratosis and ulcer formation were clearly present in the gastric nonglandular mucosa of all H. suis-inoculated pigs, while none of the sham-inoculated piglets developed gastric lesions.
Hyperkeratosis and ulceration of the nonglandular part of the stomach have been reported in many countries. Up to 80% of the market pigs in Australia (189) and 60% of the sows (106) in The Netherlands showed gastric lesions. Hessing et al. (106) found gastric ulcers in 10 to 15% of the sows.
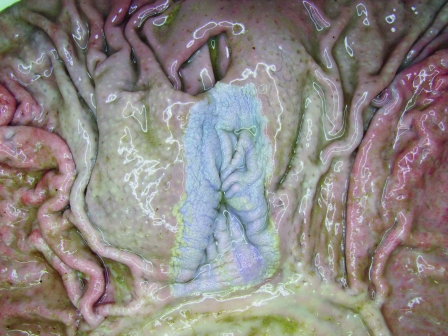
The stomach mucosa of pigs can be divided into a glandular part (cardiac gland zone, fundic gland zone, and antrum with pyloric glands) and a nonglandular part, the latter being a small rectangular area around the esophageal opening. It is also called the pars esophagea of the stomach and is covered by a stratified squamous epithelium (Fig. 3). After experimental infection, H. suis colonizes mainly the antrum and the fundic gland zone and, to a lesser extent, the cardiac gland zone (103). H. suis DNA was also detected in the pars esophagea by PCR (190), but bacteria were not detected in the nonglandular part of the stomach by microscopic examination (103). Ulceration of the porcine gastric nonglandular mucosa may result in decreased feed intake, a decrease in daily weight gain, and even sudden death (10), thus leading to significant economic losses. There is little doubt that this disease can cause pain and discomfort.
FIG. 3.
In the normal porcine stomach there is a small rectangular area around the cardia which is covered by a slightly keratinized squamous epithelium presenting as a white, slightly irregular surface on visual inspection. This area is named the pars esophagea, since the epithelium is similar to that of the esophagus.
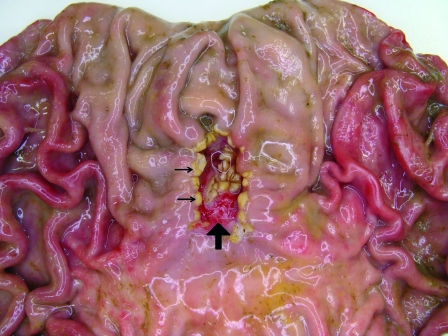
The nonglandular region and the cardiac gland zone, together representing almost 50% of the stomach, have a pH range of between 5 and 7 due to the presence of saliva and cardiac gland bicarbonate secretions (109). The distal compartment, composed of the fundic and pyloric glands, ensures postprandial pepsin digestive enzymatic activity through acid secretion. Pepsin activity is only possible at the low pH of the distal compartment. It has been suggested that no mixing of luminal content takes place between the proximal and the distal stomach compartments and that the porcine stomach normally maintains these two compartments with distinct pH and enzymatic conditions (58). Anything contributing to a breakdown in the segregation of the proximal and distal compartments may allow the stratified squamous epithelium of the nonglandular region to come into contact with the luminal content of the distal part with acid, bile (refluxed from the duodenum), and pepsin. Chronic insult of the nonglandular region will eventually lead to ulceration (Fig. 4).
FIG. 4.
In contrast to the case for most other animal species and for humans, pigs do not usually develop stomach ulcers in the pyloric antrum but rather develop them in the pars esophagea. These lesions are characterized by hyperkeratosis, which typically presents as a bile-stained thickening of the mucosa (small arrows). This can evolve to clefts, erosions, and ulcerations (large arrow).
Ulceration in the nonglandular stomachs of pigs is a disease of complex etiology in which multiple factors are involved, including dietary and stress factors. Small particle size of feed, interruption of feed intake, and presence of highly fermentable carbohydrates in the diet promote ulcera (10). In general, all conditions increasing the fluidity of the stomach contents may cause a breakdown of the pH gradient between the proximal and the distal parts of the stomach and may play a role in ulcer development (77). An infection with H. suis may result in secretion of excessive amounts of gastric acid, leading to increased contact of the nonglandular part of the stomach with hydrochloric acid. In the fundic gland regions of pigs experimentally or naturally infected with H. suis, these microorganisms were found in close contact with parietal cells, which might indicate that the bacterium may have an impact on these hydrochloric acid-producing cells (103). An H. suis infection results in gastritis, which is mainly localized in the antrum (103, 147). In H. pylori infections in humans, increased acid production has been associated with antral predominant gastritis (132). In a recent study, Sapierzynski et al. (192) demonstrated that an H. suis infection in pigs results in an increased number of gastrin-producing cells and a decreased number of somatostatin-producing cells. Since gastrin stimulates and somatostatin inhibits the secretion of hydrochloric acid by parietal cells, this may also result in excessive acid production. However, Silva et al. (202) did not find increased postprandial serum gastrin concentrations in pigs with ulceration of the pars esophagea.
Krakowka et al. (129) isolated a curve-shaped Helicobacter species from naturally infected young piglets that was different from the tightly coiled H. suis. This microorganism is morphologically similar to but antigenically different from H. pylori. As far as we know, no genomic data on this H. pylori-like bacterium have been published yet. Gnotobiotic piglets experimentally inoculated with this microorganism developed ulcers of the pars esophagea (130).
Hänninen et al. (93, 94) demonstrated that spindle-shaped microorganisms that had been isolated from the stomachs and feces of pigs and that had tufts of sheathed flagella at both ends and external fibrils outside the cell belonged to the species H. bilis and H. trogontum. These are enterohepatic helicobacters that were originally isolated from mice and rats, respectively, and were provisionally called “flexispira.” The pathogenicity of these microorganisms for pigs is unknown, and their main site of colonization is most probably the lower intestinal tract. They are urease positive, which may help them to survive during passage through the stomach. It remains to be determined whether they are able to colonize the porcine stomach, as has been shown for some other urease-positive enterohepatic helicobacters in other animals, such as H. aurati in Syrian hamsters (177) and H. muridarum in mice (183).
Gastric Helicobacters Associated with Dogs and Cats
The majority of Helicobacter infections of the canine and feline gastric mucosa are mixed infections of various Helicobacter species, including H. felis, H. bizzozeronii, H. salomonis, and “Candidatus Helicobacter heilmannii.” Recently, one additional species was isolated from the stomach of a dog, namely, H. cynogastricus (233), and one additional species was isolated from the stomach of a cat, namely, H. baculiformis (15).
In dogs, spiral-shaped bacteria are commonly found in the stomach. They are present in 67 to 86% of clinically healthy dogs and in 61 to 100% of dogs presenting chronic vomiting (105, 112). In cats, spiral-shaped organisms have been detected in 41 to 100% of the animals investigated, with a slightly higher rate in animals presenting chronic vomiting (62, 80, 105, 112, 161, 171, 172, 236, 243). Bridgeford et al. (23) hypothesized that gastric Helicobacter species may be a cause of feline gastric lymphoma.
The pathogenic significance of gastric Helicobacter species in dogs and cats remains enigmatic and may be Helicobacter species or even strain dependent.
Cats experimentally infected with H. felis presented a pangastric mononuclear infiltration throughout the gastric mucosa, which was equivalent to the inflammatory response in uninfected animals. However, follicular organization of the inflammatory cells was restricted to the infected animals (193, 204).
The only spiral organism which has been identified in dogs with chronic active gastritis, and not in dogs with a normal gastric histology, is H. felis (51). Also, young gnotobiotic dogs experimentally infected with H. felis presented marked lymphoid hyperplasia in the fundus and the body of the stomach (137). These observations suggest a cytopathogenic effect in the canine stomach for at least H. felis, which may be enhanced due to a possible synergistic effect with H. bizzozeronii. However, Simpson and others (204) found a similar degree of inflammation both in mature specific-pathogen-free dogs experimentally infected with H. felis and in uninfected control dogs. These conflicting observations may be due to differences in virulence between different H. felis isolates, as has also been described for H. pylori (59, 132). Very little is known, however, about differences in pathogenicity between different strains within the same species of non-H. pylori helicobacters. De Bock et al. (36) reported significant differences in inflammation scores in the gastric mucosa of SJL mice at 3 weeks following experimental infection with two different H. felis strains, but it is unclear whether this difference in inflammation score persists over time. Inflammation is considered only one aspect of pathogenicity, and it is not known whether these strains differ in other aspects of pathogenicity as well or whether this observation also holds in other animal species.
Gastric and duodenal ulcers are reported infrequently in dogs and cats, and no clear association has been made with Helicobacter infections (25).
Several research groups concluded on the basis of a species-specific ureB PCR (13, 161) that H. bizzozeronii is the predominant Helicobacter species in the canine stomach (182, 230, 241). Using a multiplex PCR, it was found that more than 50% of the Belgian dogs and cats investigated harbored H. felis (230). The prevalence of “Candidatus Helicobacter heilmannii” (designated “H. heilmannii” or “HLO135” at that time) was found to vary from 20 and 100% in both cats and dogs (112, 161, 218, 230, 241). According to Van den Bulck et al. (230), H. felis and “Candidatus Helicobacter heilmannii” (designated “HLO135”) are the predominant Helicobacter species in cats. H. salomonis has only sporadically been detected in both dogs and cats. The prevalence of H. cynogastricus and H. baculiformis in these animal species is presently unknown.
Gastric Helicobacters Associated with Rabbits
To our knowledge, only two reports describe the detection of Helicobacter DNA in the stomachs of rabbits (231, 234). This concerned H. felis and H. salomonis. No attempts were made to cultivate these organisms from rabbits, nor is there anything known about their pathogenicity toward this animal species. Further research is recommended, especially since rabbits are gaining importance as pet animals, often living in intimate contact with their owners.
Gastric Helicobacters Associated with Ferrets
Shortly after the discovery of H. pylori in humans, spiral organisms were isolated from a gastric ulcer of a ferret and from the gastric mucosa of two healthy ferrets (66). These organisms were first described as Campylobacter pylori subsp. mustelae (68). Later they were designated C. mustelae (69) and, finally, H. mustelae (85). Only a minority of ferrets younger than 6 weeks are colonized by this bacterium, in contrast to approximately 100% of the adult ferrets (67). This indicates that widespread colonization occurs after weaning, and it seems to persist throughout the adult life of the ferret (71).
In ferrets naturally infected with H. mustelae, often only a superficial gastritis is present in the corpus region, where these bacteria colonize the mucosal surface (140). In the antrum, however, a diffuse mononuclear gastritis is observed with inflammatory cells often occupying the full thickness of the mucosa (72). In this stomach region, H. mustelae colonizes the surface, gastric pits, and superficial portion of the glands (72). A retrospective study revealed that persistent colonization with H. mustelae over time increases the severity of gastric disease (71).
Gastric and, to a lesser extent, duodenal ulcers have been reported in ferrets infected with H. mustelae (66, 70), and the incidence of gastric ulceration in this animal species varies between 1.4 and 35% (3, 4). However, since the prevalence of H. mustelae is very high in adult ferrets, long-term observations of experimentally infected pathogen-free ferrets are needed to elucidate the exact role of H. mustelae infection in the development of peptic ulcer disease (211).
Fox and coworkers (75) reported on the presence of H. mustelae in the pyloric mucosa of two ferrets suffering from pyloric adenocarcinoma. In both cases, the invasion of neoplastic tubules into the deep submucosa was described. An increased epithelial cell proliferation has also been detected in the gastric mucosa of ferrets infected with H. mustelae. This may play a role in the development of gastric tumors (247). Gastric MALT lymphoma has also been described in ferrets infected with H. mustelae (63). Replacement of normal epithelium by uniform populations of lymphoid cells was seen, with invasion and destruction of the gastric glands. These lymphomas arose in the antrum, where H. mustelae-induced gastritis is most severe. However, for both types of gastric malignancy, evidence remains circumstantial, and the role of H. mustelae in the development of gastric tumors needs to be confirmed (211).
Gastric Helicobacters Associated with Hamsters
H. aurati has been isolated from the stomachs of hamsters. Several not-further-characterized Helicobacter spp. have also been reported to be present in the stomachs of these animals (159, 176), but no further information is available on these species.
H. aurati has been isolated from the inflamed stomachs and ceca of adult Syrian hamsters. Various features, such as the fusiform shape and the presence of periplasmic fibrils, allow morphological discrimination between H. aurati and the three other helicobacters that have thus far been identified in hamsters, namely, H. cholecystus (76), H. mesocricetorum (203), and H. cinaedi (26, 240). The presence of urease activity also distinguishes H. aurati from these three enterohepatic Helicobacter species. The preferential colonization site of H. aurati in hamsters is probably the intestinal tract, particularly the cecum, with subsequent spreading of this bacterial agent to the stomach in selected animals. The coprophagic behavior of hamsters may play a role in this gastric colonization by H. aurati. At present, the exact role of H. aurati in gastric disease of hamsters has not yet been fully clarified, although the organism has been identified in hamsters suffering from chronic gastric inflammation and intestinal metaplasia (176, 177). The same authors reported the presence of another helical, urease-negative Helicobacter species, as well as a smaller, urease-negative Campylobacter sp. in the stomachs of these hamsters. Likewise, Nambiar et al. (159) reported a case of gastritis-associated adenocarcinoma and intestinal metaplasia in a Syrian hamster naturally infected with different Helicobacter species, including H. aurati. They suggested that chronic Helicobacter-associated gastritis in hamsters may develop into an infiltrative gastric adenocarcinoma, similar to what has been described in chronic H. pylori infections in humans. There are no indications that H. aurati is of zoonotic significance.
Gastric Helicobacters Associated with Ruminants
Gastric ulcers regularly occur in calves and adult cattle, with an incidence varying between 2 and 87% (52, 96, 117, 163, 206, 239). “Candidatus Helicobacter bovis” has been demonstrated in the pyloric part of the abomasums of calves and adult cattle but has not yet been cultivated in vitro (44). Although it is highly prevalent in bovines (reference 88 and unpublished results), its involvement in gastric disease in cattle is presently unknown. In contrast, Helicobacter DNA was not detected in the abomasums of 70 goats, using a genus-specific PCR (88).
Gastric Helicobacters Associated with Horses
Gastric ulcers are common in horses, and their incidence in racehorses in active training may exceed 90% (20, 156). Various stress factors, diet, management, and training practices are regarded as potential risk factors (31, 156). The proximal half of a horse's stomach is entirely lined with a stratified squamous epithelium resembling the pars esophagea of the porcine stomach. The more distal portion is the glandular part. Ulcers are most frequently seen close to the junction between the nonglandular and the glandular parts of the stomach (31). In some studies, Helicobacter-like organisms or their DNA have been detected in the stomachs of horses, but their role in development of gastric ulcers remains speculative. Contreras et al. (31) detected Helicobacter-like DNA in the gastric mucosa of 11 thoroughbred racehorses. Sequencing of the 16S rRNA gene revealed 99% similarity with H. pylori, but all samples were negative when tested with H. pylori-specific PCR assays targeting the cagA and glmM genes, which might indicate that the DNA was from a Helicobacter species different from H. pylori. It remains to be determined whether horses may indeed be infected with a gastric Helicobacter species specifically associated with this animal host. Attempts to isolate these microorganisms from the horse's stomach should be made, both in vitro and in vivo by intragastric inoculation of specific-pathogen-free mice (166).
Gastric Helicobacters Associated with Nonhuman Primates
In several studies, nonhuman primates were used as models for human H. pylori infections (111, 128, 209, 210, 212). Captive rhesus monkeys (Macaca mulatta) are commonly infected with H. pylori (54). The rhesus monkey model, therefore, provides an opportunity to examine natural acquisition of H. pylori using an experimental setup that closely resembles human infection. Socially housed rhesus monkeys rapidly acquire H. pylori infection. Newborns from infected dams are more commonly infected than those from uninfected dams, particularly during the peripartum period, suggesting that close contact during this time facilitates oral-oral transmission (213, 214). Once acquired, infection is associated with chronic gastritis that resembles that seen in humans.
The number of reports dealing with natural infections with gastric non-H. pylori helicobacters in nonhuman primates is limited, and at present, a gastric Helicobacter species specifically associated with these animals has not yet been described. Bronsdon et al. (24) isolated and described H. nemestrinae from the stomach of a pigtailed macaque (Macaca nemestrina). This microorganism is able to grow at 42°C and possesses bipolar flagella, which is different from the case for H. pylori strains isolated from humans. Based on the sequencing of seven housekeeping genes and two flagellin genes, H. nemestrinae was later shown to be an atypical H. pylori strain (219). H. nemestrinae should therefore be considered a later heterotypic synonym of H. pylori.
In the stomachs of rhesus monkeys (Macaca mulatta), gastric non-H. pylori helicobacters which were not identified to the species level have been observed in the mucus covering the surface epithelial cells, in the lumina of the gastric glands, and in close contact with parietal cells (57). These microorganisms were able to invade and on occasion to damage parietal cells, while apparently causing hyperchlorhydria. This is in contrast to the case for H. pylori, which caused gastritis in these animals without modifying the acid output (57). Long, spiral-shaped bacteria have also been reported in the stomachs of baboons (Popio hamadryas). This was associated with gastritis by Mackie and O'Rourke (138) but not by others (34, 35). Non-H. pylori helicobacters, without clarification about the species, have been described to be naturally present in the stomachs of up to 100% of cynomolgus monkeys from many different geographic regions (55, 187). These microorganisms were found in the superficial portions of the gastric epithelium, most frequently in the fundic region. The bacteria were located in the gastric pits, in the superficial glands, or on the surface epithelium. However, no correlation was observed between the presence of these bacteria and the infiltration of lymphoplasmacytic cells and inflammatory lesions in these gastric tissues (55).
H. suis has been demonstrated in the stomachs of two mandrill monkeys (Papio sphinx), two cynomolgus monkeys (Macaca fasicularis), and one rhesus macaque (Macaca mulatta) from a zoo (167). One isolate, first described as “H. heilmannii” (114) and later identified as “Candidatus Helicobacter heilmannii” (158), was obtained by intragastric inoculation of mice with gastric tissue from a cynomolgus monkey (Macaca fascicularis). No information on the pathogenic significance of these Helicobacter species for nonhuman primates is available, and the source of infection remains to be determined.
Although it is clear that nonhuman primates may be infected with different types of gastric helicobacters, little information on these bacteria and their interactions with these hosts is available. Since these animals are closely related to humans, further research is wanted. Monkeys might serve as a possible reservoir for human infections.
NON-H. PYLORI HELICOBACTER-ASSOCIATED GASTRIC DISEASES IN HUMANS: ZOONOSES?
Although some data in the literature indicate that animals, including cats, dogs, and sheep, occasionally may be infected with H. pylori (53, 74, 226), it is unlikely that animals play an important role in the transmission of this microorganism to humans. Moreover, it cannot be excluded that in some of these cases H. pylori-like organisms, but not H. pylori itself, were involved. H. pylori has been demonstrated by culture and PCR methods in the gastric mucosa of specific-pathogen-free laboratory cats in one study (90). This observation may be related to an anecdotic anthroponosis, especially since these bacteria have not been identified in stray cats (62).
Human infections with non-H. pylori Helicobacter organisms, however, most likely originate from animals, although the fingerprinting of Helicobacter species present in the human and animal gastric mucosa should be considered in order to fully understand the zoonotic hazard originating from different animal species.
O'Rourke et al. (166) demonstrated that 16S rRNA gene sequences and partial ureA and ureB gene sequences from three human and four porcine non-H. pylori Helicobacter strains isolated in vivo by inoculation in specific-pathogen-free mice showed a very high degree of homology, ≥99.3%. This led to the conclusion that they represent the same species, later described as H. suis (14, 166).
Another human non-H. pylori Helicobacter strain, also isolated in vivo in specific-pathogen-free mice, could be easily differentiated from H. suis. Urease gene sequence analysis demonstrated that it clustered with helicobacters from domestic and exotic feline species. These microorganisms were designated “Candidatus Helicobacter heilmannii” (166).
Two urease-based PCRs, one developed by O'Rourke et al. (166) and another one by Neiger et al. (161), detected only “Candidatus Helicobacter heilmannii” DNA and not DNA from pure in vitro cultures of H. suis, H. felis, H. salomonis, H. bizzozeronii, H. baculiformis, and H. cynogastricus strains (unpublished results) and can therefore be considered species-specific. Using the test described by Neiger et al. (161), Chisholm and Owen (27) demonstrated the presence of Candidatus Helicobacter heilmannii DNA in one of 113 gastric biopsies from human patients with dyspeptic symptoms.
Trebesius et al. (227) used FISH and partial 16S ribosomal sequencing for analyzing 89 gastric biopsy samples from humans in Germany with histological evidence of non-H. pylori helicobacters. Five short 16S rRNA-directed probes of about 20 nucleotides were used in FISH. In total, 71 (80%) of these samples hybridized with a probe designated Hhe-1, which recognizes fragments of the 16S rRNA gene of H. suis. The 16S ribosomal gene sequences of the former “H. heilmannii” type 2 are highly related, and results obtained with the other probes are therefore more difficult to interpret. Two probes (Hhe2 and Hhe4) are identical to fragments of the 16S rRNA gene of “Candidatus Helicobacter heilmannii,” leading to the conclusion that DNA of this helicobacter was detected in 17 (19%) of the samples. Probe Hhe5 recognizes the 16S rRNA gene of H. felis, H. bizzozeronii and H. salomonis. Five samples (6%) hybridized with this probe. Finally, one sample hybridized with probe Hhe3. It is possible that this probe recognizes a not-yet-described Helicobacter species, since the sequence obtained from HHLO-3 (GenBank accession no. AY014859) shows less than 97% similarity with any of the known Helicobacter species. More research is needed to confirm this, however.
De Groote et al. (45) used three PCR assays targeting the 16S rRNA gene for screening of paraffin-embedded gastric biopsy specimens of 101 patients with chronic active gastritis and histological evidence of a non-H. pylori Helicobacter infection. Fourteen samples tested positive in a PCR assay that specifically detected H. suis DNA. DNA of “Candidatus Helicobacter bovis” was detected in one sample. Samples of 49 patients tested positive in a third assay that simultaneously detected H. felis, H. bizzozeronii, H. salomonis, “Candidatus Helicobacter heilmannii,” H. bacculiformis, and H. cynogastricus.
Van den Bulck et al. (230) studied the presence of Helicobacter species in 123 gastric biopsies of humans from Belgium and Germany with histological evidence of a non-H. pylori Helicobacter infection, using a multiplex PCR based on the tRNA intergenic spacers, the urease gene, and the 16S rRNA gene (13). In 37% of the samples, H. suis was detected. In descending order, H. salomonis (21%), H. felis (15%), “Candidatus Helicobacter heilmannii” (8%, designated HLO135 by the authors), and H. bizzozeronii (4%) were found.
The data presented above show that H. suis is the most prevalent gastric non-H. pylori Helicobacter species in humans and that there are strong indications that pigs may be a source of infection for humans. Apart from the stomachs of pigs and humans, H. suis has also been detected in the stomachs of macaques and mandrill monkeys, as has been demonstrated by 16S rRNA gene and urease gene sequencing (167), and in one cat (230).
Dogs, cats, and perhaps also pet rabbits may serve as a source of infection in humans with H. felis, “Candidatus Helicobacter heilmannii,” and H. bizzozeronii. Anecdotal reports of the presence of the same Helicobacter species in the stomach of a person and his favorite pet animal further point in the same direction. Several reports indeed suggest the transmission of gastric non-H. pylori spiral bacteria from dogs to humans (39, 116, 226) or from cats to humans (50, 107, 133, 217, 235). In 1999, Andersen et al. (2) succeeded in the isolation of a non-H. pylori helicobacter strain from human gastric mucosa. Later, phenotypic analysis, sequencing of the 16S rRNA gene, DNA-DNA hybridization analysis, and whole-cell protein profiling revealed that this isolate belongs to the species H. bizzozeronii (116). Recently, another in vitro isolate was obtained from a human gastric non-H. pylori Helicobacter infection. Despite the low prevalence of H. bizzozeronii in human biopsies, polyphasic identification analysis revealed that this isolate also belonged to this species (127). In the study by Van den Bulck et al. (230), H. salomonis represented 21% of the human samples. The frequent identification of H. salomonis in human gastric biopsies, however, is in contrast to its rare identification in pet carnivore samples, thus inclining us to suspect additional sources of infection. In none of the studies described above were tests that specifically detect H. baculiformis and H. cynogastricus included, and it is presently not known whether these recently described species are able to colonize the human stomach.
GASTRIC DISEASE IN HUMANS INFECTED WITH NON-H. PYLORI HELICOBACTER SPECIES
Studying the effects of non-H. pylori helicobacters in humans is complicated by the fact that these infections are uncommon and it seems likely that there may be variation among non-H. pylori helicobacters in their ability to cause inflammation or disease in humans. In addition, the presence of H. pylori must be excluded in order to assess the effects of the species under consideration. De Groote et al. (45) detected H. pylori DNA in 7 of 64 and Van den Bulck et al. (230) in 6 of 89 human gastric biopsy specimens that were positive for non-H. pylori helicobacters. Human H. pylori and non-H. pylori Helicobacter coinfections have also been identified by histology (113), but the possible significance of this in terms of disease development has not been determined.
Non-H. pylori Helicobacter infections of the human stomach are consistently accompanied by active chronic gastritis. The lesions, however, appear less severe than those associated with H. pylori (216). Acute gastritis is also occasionally observed (246). There may be glandular atrophy or intestinal metaplasia of the fundic mucosa, but these lesions are less common with non-H. pylori Helicobacter than with H. pylori infections (216). Gastric erosions located mainly in the antrum (22, 41, 50, 199, 221, 246) and duodenal ulcers (21, 81, 118, 194) have also been reported in association with non-H. pylori Helicobacter infections. Furthermore, these infections have been associated with low-grade MALT lymphoma of the stomach, and the risk of developing MALT lymphoma is higher with non-H. pylori helicobacters than with H. pylori (153, 154). Both the gastritis and the MALT lymphomas have been reported to resolve after clearance of the non-H. pylori Helicobacter infections, further underlining the causal relationship (154).
Clinical symptoms associated with non-H. pylori helicobacters in humans can be characterized by atypical complaints such as acute or chronic epigastric pain and nausea. Other aspecific symptoms include hematemesis, recurrent dyspepsia, irregular defecation frequency and consistency, vomiting, heartburn, and dysphagia, often accompanied by a decreased appetite (50, 81, 101, 123, 148, 164, 194, 199, 221, 235, 238, 245, 246). Some people infected with non-H. pylori helicobacters do not present obvious clinical signs (142).
Inspection of the gastric mucosa of people infected with long spiral bacteria through endoscopy reveals a variety of lesions, ranging from a normal to slightly hyperemic mucosa to mucosal edema and to multiple erosions and ulcerations in the antrum or in the duodenum (50, 81, 194, 199, 221, 235, 244, 246).
Histologically, the inflammation induced by non-H. pylori helicobacters in the gastric tissue is generally characterized by lymphocytic exudation into gastric foveolae, sometimes admixed with plasma cells. In some cases, lymphocytes are organized into lymphoid aggregates. Epithelial mucus is occasionally depleted (65, 113, 123, 154, 164, 194).
In human patients presenting severe pathology and clinical symptoms associated with the presence of non-H. pylori helicobacters, treatment is indicated, although the efficacy of such treatment is not always easy to determine due to the lack of randomized trials. Such trials are difficult to organize in view of the low frequency of these infections in humans. In practice, treatment regimens identical to those used for H. pylori have been prescribed. Triple therapy using combinations of a proton pump inhibitor and two antimicrobial agents selected from clarithomycin, metronidazole, amoxicillin, and tetracycline may be effective (39, 81, 123, 221, 235).
Because of the low number of in vitro isolates available, very little data exist on the antimicrobial susceptibility and acquired resistance of gastric non-H. pylori Helicobacter species. Determination of MICs of various antimicrobials against in vitro isolates indicated that acquired resistance to metronidazole may occur in H. bizzozeronii and H. felis strains of animal origin (232). Experimental H. felis infections in mice showed that several therapies using only one antimicrobial were effective in only 25 to 70% of the animals tested, while triple therapy using metronidazole, tetracycline, and bismuth subcitrate eradicated H. felis from all the animals (49). In a BALB/c mouse model it was shown that treatment with ampicillin/omeprazole results in the suppression of H. suis. Differences in sensitivity were seen between different H. suis isolates, which might indicate acquired antimicrobial resistance (102).
TRANSMISSION OF GASTRIC NON-H. PYLORI HELICOBACTER SPECIES
Few data are available on how non-H. pylori Helicobacter species are transmitted among their hosts.
Hellemans et al. (104) screened stomach samples of pigs of different ages from different herds for the presence of H. suis. The prevalence of the infection was very low in suckling piglets, increased rapidly after weaning, and reached 90% in the adult boars and sows. The low degree of infection before weaning despite the high prevalence in adult pigs and thus most probably in the dams of the suckling piglets may indicate maternal protection through antibodies or other antibacterial factors present in sow milk (89), which disappears at weaning. The regrouping of animals at the time of weaning most probably favors the spread of the microorganism from the few piglets infected before weaning to noninfected animals. Another factor that may enhance H. suis colonization in weaned pigs is that after weaning the lactobacilli disappear temporarily from the stomach. An antagonistic effect of lactobacilli against H. pylori (1, 119, 200) and H. felis (30) infection has been demonstrated in a murine model. The fact that the H. suis infection persisted in the adult boars and sows indicates that any natural immune response against this microorganism did not lead to its clearance. Despite numerous attempts, we were not able to detect H. suis in feces of infected pigs, which may indicate that fecal-oral spread between pigs and from pigs to humans is limited. Transmission of H. suis may be oral-oral via saliva or gastric-oral via vomitus, but this remains to be investigated. Persistent stomach colonization with H. suis can be achieved in laboratory mice (102), and it may be worthwhile to determine whether wild mice can act as vectors or even as reservoirs of the infection.
Hardly any data on the transmission of non-H. pylori Helicobacter infections in dogs and cats are available in the literature. Hänninen et al. (91) described the transmission of H. salomonis from a dam to her puppies, as well as between infected and noninfected pups, which was proved by the similar pattern found for all cultured isolates using pulsed-field gel electrophoresis. Transmission is suspected to be through oral-oral or gastric-oral contact, as nursing dogs have very intimate contact with their offspring and puppies eat material vomited by the dam. Helicobacter DNA was detected in the oral cavities of dogs (186). Lee et al. (135) found that gastric mucus derived from Helicobacter-infected cats was highly infectious for mice, while rectal contents were not. Moreover, the isolation procedure starting from fecal material was unsuccessful (91). These observations may indicate that fecal-oral transmission is less important.
Fecal-oral transmission has, however, been suggested for H. mustelae (73). In a group of 36 ferrets, H. mustelae was isolated from the feces of 11 animals. A correlation was found with periods of transient hypochlorhydria, also seen in experimentally infected animals (72), which may allow larger numbers of H. mustelae to exit the stomach. Keeping in mind the ease by which ferrets vomit, oral-oral and gastric-oral contact may also play a role in transmission of this bacterium (71).
It is not exactly known how gastric helicobacters are transmitted from animals to humans, but most likely it occurs through direct contact. Living in close proximity to dogs, cats, and especially swine has indeed been identified as a significant risk factor for these infections (108, 144, 220). The intensity of contact with animals is thought to be important as well, since a higher incidence of these infections has been noted in pig farmers, the staff of pig slaughterhouses, and people having intensive contact with pet animals (217, 221, 235, 246). It is remarkable that H. suis is the most prevalent gastric non-H. pylori Helicobacter species in humans (27, 227, 230). This might indicate that the infectivity in humans of cat- or dog-related strains is less than that of H. suis.
It is not known whether, besides direct contact with animals, other routes of transmission of non-H. pylori helicobacters are of importance. Recently, it was shown that gastric helicobacters can survive in water for more than 4 days, a fact which may suggest a possible role for water in the transmission of Helicobacter species between hosts (11). There are no data available on the survival of H. suis on carcasses of slaughtered pigs, and it remains to be determined whether raw or undercooked pork meat might be a source of infection for humans.
It is also not known how frequently transmission of non-H. pylori helicobacters from animals to humans occurs. Only in a low percentage of human patients with severe gastric complaints are long spiral-shaped bacteria found at microscopic examination of gastric biopsies. However, it is possible that this represents only the tip of the iceberg, and it cannot be excluded that infections with these bacteria often pass while remaining unapparent or result in mild disease signs which are not further examined (142).
VIRULENCE FACTORS OF GASTRIC NON-H. PYLORI HELICOBACTER SPECIES
Most of the research concerning Helicobacter virulence factors and the evoked host response has been done with H. pylori, and much less information is available about the virulence mechanisms of non-H. pylori helicobacters. Although some virulence factors of these bacteria may indeed be similar to those described for H. pylori, there may also be differences.
H. pylori is a diverse pathogen, and several bacterial virulence factors are considered to play a role in pathogenesis of infections with this agent. The key enzymes and proteins found in H. pylori that are important for colonization include the urease system, alpha carbonic anhydrase, sheathed flagella, the pH taxis tlpB gene, arginase, and several adhesins. Key virulence factors also include the cag pathogenicity island (cag PAI) and the vacuolating toxin VacA. Describing these virulence factors in detail goes beyond the scope of this article, and readers are referred to recent reviews dealing with this subject (1a, 100, 132, 152, 160, 179).
All gastric Helicobacter species require a family of genes that are involved in the production of urease. This enzyme consists of two subunits, UreA and UreB. It hydrolyzes urea to ammonia and carbon dioxide and is an important mechanism of survival that is required for colonization of the stomach. The ammonia produced neutralizes the hydrochloric acid of the stomach, creating a neutral microenvironment around the bacterium. Urease is localized mainly in the cytoplasm but also becomes associated with the surface of the viable bacteria after autolysis of surrounding bacteria (131, 139, 178). In vitro, this autolysis occurs at the culture stage when the growth of H. pylori ceases (78). Although urease has been associated with the outer membrane (16), the concentration of surface-bound urease is probably too low to contribute to acid resistance (197). The proton-gated urea channel UreI regulates the rate of the urea entry into the cytoplasm and is required for acid survival and gastric infection (151, 205, 237). When UreI is activated by an acidic pH of the medium, urease moves from the inner portion to the outer portion of H. pylori, closer to the source of urea, so presumably ammonia production occurs at or near the inner membrane (110). In H. felis, a second urease system, UreA2B2, has been detected (180), but its function and regulation are currently unknown. One study showed that an H. pylori strain unable to produce functional urease was able to colonize and damage the gastric mucosa of Mongolian gerbils (150). This seems to contrast with results from many other studies stating that urease is essential for colonization of the stomachs of several animals, including gnotobiotic piglets (61). An isogenic urease-negative mutant of H. mustelae (208) produced no detectable urease and failed to colonize the ferret stomach (5). In any case, the role of urease seems not to be limited to colonization. Ammonia is probably also used as a nitrogen source (242) and is thought to assist in damaging the mucosal barrier, thereby releasing nutrients for the bacterium and maintaining the inflammation process (180, 207). In vitro experiments have shown that H. pylori urease is capable of activating peripheral blood mononuclear leukocytes and mucosal macrophages, resulting in production of proinflammatory cytokines (98, 99). Moreover, the H. pylori urease B subunit is able to induce NF-κB activation and interleukin-8 production (19). Urease may also play a role in chemotaxis (157).
Motility is essential for stomach colonization by helicobacters, allowing them to move toward the gastric mucosa (122, 170), which has a neutral pH. Gastric helicobacters possess monopolar, bipolar, or peritrichous bundles of 2 to 23 flagella. The flagella consist of a body, hook, and flagellar filament. The filament is composed of two flagellin subunits, namely, the predominant FlaA and the minor FlaB. It works as a propeller and is covered by a sheath which is suspected to play a role in acid protection, masking of antigens, and maybe adhesion (120). The basal body of the flagellum is embedded in the bacterial cell wall and contains proteins required for rotation and chemotaxis. The hook links the body and the filament. H. mustelae mutants defective in hook production are nonmotile and devoid of flagellar filaments (169). H. mustelae flaA flaB double mutants are completely nonmotile and unable to colonize the ferret, whereas single flaA and flaB mutants have decreased motility (6, 121). These single mutant strains were still able to initially colonize the ferret's stomach at a low level and establish persistent infection, with increasing numbers of organisms over time (6).
Using a microscope slide-based pH gradient assay, it has been shown that H. pylori displays pH-tactic behavior. In response to hydrochloric acid, the microorganism moves away from the strong acid. The chemotaxis receptor TlpB is required for this pH taxis, and tlpB mutants are defective for mouse colonization (33). Homologous genes in gastric non-H. pylori helicobacters have not yet been described, although it seems likely that similar mechanisms exist, allowing them to escape from the highly acidic stomach lumen.
Once highly motile H. pylori bacteria have escaped pH stress, motility decreases or ceases. The ability of H. pylori to stop swimming in response to a neutral pH environment might optimize attachment to gastric epithelial cells (33). While the bacteria can persist deep in the mucus layer, they also attach tightly to gastric epithelial cells via a number of adhesins (162). Nothing is known about the adhesins of non-H. pylori helicobacters, and information about specific localizations of these microorganisms in the stomach is limited. Gerbils have been experimentally infected with H. felis and H. bizzozeronii as a model for the study of pathogenesis and virulence mechanisms. Transmission electron microscopy revealed H. felis bacteria often in close proximity of parietal cells, in contrast to what was found for H. bizzozeronii (38). A close apposition between the microvillus membrane of parietal cells and the outer membrane of the bacteria at the level of the periplasmic fibrils was regularly seen. Bacteria were also found surrounded by necrotic debris of parietal cells. H. suis cells were found in close proximity of mucus-producing epithelial cells and parietal cells of the stomachs of experimentally infected pigs. This has also been seen in naturally infected pigs, and in these animals the bacteria were also found inside the canaliculi of the parietal cells (103). H. mustelae adheres firmly to the gastric epithelium, and only few bacteria are seen lying in the mucus (165). The exact mechanisms promoting its adhesion remain unknown. Most strains of H. mustelae agglutinate red blood cells from various hosts (223). Probably, more than one receptor is involved. In H. mustelae-infected ferrets, the gastric mucosal hydrophobicity is reduced, which is correlated with the degree of mucosal inflammation (84). This may promote the attachment of H. mustelae, which is thought to be mainly hydrophilic. H. mustelae binds to the same receptor lipids as H. pylori, particularly phosphatidylethanolamine (82). Adhesion to eukaryotic cells in vitro correlates with the amount of phosphatidylethanolamine present (83). Clyne et al. (29) showed that flagella do not play a direct role in promoting adherence of H. mustelae to gastric epithelial cells.
Several virulence factors and genes are thought to be important in the pathogenesis of H. pylori infections once contact with the host cell epithelium is established, including VacA and the cag PAI (1a, 17, 100, 132, 160, 179, 229). The cag PAI encodes a type IV secretion system, which forms a syringe-like structure capable of penetrating gastric epithelial cells and delivering CagA into the host cells. Once delivered inside the cell, CagA becomes phosphorylated on tyrosine residues, which are present in EPIYA motifs, resulting in morphological epithelial cell changes (162, 198). H. pylori strains that deliver CagA with more phosphorylation motifs are able to induce more severe cytoskeletal changes and are most often associated with gastric cancer (7, 12). The vacuolating toxin VacA plays a role in the development of vacuoles in epithelial cells and in the induction of apoptosis of these cells (32, 132). It is involved in the osmotic swelling of endosomes into vacuoles (79) and is encoded by the vacA gene, which possesses several polymorphic sites, namely, the signal region, the midregion and the intermediate region. For the first two regions, H. pylori strains with vacA type s1/m1 have been shown to be associated with duodenal and gastric ulceration and gastric adenocarcinoma, whereas the intermediate region is an important marker for H. pylori strains associated with gastric adenocarcinoma (188). Recombinant strains with altered toxicity can emerge during human infection, both for vacA and the cag PAI (9, 124). Moreover, these two genes are thought to downregulate each other's effects on epithelial cells, raising the possibility of avoiding excessive cellular damage (8). Although homologous genes have not been found in gastric non-H. pylori helicobacters, H. felis, in particular, induces extensive apoptosis and necrosis of parietal cells in experimentally infected gerbils (37). H. felis and H. suis induce apoptosis in the murine gastric epithelial cell line GSM06 and in the human gastric adenocarcinoma cell line AGS (unpublished results). Gamma-glutamyl-transpeptidase, which has been associated with the induction of apoptosis in gastric epithelial cells and the inhibition of T-cell proliferation by H. pylori (125, 195, 201), is also produced by gastric non-H. pylori Helicobacter species (Table 2).
H. mustelae produces an array of surface rings which have not yet been described in other Helicobacter species. These rings are composed of the Helicobacter surface ring (Hsr) protein, comprising approximately 25% of the total envelope protein of H. mustelae (168). An Hsr-deficient mutant strain was able to colonize the ferret stomach, but cultures from mutant-dosed ferrets showed reduced levels of bacteria (175). Moreover, animals inoculated with the Hsr-negative strain showed reduced gastric inflammation compared to ferrets infected with the wild-type strain. This underlines the impact of these surface rings on the long-term survival of H. mustelae in the ferret stomach.
STUDYING GASTRIC HELICOBACTER INFECTIONS IN DOMESTIC ANIMALS: WHAT MIGHT IT TEACH US ABOUT THESE INFECTIONS IN HUMANS?
One of the main reasons for the lack of knowledge of the main characteristics and bacterium-host interactions of non-H. pylori helicobacters colonizing the human stomach is that these bacteria are infrequently cultured in vitro, if at all, from gastric biopsies taken from infected humans. Hence, there are two major drawbacks to this approach: the difficulty of obtaining fresh human non-H. pylori spiral bacterium-positive stomach biopsies and the small size of these samples. Tackling this problem from a veterinary perspective may be helpful. Indeed, all known gastric non-H. pylori Helicobacter species infecting humans, except “Candidatus Helicobacter heilmannii” and “Candidatus Helicobacter bovis,” have now been isolated from the stomachs of animals and can be cultivated in vitro (14, 92, 115, 134). This will facilitate the study of bacterium-host interactions and will make it possible to determine the antimicrobial susceptibilities of these microorganisms. Whole-genome sequencing of non-H. pylori helicobacters may be used for detecting genes homologous to H. pylori virulence genes. The H. mustelae and H. suis genome have recently been completed, but results have not yet been published. Studying the genetic diversity of different isolates belonging to a single species may make it possible to determine whether certain genotypes are more often associated with disease in animals and humans than other genotypes. The availability of pure isolates should also enable the development of typing methods, such as multilocus sequence typing, which can be directly applied on gastric tissue. These techniques can then be used to determine whether animal and human strains are clonally related. It can indeed not be excluded at this time that gastric non-H. pylori Helicobacter strains infecting humans may be somewhat different from those found in animals.
CONCLUSIONS
There are clear indications that gastric helicobacters other than H. pylori can cause disease in humans. These tightly coiled microorganisms comprise at least five different Helicobacter species. Diagnostic methods enabling the identification of these bacteria to the species level are needed to help clarify the epidemiology and pathology of these infections in humans. Evidence is accumulating that especially pigs, dogs, and cats constitute reservoir hosts for gastric Helicobacter species with zoonotic potential. The recent successes with in vitro isolation of these fastidious microorganisms from domestic animals open new perspectives for developing typing techniques that can be directly applied on gastric biopsies from humans. These techniques should make it possible to determine whether animal and human strains belonging to the same Helicobacter species are clonally related. The availability of in vitro isolates also opens new perspectives for better understanding the pathogenesis of non-H. pylori Helicobacter-associated gastric pathology and for developing treatment and prevention measures.
Biography
 Freddy Haesebrouck is a veterinarian and Ph.D. from Ghent University, Belgium. In 1988, he became professor in veterinary bacteriology and mycology, and since 1993, he has been head of the Department of Pathology, Bacteriology and Avian Diseases at the Faculty of Veterinary Medicine, Ghent University. His expertise relates primarily to the study of bacterium-host interactions, taxonomy, and antimicrobial resistance. The taxonomic line of research is used as an auxiliary to the study of bacterium-host interactions (characterization of zoonotic bacteria and bacteria that cause disease in animals). His study of Helicobacter spp. began in 1995 and was initially focused on porcine gastric disease. Other studies focused on canine and feline gastric Helicobacter species and enterohepatic helicobacters of poultry and horses. The cultivation method for these bacteria was optimized. This resulted in the isolation and description of new Helicobacter species H. cynogastricus, H. baculiformis, H. suis, and H. equorum.
Freddy Haesebrouck is a veterinarian and Ph.D. from Ghent University, Belgium. In 1988, he became professor in veterinary bacteriology and mycology, and since 1993, he has been head of the Department of Pathology, Bacteriology and Avian Diseases at the Faculty of Veterinary Medicine, Ghent University. His expertise relates primarily to the study of bacterium-host interactions, taxonomy, and antimicrobial resistance. The taxonomic line of research is used as an auxiliary to the study of bacterium-host interactions (characterization of zoonotic bacteria and bacteria that cause disease in animals). His study of Helicobacter spp. began in 1995 and was initially focused on porcine gastric disease. Other studies focused on canine and feline gastric Helicobacter species and enterohepatic helicobacters of poultry and horses. The cultivation method for these bacteria was optimized. This resulted in the isolation and description of new Helicobacter species H. cynogastricus, H. baculiformis, H. suis, and H. equorum.
 Frank Pasmans obtained his degree in veterinary medicine in 1998 and a Ph.D. in veterinary sciences in 2002 at Ghent University, Belgium. After three years of postdoctoral positions at the Department of Pathology, Bacteriology and Avian Diseases, Ghent University, he was appointed a professor in the same department in the disciplines of veterinary bacteriology and mycology and diseases of reptiles and amphibians. His research focuses on host-pathogen interactions of bacteria and fungi with animals. In this respect, gastric non-Helicobacter pylori Helicobacter infections in animals are of special interest because they represent an exciting new area in which nearly all aspects of the pathogenesis await discovery. Recent findings confirm the suspected role of H. suis in gastric disease in swine, rendering this research highly relevant for both animal and human health.
Frank Pasmans obtained his degree in veterinary medicine in 1998 and a Ph.D. in veterinary sciences in 2002 at Ghent University, Belgium. After three years of postdoctoral positions at the Department of Pathology, Bacteriology and Avian Diseases, Ghent University, he was appointed a professor in the same department in the disciplines of veterinary bacteriology and mycology and diseases of reptiles and amphibians. His research focuses on host-pathogen interactions of bacteria and fungi with animals. In this respect, gastric non-Helicobacter pylori Helicobacter infections in animals are of special interest because they represent an exciting new area in which nearly all aspects of the pathogenesis await discovery. Recent findings confirm the suspected role of H. suis in gastric disease in swine, rendering this research highly relevant for both animal and human health.
 Bram Flahou is a veterinarian who graduated at the Faculty of Veterinary Medicine, Ghent University, Belgium. In 2005, he started his Ph.D. studies on animal-associated gastric helicobacters at the Department of Pathology, Bacteriology and Avian Diseases of the same faculty. His research focuses on the interactions of these microorganisms with their hosts, with emphasis on H. suis.
Bram Flahou is a veterinarian who graduated at the Faculty of Veterinary Medicine, Ghent University, Belgium. In 2005, he started his Ph.D. studies on animal-associated gastric helicobacters at the Department of Pathology, Bacteriology and Avian Diseases of the same faculty. His research focuses on the interactions of these microorganisms with their hosts, with emphasis on H. suis.
 Koen Chiers is a veterinarian and Ph.D. from Ghent University, Belgium. In 1995, he became an assistant and later an assistant professor in the Department of Pathology, Bacteriology and Avian Diseases, Faculty of Veterinary Medicine, Ghent University. Currently, he is employed as a professor of veterinary pathology at the same department. In 2006, he became a diplomate of the European College of Veterinary Pathologists (ECVP) and the European College of Porcine Health Management (ECPHM). Since 2001, he has been involved in several studies dealing with animal-associated helicobacters.
Koen Chiers is a veterinarian and Ph.D. from Ghent University, Belgium. In 1995, he became an assistant and later an assistant professor in the Department of Pathology, Bacteriology and Avian Diseases, Faculty of Veterinary Medicine, Ghent University. Currently, he is employed as a professor of veterinary pathology at the same department. In 2006, he became a diplomate of the European College of Veterinary Pathologists (ECVP) and the European College of Porcine Health Management (ECPHM). Since 2001, he has been involved in several studies dealing with animal-associated helicobacters.
 Margo Baele studied bioscience engineering at Ghent University and graduated in 1997. After her studies, she joined the Department of Pathology, Bacteriology and Avian Diseases at the Faculty of Veterinary Medicine of the same university and obtained her Ph.D. in 2001. During this period, she performed taxonomical research, developed PCR-based methods for the identification and typing of bacteria, and described several new bacterial species. After obtaining her Ph.D. degree, she joined the Helicobacter team of the same department and studied the taxonomy of Helicobacter species from dogs, cats, and pigs. This research resulted in the description of several new Helicobacter species and the first cultivation of and taxonomical studies on Helicobacter suis. Since 2008, Margo Baele has joined the Department of Research Affairs of Ghent University, advising researchers in applying for and managing European Research projects in the European Framework Programme.
Margo Baele studied bioscience engineering at Ghent University and graduated in 1997. After her studies, she joined the Department of Pathology, Bacteriology and Avian Diseases at the Faculty of Veterinary Medicine of the same university and obtained her Ph.D. in 2001. During this period, she performed taxonomical research, developed PCR-based methods for the identification and typing of bacteria, and described several new bacterial species. After obtaining her Ph.D. degree, she joined the Helicobacter team of the same department and studied the taxonomy of Helicobacter species from dogs, cats, and pigs. This research resulted in the description of several new Helicobacter species and the first cultivation of and taxonomical studies on Helicobacter suis. Since 2008, Margo Baele has joined the Department of Research Affairs of Ghent University, advising researchers in applying for and managing European Research projects in the European Framework Programme.
 Tom Meyns graduated as a veterinarian in 2002 at the Faculty of Veterinary Medicine, Ghent University, Belgium. In 2007, he obtained a Ph.D. degree for his research on Mycoplasma hyopneumoniae infections in pigs, and thereafter, he became a postdoctoral researcher at the Department of Pathology, Bacteriology and Avian Diseases, Ghent University. His current research focuses on the role of Helicobacter suis in the development of gastric ulcers in pigs.
Tom Meyns graduated as a veterinarian in 2002 at the Faculty of Veterinary Medicine, Ghent University, Belgium. In 2007, he obtained a Ph.D. degree for his research on Mycoplasma hyopneumoniae infections in pigs, and thereafter, he became a postdoctoral researcher at the Department of Pathology, Bacteriology and Avian Diseases, Ghent University. His current research focuses on the role of Helicobacter suis in the development of gastric ulcers in pigs.
 Annemie Decostere received her education at the Faculty of Veterinary Medicine, Ghent University, Belgium. She obtained a Master of Science degree in Aquatic Veterinary Studies at the Institute of Aquaculture, Stirling University, Scotland. Until December 2006, she was employed full-time as a professor of veterinary bacteriology and mycology. Since the beginning of 2007, she has been working at pharma.be (the Belgian association of the pharmaceutical industry) as an Advisor in Public Health and Regulatory Affairs Manager. She is currently still involved as a visiting professor in teaching and supervising scientific research at the faculty listed above. Her active involvement in research on helicobacters in domestic animals started in 2001. This resulted in her being the promoter of various defended doctoral dissertations on helicobacters in domestic animals, mainly focusing on taxonomy and clinical significance both in animals and human beings.
Annemie Decostere received her education at the Faculty of Veterinary Medicine, Ghent University, Belgium. She obtained a Master of Science degree in Aquatic Veterinary Studies at the Institute of Aquaculture, Stirling University, Scotland. Until December 2006, she was employed full-time as a professor of veterinary bacteriology and mycology. Since the beginning of 2007, she has been working at pharma.be (the Belgian association of the pharmaceutical industry) as an Advisor in Public Health and Regulatory Affairs Manager. She is currently still involved as a visiting professor in teaching and supervising scientific research at the faculty listed above. Her active involvement in research on helicobacters in domestic animals started in 2001. This resulted in her being the promoter of various defended doctoral dissertations on helicobacters in domestic animals, mainly focusing on taxonomy and clinical significance both in animals and human beings.
 Richard Ducatelle is a veterinarian and Ph.D. from Ghent University, Belgium. After a few years of working for a Belgian research grant organization as a research grant evaluator, he joined Ghent University, where he became professor of avian pathology, general and special animal pathology, and animal hygiene. Currently, he is a full professor of general and special animal pathology and animal hygiene. The main focus of his research is on interactions between bacteria and the animal host. For many years, he has been interested in the problem of gastric ulcers in domestic animals. A major breakthrough in this field was the recent isolation of Helicobacter suis which, together with a number of other animal helicobacters, appears to play a role in human gastric pathology as well.
Richard Ducatelle is a veterinarian and Ph.D. from Ghent University, Belgium. After a few years of working for a Belgian research grant organization as a research grant evaluator, he joined Ghent University, where he became professor of avian pathology, general and special animal pathology, and animal hygiene. Currently, he is a full professor of general and special animal pathology and animal hygiene. The main focus of his research is on interactions between bacteria and the animal host. For many years, he has been interested in the problem of gastric ulcers in domestic animals. A major breakthrough in this field was the recent isolation of Helicobacter suis which, together with a number of other animal helicobacters, appears to play a role in human gastric pathology as well.
REFERENCES
- 1.Aiba, Y., N. Suzuki, A. M. Kabir, A. Takagi, and Y. Koga. 1998. Lactic acid-mediated suppression of Helicobacter pylori by the oral administration of Lactobacillus salivarius as a probiotic in a gnotobiotic murine model. Am. J. Gastroenterol. 93:2097-101. [DOI] [PubMed] [Google Scholar]
- 1a.Algood, H. M. S., and T. L. Cover. 2006. Helicobacter pylori persistence: an overview of interactions between H. pylori and immune defenses. Clin. Microbiol. Rev. 19:597-613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Andersen, L. P., A. Norgaard, S. Holck, J. Blom, and L. Elsborg. 1996. Isolation of a “Helicobacter heilmannii”-like organism from the human stomach. Eur. J. Clin. Microbiol. Infect. Dis. 15:95-96. [DOI] [PubMed] [Google Scholar]
- 3.Andrews, P. L., O. Illman, and A. Mellersh. 1979. Some observations of anatomical abnormalities and disease states in a population of 350 ferrets (Mustela furo L.). Z. Verstierkd. 21:346-353. [PubMed] [Google Scholar]
- 4.Andrews, P. L., O. Illman, and R. D. Wynne. 1976. Gastric ulceration in the ferret (Mustela furo L.). Z. Verstierkd. 18:285-291. [PubMed] [Google Scholar]
- 5.Andrutis, K. A., J. G. Fox, D. B. Schauer, R. P. Marini, J. C. Murphy, L. Yan, and J. V. Solnick. 1995. Inability of an isogenic urease-negative mutant strain of Helicobacter mustelae to colonize the ferret stomach. Infect. Immun. 63:3722-3725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Andrutis, K. A., J. G. Fox, D. B. Schauer, R. P. Marini, X. Li, L. Yan, C. Josenhans, and S. Suerbaum. 1997. Infection of the ferret stomach by isogenic flagellar mutant strains of Helicobacter mustelae. Infect. Immun. 65:1962-1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Argent, R. H., M. Kidd, R. J. Owen, R. J. Thomas, M. C. Limb, and J. C. Atherton. 2004. Determinants and consequences of different levels of CagA phosphorylation for clinical isolates of Helicobacter pylori. Gastroenterology 127:514-523. [DOI] [PubMed] [Google Scholar]
- 8.Argent, R. H., R. J. Thomas, D. P. Letley, M. G. Rittig, K. R. Hardie, and J. C. Atherton. 2008. Functional association between the Helicobacter pylori virulence factors VacA and CagA. J. Med. Microbiol. 57:145-150. [DOI] [PubMed] [Google Scholar]
- 9.Aviles-Jimenez, F., D. P. Letley, G. Gonzalez-Valencia, N. Salama, J. Torres, and J. C. Atherton. 2004. Evolution of the Helicobacter pylori vacuolating cytotoxin in a human stomach. J. Bacteriol. 186:5182-5185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ayles, H. L., R. M. Friendship, and E. O. Ball. 1996. Effect of dietary particle size on gastric ulcers, assessed by endoscopic examination, and relationship between ulcer severity and growth performance of individually fed pigs. Swine Health Prod. 4:211-216. [Google Scholar]
- 11.Azevedo, N. F., C. Almeida, I. Fernandes, L. Cerqueira, S. Dias, C. W. Keevil, and M. J. Vieira. 2008. Survival of gastric and enterohepatic Helicobacter species in water: implications for transmission. Appl. Environ. Microbiol. 74:1805-1811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Azuma, T., A. Yamakawa, S. Yamazaki, K. Fukuta, M. Ohtani, Y. Ito, M. Dojo, Y. Yamazaki, and M. Kuriyama. 2002. Correlation between variation of the 3′ region of the cagA gene in Helicobacter pylori and disease outcome in Japan. J. Infect. Dis. 186:1621-1630. [DOI] [PubMed] [Google Scholar]
- 13.Baele, M., K. Van den Bulck, A. Decostere, P. Vandamme, M. L. Hanninen, R. Ducatelle, and F. Haesebrouck. 2004. Multiplex PCR assay for differentiation of Helicobacter felis, H. bizzozeronii, and H. salomonis. J. Clin. Microbiol. 42:1115-1122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Baele, M., A. Decostere, P. Vandamme, L. Ceelen, A. Hellemans, K. Chiers, R. Ducatelle, and F. Haesebrouck. 2008. Isolation and characterization of Helicobacter suis sp. nov. from pig stomachs. Int. J. Syst. Evol. Microbiol. 58:1350-1358. [DOI] [PubMed] [Google Scholar]
- 15.Baele, M., F. Haesebrouck, P. Vandamme, K. Van den Bulck, I. Gruntar, J. Mehle, J. Mast, R. Ducatelle, and A. Decostere. 2008. Helicobacter baculiformis sp. nov. isolated from feline stomach mucosa. Int. J. Syst. Evol. Microbiol. 58:357-364. [DOI] [PubMed] [Google Scholar]
- 16.Baik, S. C., K. M. Kim, S. M. Song, D. S. Kim, J. S. Jun, S. G. Lee, J. Y. Song, J. U. Park, H. L. Kang, W. K. Lee, M. J. Cho, H. S. Youn, G. H. Ko, and K. H. Rhee. 2004. Proteomic analysis of the sarcosine-insoluble outer membrane fraction of Helicobacter pylori strain 26695. J. Bacteriol. 186:949-955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Baldwin, D. N., B. Shepherd, P. Kraemer, M. K. Hall, L. K. Sycuro, D. M. Pinto-Santini, and N. R. Salama. 2007. Identification of Helicobacter pylori genes that contribute to stomach colonization. Infect. Immun. 75:1005-1016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Barbosa, A. J. A., J. C. P. Silva, A. M. M. F. Nogueira, E. Paulino, and C. R. Miranda. 1995. Higher incidence of Gastrospirillum sp. in swine with gastric ulcer of the pars oesophagea. Vet. Pathol. 32:134-139. [DOI] [PubMed] [Google Scholar]
- 19.Beswick, E. J., I. V. Pinchuk, K. Minch, G. Suarez, J. C. Sierra, Y. Yamaoka, and V. E. Reyes. 2006. The Helicobacter pylori urease b subunit binds to CD74 on gastric epithelial cells and induces NF-κB activation and interleukin-8 production. Infect. Immun. 74:1148-1155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bezdukova, B., P. Jahn, M. Vyskocil, and J. Plachy. 2005. Gastric ulceration and exercise intensity in standardbred racehorses in Czech Republic. Acta Vet. 74:67-71. [Google Scholar]
- 21.Borody, T. J., L. L. George, S. Brandl, P. Andrews, N. Ostapowicz, L. Hyland, and M. Devine. 1991. Helicobacter pylori-negative duodenal ulcer. Am. J. Gastroenterol. 86:1154-1157. [PubMed] [Google Scholar]
- 22.Boyanova, L., R. Koumanova, E. Lazarova, and C. Jelev. 2003. Helicobacter pylori and Helicobacter heilmannii in children. A Bulgarian study. Diagn. Microbiol. Infect. Dis. 46:249-252. [DOI] [PubMed] [Google Scholar]
- 23.Bridgeford, E. C., R. P. Marini, Y. Feng, N. M. A. Parry, B. Rickman, and J. G. Fox. 2008. Gastric Helicobacter species as a cause of feline gastric lymphoma: a viable hypothesis. Vet. Immunol. Immunopathol. 123:106-113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bronsdon, M. A., C. S. Goodwin, L. I. Sly, T. Chilvers, and F. D. Schoenknecht. 1991. Helicobacter nemestrinae sp. nov., a spiral bacterium found in the stomach of a pigtailed macaque (Macaca nemestrina). Int. J. Syst. Bacteriol. 41:148-153. [DOI] [PubMed] [Google Scholar]
- 25.Brown, C. C., D. C. Baker, and I. K. Barker. 2007. Alimentary system, p. 1-269. In M. G. Maxie (ed.), Jubb, Kennedy and Palmer's pathology of domestic animals, 5th ed., vol. 2. Elsevier Saunders, New York, NY. [Google Scholar]
- 26.Ceelen, L., F. Haesebrouck, R. Ducatelle, and A. Decostere. 2007. The occurrence and clinical significance of enterohepatic Helicobacter species in laboratory rodents. Vlaams Diergen. Tijds. 76:103-116. [Google Scholar]
- 27.Chisholm, S. A., and R. J. Owen. 2003. Development and application of a novel screening PCR assay for direct detection of “Helicobacter heilmannii”-like organisms in human gastric biopsies in Southeast England. Diagn. Microbiol. Infect. Dis. 46:1-7. [DOI] [PubMed] [Google Scholar]
- 28.Choi, Y. K., J. H. Han, and H. S. Joo. 2001. Identification of novel Helicobacter species in pig stomachs by PCR and partial sequencing. J. Clin. Microbiol. 39:3311-3315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Clyne, M., T. Ó. Cróinín, S. Suerbaum, C. Josenhans, and B. Drumm. 2000. Adherence of isogenic flagellum-negative mutants of Helicobacter pylori and Helicobacter mustelae to human and ferret gastric epithelial cells. Infect. Immun. 68:4335-4339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Coconnier, M. H., V. Lievin, E. Hemery, and A. L. Servin. 1998. Antagonistic activity against Helicobacter infection in vitro and in vivo by the human Lactobacillus acidophilus strain LB. Appl. Environ. Microbiol. 64:4573-4580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Contreras, M., A. Morales, M. A. Garcia-Amado, M. De Vera, V. Bermúdez, and P. Gueneau. 2007. Detection of Helicobacter-like DNA in the gastric mucosa of thoroughbred horses. Lett. Appl. Microbiol. 45:553-557. [DOI] [PubMed] [Google Scholar]
- 32.Cover, T. L., U. S. Krishna, D. A. Israel, and R. M. Peek, Jr. 2003. Induction of gastric epithelial cell apoptosis by Helicobacter pylori vacuolating cytotoxin. Cancer Res. 63:951-957. [PubMed] [Google Scholar]
- 33.Croxen, M. A., S. Sisson, R. Melano, and P. S. Hoffman. 2006. The Helicobacter pylori chemotaxis receptor TlpB (HP0103) is required for pH taxis and for colonization of the gastric mucosa. J. Bacteriol. 188:2656-2665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Curry, A., D. M. Jones, and J. Eldridge. 1987. Spiral organisms in the baboon stomach. Lancet ii:634-635. [DOI] [PubMed] [Google Scholar]
- 35.Curry, A., D. M. Jones, and P. Skelton-Stroud. 1989. Novel ultrastructural findings in a helical bacterium found in the baboon (Papio anubis) stomach. J. Gen. Microbiol. 135:2223-2231. [Google Scholar]
- 36.De Bock, M., A. Decostere, K. Van den Bulck, M. Baele, L. Duchateau, F. Haesebrouck, and R. Ducatelle. 2005. The inflammatory response in the mouse stomach to Helicobacter bizzozeronii, Helicobacter salomonis and two Helicobacter felis strains. J. Comp. Pathol. 133:83-91. [DOI] [PubMed] [Google Scholar]
- 37.De Bock, M., A. Decostere, A. Hellemans, F. Haesebrouck, and R. Ducatelle. 2006. Helicobacter felis and Helicobacter bizzozeronii induce gastric parietal cell loss in Mongolian gerbils. Microb. Infect. 8:503-510. [DOI] [PubMed] [Google Scholar]
- 38.De Bock, M., K. D'Herde, L. Duchateau, A. Hellemans, A. Decostere, F. Haesebrouck, and R. Ducatelle. 2006. The pathogenic effect of Helicobacter felis and Helicobacter bizzozeronii on the gastric mucosa in Mongolian gerbils: a sequential pathological study. J. Comp. Pathol. 135:226-236. [DOI] [PubMed] [Google Scholar]
- 39.De Bock, M., K. Van den Bulck, A. Hellemans, S. Daminet, J. C. Coche, J. C. Debongnie, A. Decostere, F. Haesebrouck, and R. Ducatelle. 2007. Peptic ulcer disease associated with Helicobacter felis in a dog owner. Eur. J. Gastroenterol. Hepatol. 19:79-82. [DOI] [PubMed] [Google Scholar]
- 40.Debongnie, J. C., M. Donnay, and J. Mairesse. 1995. Gastrospirillum hominis (“Helicobacter heilmannii”): a cause of gastritis, sometimes transient, better diagnosed by touch cytology? Am. J. Gastroenterol. 90:411-416. [PubMed] [Google Scholar]
- 41.Debongnie, J. C., M. Donnay, J. Mairesse, V. Lamy, X. Dekoninck, and B. Ramdani. 1998. Gastric ulcers and Helicobacter heilmannii. Eur. J. Gastroenterol. Hepatol. 10:251-254. [DOI] [PubMed] [Google Scholar]
- 42.De Groote, D., R. Ducatelle, L. J. Van Doorn, K. Tilmant, A. Verschuuren, and F. Haesebrouck. 2000. Detection of “Candidatus Helicobacter suis” in gastric samples of pigs by PCR: comparison with other invasive diagnostic techniques. J. Clin. Microbiol. 38:1131-1135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.De Groote, D., L. J. Van Doorn, R. Ducatelle, A. Verschuuren, F. Haesebrouck, W. G. Quint, K. Jalava, and P. Vandamme. 1999. “Candidatus Helicobacter suis,” a gastric Helicobacter from pigs, and its phylogenetic relatedness to other gastrospirilla. Int. J. Syst. Bacteriol. 49:1769-1777. [DOI] [PubMed] [Google Scholar]
- 44.De Groote, D., L. J. Van Doorn, R. Ducatelle, A. Verschuuren, K. Tilmant, W. G. V. Quint, F. Haesebrouck, and P. Vandamme. 1999. Phylogenetic characterization of ‘Candidatus Helicobacter bovis,’ a new gastric Helicobacter in cattle. Int. J. Syst. Bacteriol. 49:1707-1715. [DOI] [PubMed] [Google Scholar]
- 45.De Groote, D., L. J. Van Doorn, K. Van Den Bulck, P. Vandamme, M. Vieth, M. Stolte, J. C. Debongnie, A. Burette, F. Haesebrouck, and R. Ducatelle. 2005. Detection of non-pylori Helicobacter species in “Helicobacter heilmannii”-infected humans. Helicobacter 10:398-406. [DOI] [PubMed] [Google Scholar]
- 46.Dent, J. C., C. A. M. McNulty, J. C. Uff, S. P. Wilkinson, and M. W. Gear. 1987. Spiral organisms in the gastric antrum. Lancet ii:96. [DOI] [PubMed] [Google Scholar]
- 47.Dewhirst, F. E., J. G. Fox, E. N. Mendes, B. J. Paster, C. E. Gates, C. A. Kirkbride, and K. A. Eaton. 2000. ‘Flexispira rappini’ strains represent at least 10 Helicobacter taxa. Int. J. Syst. Evol. Microbiol. 50:1781-1787. [DOI] [PubMed] [Google Scholar]
- 48.Dewhirst, F. E., Z. Shen, M. S. Scimeca, L. N. Stokes, T. Boumenna, T. Chen, B. J. Paster, and J. G. Fox. 2005. Discordant 16S and 23S rRNA gene phylogenies for the genus Helicobacter: implications for phylogenetic inference and systematics. J. Bacteriol. 187:6106-6118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Dick-Hegedus, E., and A. Lee. 1991. Use of a mouse model to examine anti-Helicobacter pylori agents. Scand. J. Gastroenterol. 26:909-915. [DOI] [PubMed] [Google Scholar]
- 50.Dieterich, C., P. Wiesel, R. Neiger, A. Blum, and I. Corthesy-Theulaz. 1998. Presence of multiple “Helicobacter heilmannii” strains in an individual suffering from ulcers and in his two cats. J. Clin. Microbiol. 36:1366-1370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Diker, K. S., R. Haziroglu, M. Akan, S. Celik, and N. Kabakci. 2002. The prevalence, colonization sites and pathological effects of gastric helicobacters in dogs. Turk. J. Vet. Anim. Sci. 26:345-351. [Google Scholar]
- 52.Dirksen, G., K. Doll, J. Einhellig, A. Seitz, G. Rademacher, W. Breitner, and W. Klee. 1997. Labmagen geschwüren beim Kalb: klinische Untersuchungen und Erfahrungen. Tierärztl. Prax. 25:318-328. [PubMed] [Google Scholar]
- 53.Dore, M. P., M. Bilotta, D. Vaira, A. Manca, G. Massarelli, G. Leandro, A. Atzei, G. Pisanu, D. Y. Graham, and G. Realdi. 1999. High prevalence of Helicobacter pylori infection in shepherds. Digest. Dis. Sci. 44:1161-1164. [DOI] [PubMed] [Google Scholar]
- 54.Drazek, E. S., A. Dubois, and R. K. Holmes. 1994. Characterization and presumptive identification of Helicobacter pylori isolates from rhesus monkeys. J. Clin. Microbiol. 32:1799-1804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Drevon-Gaillot, E., M.-F. Perron-Lepage, C. Clement, and R. Burnett. 2006. A review of background findings in cynomolgus monkeys (Macaca fascicularis) from three different geographical origins. Exp. Toxicol. Pathol. 58:77-88. [DOI] [PubMed] [Google Scholar]
- 56.Dubois, A., D. E. Berg, E. T. Incecik, N. Fiala, L. M. Heman-Ackah, G. I. Perez-Perez, and M. J. Blaser. 1996. Transient and persistent experimental infection of nonhuman primates with Helicobacter pylori: implications for human disease. Infect. Immun. 64:2885-2891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Dubois, A., A. Tarnawski, D. G. Newell, N. Fiala, W. Dabros, J. Stachura, H. Krivan, and L. M. Heman-Ackah. 1991. Gastric injury and invasion of parietal cells by spiral bacteria in rhesus monkeys. Are gastritis and hyperchlorhydria infectious diseases? Gastroenterology 100:884-891. [DOI] [PubMed] [Google Scholar]
- 58.Dufresne, L. 1998. Alimentary tract disorders of growing pigs, p. 71-77. Proceedings of the 15th IPVS Congress, Birmingham, England.
- 59.Dunn, B. E., H. Cohen, and M. J. Blaser. 1997. Helicobacter pylori. Clin. Microbiol. Rev. 10:720-741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Eaton, K. A., F. E. Dewhirst, M. J. Radin, J. G. Fox, B. J. Paster, S. Krakowka, and D. R. Morgan. 1993. Helicobacter acinonyx sp. nov., isolated from cheetahs with gastritis. Int. J. Syst. Bacteriol. 43:99-106. [DOI] [PubMed] [Google Scholar]
- 61.Eaton, K. A., and S. Krakowka. 1994. Effect of gastric pH on urease-dependent colonization of gnotobiotic piglets by Helicobacter pylori. Infect. Immun. 62:3604-3607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.El-Zaatari, F. A., J. S. Woo, A. Badr, M. S. Osato, H. Serna, L. M. Lichtenberger, R. M. Genta, and D. Y. Graham. 1997. Failure to isolate Helicobacter pylori from stray cats indicates that H. pylori in cats may be an anthroponosis—an animal infection with a human pathogen. J. Med. Microbiol. 46:372-376. [DOI] [PubMed] [Google Scholar]
- 63.Erdman, S. E., P. Correa, L. A. Coleman, M. D. Schrenzel, X. Li, and J. G. Fox. 1997. Helicobacter mustelae-associated gastric MALT lymphoma in ferrets. Am. J. Pathol. 151:273-280. [PMC free article] [PubMed] [Google Scholar]
- 64.Felsenstein, J. 1989. PHYLIP—phylogeny inference package (version 3.2). Cladistics 5:164-166. [Google Scholar]
- 65.Fléjou, J. F., I. Diomandé, G. Molas, D. Goldfain, A. Rotenberg, M. Florent, and F. Potet. 1990. Gastrite chronique associée chez l'homme à la presence de germes spiralés non-Helicobacter pylori (Gastrospirillum hominis). Gastroenterol. Clin. Biol. 14:806-810. [PubMed] [Google Scholar]
- 66.Fox, J. G., B. M. Edrise, E. B. Cabot, C. Beaucage, J. C. Murphy, and K. S. Prostak. 1986. Campylobacter-like organisms isolated from gastric mucosa of ferrets. Am. J. Vet. Res. 47:236-239. [PubMed] [Google Scholar]
- 67.Fox, J. G., E. B. Cabot, N. S. Taylor, and R. Laraway. 1988. Gastric colonization by Campylobacter pylori subsp. mustelae in ferrets. Infect. Immun. 56:2994-2996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Fox, J. G., N. S. Taylor, P. Edmonds, and D. J. Brenner. 1988. Campylobacter pylori subsp. mustelae subsp. nov., isolated from the gastric mucosa of ferrets (Mustela putorius furo), and an emended description of Campylobacter pylori. Int. J. Syst. Bacteriol. 38:367-370. [Google Scholar]
- 69.Fox, J. G., T. Chilvers, C. S. Goodwin, N. S. Taylor, P. Edmonds, L. I. Sly, and D. Brenner. 1989. Campylobacter mustelae, a new species resulting from the elevation of Campylobacter pylori subsp. mustelae to species status. Int. J. Syst. Bacteriol. 39:301-303. [Google Scholar]
- 70.Fox, J. G., P. Correa, N. S. Taylor, A. Lee, G. Otto, J. C. Murphy, and R. Rose. 1990. Helicobacter mustelae-associated gastritis in ferrets. Gastroenterology 99:352-361. [DOI] [PubMed] [Google Scholar]
- 71.Fox, J. G., G. Otto, J. C. Murphy, N. S. Taylor, and A. Lee. 1991. Gastric colonization of the ferret with Helicobacter species: natural and experimental infections. Rev. Infect. Dis. 13(Suppl. 8):S671-S680. [DOI] [PubMed] [Google Scholar]
- 72.Fox, J. G., G. Otto, N. S. Taylor, W. Rosenblad, and J. C. Murphy. 1991. Helicobacter mustelae-induced gastritis and elevated gastric pH in the ferret (Mustela putorius furo). Infect. Immun. 59:1875-1880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Fox, J. G., B. J. Paster, F. E. Dewhirst, N. S. Taylor, L.-L. Yan, P. J. Macuch, and L. M. Chmura. 1992. Helicobacter mustelae isolation from feces of ferrets: evidence to support fecal-oral transmission of a gastric Helicobacter. Infect. Immun. 60:606-611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Fox, J. G. 1995. Non-human reservoirs of Helicobacter pylori. Aliment. Pharmacol. Ther. 9(Suppl. 2):93-103. [PubMed] [Google Scholar]
- 75.Fox, J. G., C. A. Dangler, W. sager, R. Borkowski, and J. M. Gliatto. 1997. Helicobacter mustelae-associated gastric adenocarcinoma in ferrets (Mustela putorius furo). Vet. Pathol. 34:225-229. [DOI] [PubMed] [Google Scholar]
- 76.Franklin, C. L., C. S. Beckwith, R. S. Livingston, L. K. Riley, S. V. Gibson, C. L. Besch-Williford, and R. R. Hook. 1996. Isolation of a novel Helicobacter species, Helicobacter cholecystus sp. nov., from the gallbladders of Syrian hamsters with cholangiofibrosis and centrilobular pancreatitis. J. Clin. Microbiol. 34:2952-2958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Friendship, R. M. 2006. Gastric ulcers, p. 891-899. In B. E. Straw, J. J. Zimmerman, S. D'Allaire, and D. J. Taylor (ed.), Diseases of swine, 9th ed. Blackwell Publishing, Ames, IA.
- 78.Fujita, Y., K. Yamaguchi, T. Kamegaya, H. Sato, K. Semura, K. Mutoh, T. Kashimoto, H. Ohori, and T. Mukai. 2005. A novel mechanism of autolysis in Helicobacter pylori: possible involvement of peptidergic substances. Helicobacter 10:567-576. [DOI] [PubMed] [Google Scholar]
- 79.Genisset, C., A. Puhar, F. Calore, M. de Bernard, P. Dell'Antone, and C. Montecucco. 2007. The concerted action of the Helicobacter pylori cytotoxin VacA and of the v-ATPase proton pump induces swelling of isolated endosomes. Cell. Microbiol. 9:1481-1490. [DOI] [PubMed] [Google Scholar]
- 80.Geyer, C., F. Colbatzky, J. Lechner, and W. Hermanns. 1993. Occurrence of spiral-shaped bacteria in gastric biopsies of dogs and cats. Vet. Rec. 133:18-19. [DOI] [PubMed] [Google Scholar]
- 81.Goddard, A. F., R. P. Logan, J. C. Atherton, D. Jenkins, and R. C. Spiller. 1997. Healing of duodenal ulcer after eradication of Helicobacter heilmannii. Lancet 349:1815-1816. [DOI] [PubMed] [Google Scholar]
- 82.Gold, B. D., M. Huesca, P. M. Sherman, and C. A. Lingwood. 1993. Helicobacter mustelae and Helicobacter pylori bind to common lipid receptors in vitro. Infect. Immun. 61:2632-2638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Gold, B. D., M. Dytoc, M. Huesca, D. Philpott, A. Kuksis, S. Czinn, C. A. Lingwood, and P. M. Sherman. 1995. Comparison of Helicobacter mustelae and Helicobacter pylori adhesion to eukaryotic cells in vitro. Gastroenterology 109:692-700. [DOI] [PubMed] [Google Scholar]
- 84.Gold, B. D., P. Islur, Z. Policova, S. Czinn, A. W. Neumann, and P. M. Sherman. 1996. Surface properties of Helicobacter mustelae and ferret gastrointestinal mucosa. Clin. Investig. Med. 19:92-100. [PubMed] [Google Scholar]
- 85.Goodwin, C. S., J. A. Armstrong, T. Chilvers, M. Peters, M. D. Colins, L. Sly, W. McConnel, and W. E. S. Harper. 1989. Transfer of Campylobacter pylori and Campylobacter mustelae to Helicobacter gen. nov. as Helicobacter pylori comb. nov. and Helicobacter mustelae comb. nov., respectively. Int. J. Syst. Bacteriol. 39:397-405. [Google Scholar]
- 86.Goto, K., H. Ohashi, A. Takakura, and T. Itoh. 2000. Current status of Helicobacter contamination of laboratory mice, rats, gerbils, and house musk shrews in Japan. Curr. Microbiol. 41:161-166. [DOI] [PubMed] [Google Scholar]
- 87.Grasso, G. M., G. Ripabelli, M. L. Sammarco, and A. Ruberto. 1996. Prevalence of Helicobacter-like organisms in porcine gastric mucosa: a study of swine slaughtered in Italy. Comp. Immunol. Microbiol. Infect. Dis. 19:213-217. [DOI] [PubMed] [Google Scholar]
- 88.Gueneau, P., J. Fuenmayor, O. C. Aristimuno, S. Cedeno, E. Baez, N. Reyes, F. Michelangeli, and M. G. Dominguez-Bello. 2002. Are goats naturally resistant to gastric Helicobacter infection? Vet. Microbiol. 84:115-121. [DOI] [PubMed] [Google Scholar]
- 89.Gustafsson, A., A. Hultberg, R. Sjostrom, I. Kacskovics, M. E. Breimer, T. Boren, L. Hammarstrom, and J. Holgersson. 2006. Carbohydrate-dependent inhibition of Helicobacter pylori colonization using porcine milk. Glycobiology 16:1-10. [DOI] [PubMed] [Google Scholar]
- 90.Handt, L. K., J. G. Fox, I. H. Stalis, R. Rufo, G. Lee, J. Linn, X. Li, and H. Kleanthous. 1995. Characterization of feline Helicobacter pylori strains and associated gastritis in a colony of domestic cats. J. Clin. Microbiol. 33:2280-2289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Hänninen, M. L., I. Happonen, and K. Jalava. 1998. Transmission of canine gastric Helicobacter salomonis infection from dam to offspring and between puppies. Vet. Microbiol. 62:47-58. [DOI] [PubMed] [Google Scholar]
- 92.Hänninen, M. L., I. Happonen, S. Saari, and K. Jalava. 1996. Culture and characteristics of Helicobacter bizzozeronii, a new canine gastric Helicobacter sp. Int. J. Syst. Bacteriol. 46:160-166. [DOI] [PubMed] [Google Scholar]
- 93.Hänninen, M. L., R. I. Kärenlampi, J. M. K. Koort, T. Mikkonen, and K. J. Björkroth. 2005. Extension of the species Helicobacter bilis to include the reference strains of Helicobacter sp. flexispira taxa 2, 3 and 8 and Finnish canine and feline flexispira strains. Int. J. Syst. Evol. Microbiol. 55:891-898. [DOI] [PubMed] [Google Scholar]
- 94.Hänninen, M. L., M. Utriainen, I. Happonen, and F. E. Dewhirst. 2003. Helicobacter sp. flexispira 16S rDNA taxa 1, 4 and 5 and Finnish porcine Helicobacter isolates are members of the species Helicobacter trogontum (taxon 6). Int. J. Syst. Evol. Microbiol. 53:425-433. [DOI] [PubMed] [Google Scholar]
- 95.Hannula, M., and M. L. Hänninen. 2007. Phylogenetic analysis of Helicobacter species based on partial gyrB gene sequences. Int. J. Syst. Evol. Microbiol. 57:444-449. [DOI] [PubMed] [Google Scholar]
- 96.Haringsma, P. C., and J. M. V. M. Mouwen. 1992. Mogelijke betekenis van spirilvormige bacteriën bij het ontstaan van lebmaagzweren bij het volwassen rund. Tijdschr. Diergeneesk. 117:485-486. [PubMed] [Google Scholar]
- 97.Harper, C. G., Y. Feng, S. Xu, N. S. Taylor, M. KInsel, F. E. Dewhirst, B. J. Paster, M. Greenwell, G. Levine, A. Rogers, and J. G. Fox. 2002. Helicobacter cetorum sp. nov., a urease-positive Helicobacter species isolated from dolphins and whales. J. Clin. Microbiol. 40:4536-4543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Harris, P. R., H. L. T. Mobley, G. I. Perez-Perez, M. J. Blaser, and P. D. Smith. 1996. Helicobacter pylori urease is a potent stimulus of mononuclear phagocyte activation and inflammatory cytokine production. Gastroenterology 111:419-425. [DOI] [PubMed] [Google Scholar]
- 99.Harris, P. R., P. B. Ernst, S. Kawabata, H. Kiyono, M. F. Graham, and P. D. Smith. 1998. Recombinant Helicobacter pylori urease activates primary mucosal macrophages. J. Infect. Dis. 181:783-786. [DOI] [PubMed] [Google Scholar]
- 100.Hatakeyama, M. 2008. SagA of CagA in Helicobacter pylori pathogenesis. Curr. Opin. Microbiol. 11:30-37. [DOI] [PubMed] [Google Scholar]
- 101.Heilmann, K., and F. Borchard. 1991. Gastritis due to spiral-shaped bacteria other than Helicobacter pylori: clinical, histological and ultrastructural findings. Gut 32:137-140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Hellemans, A., A. Decostere, F. Haesebrouck, and R. Ducatelle. 2005. Evaluation of antibiotic treatment for eradication of “Candidatus Helicobacter suis” in a mouse model. Antimicrob. Agents Chemother. 49:4530-4535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Hellemans, A., K. Chiers, A. Decostere, M. De Bock, F. Haesebrouck, and R. Ducatelle. 2007. Experimental infection of pigs with “Candidatus Helicobacter suis.” Vet. Res. Commun. 31:385-395. [DOI] [PubMed] [Google Scholar]
- 104.Hellemans, A., K. Chiers, D. Maes, M. De Bock, A. Decostere, F. Haesebrouck, and R. Ducatelle. 2007. Prevalence of “Candidatus Helicobacter suis” in pigs of different ages. Vet. Rec. 161:182-192. [DOI] [PubMed] [Google Scholar]
- 105.Hermanns, W., K. Kregel, W. Breuer, and J. Lechner. 1995. Helicobacter-like organisms: histopathological examination of gastric biopsies from dogs and cats. J. Comp. Pathol. 112:307-318. [DOI] [PubMed] [Google Scholar]
- 106.Hessing, J. J. C., M. J. Geudeke, C. J. M. Scheepens, M. J. M. Tielen, W. G. P. Schouten, and P. R. Wiepkema. 1992. Changes in the mucous membrane of the oesophageal part of the stomach-prevalence and relations to stress. Tijdschr Diergeneeskd. 117:445-450. [PubMed] [Google Scholar]
- 107.Hiroshi, O., H. Hiroko, H. Yoshikazu, T. Kazuhiko, M. Kenjiro, M. Yasuhiro, and S. Isao. 2008. Acute gastritis associated with invading Helicobacter heilmannii organisms from a previously homeless cat. Clin. Gastroenterol. 42:216-217. [DOI] [PubMed] [Google Scholar]
- 108.Holck, S., P. Ingeholm, J. Blom, A. Norgaard, L. Elsborg, S. Adamsen, and L. P. Andersen. 1997. The histopathology of human gastric mucosa inhabited by Helicobacter heilmannii-like (Gastrospirillum hominis) organisms, including the first culturable case. APMIS 105:746-756. [DOI] [PubMed] [Google Scholar]
- 109.Höller, H. 1970. Studies on the secretion of the cardiac gland zone in the pig stomach. II. Studies on the influencing of spontaneous secretion of the isolated cardiac gland zone, fluid and electrolyte secretion in the isolated small stomach containing different fluids. Zentralbl. Veterinarmed. A 17:857-873. [PubMed] [Google Scholar]
- 110.Hong, W., K. Sano, S. Morimatsu, D. R. Scott, D. L. Weeks, G. Sachs, T. Goto, S. Mohan, F. Harada, N. Nakajima, and T. Nakano. 2003. Medium pH-dependent redistribution of the urease of Helicobacter pylori. J. Med. Microbiol. 52:211-216. [DOI] [PubMed] [Google Scholar]
- 111.Hornsby, M. J., J. L. Huff, R. J. Kays, D. R. Canfield, C. L. Bevins, and J. V. Solnick. 2008. Helicobacter pylori induces an antimicrobial response in rhesus macaques in a cag pathogenicity island-dependent manner. Gastroenterology 134:1049-1057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Hwang, C. Y., H. R. Han, and H. Y. Youn. 2002. Prevalence and clinical characterization of gastric Helicobacter species infection in dogs and cats in Korea. J. Vet. Sci. 3:123-133. [PubMed] [Google Scholar]
- 113.Ierardi, E., R. A. Monno, A. Gentile, R. Francavilla, O. Burattini, L. Maran, and A. Francavilla. 2001. Helicobacter heilmannii gastritis: a histological and immunohistochemical trait. J. Clin. Pathol. 54:774-777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Itoh, T., Y. Yanagawa, M. Singaki, N. Masubuchi, S. Takahashi, and S. Saito. 1994. Isolation of Helicobacter heilmannii like organism from the stomachs of cynomolgus monkeys and colonization of them in mice. Gastroenterology 106:A99. [Google Scholar]
- 115.Jalava, K., M. Kaartinen, M. Utriainen, I. Happonen, and M. L. Hanninen. 1997. Helicobacter salomonis sp. nov., a canine gastric Helicobacter sp. related to Helicobacter felis and Helicobacter bizzozeronii. Int. J. Syst. Bacteriol. 47:975-982. [DOI] [PubMed] [Google Scholar]
- 116.Jalava, K., S. L. On, C. S. Harrington, L. P. Andersen, M. L. Hänninen, and P. Vandamme. 2001. A cultured strain of “Helicobacter heilmannii,” a human gastric pathogen, identified as H. bizzozeronii: evidence for zoonotic potential of Helicobacter. Emerg. Infect. Dis. 7:1036-1038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Jelinski, M. D., C. S. Ribble, M. Chirino-Trejo, E. G. Clark, and E. D. Janzen. 1995. The relationship between the presence of Helicobacter pylori, Clostridium perfringens type A, Campylobacter spp, or fungi and fatal abomasal ulcers in unweaned beef calves. Can. Vet. J. 36:379-382. [PMC free article] [PubMed] [Google Scholar]
- 118.Jhala, D., N. Jhala, J. Lechago, and M. Haber. 1999. Helicobacter heilmannii gastritis: association with acid peptic diseases and comparison with Helicobacter pylori gastritis. Mod. Pathol. 12:534-538. [PubMed] [Google Scholar]
- 119.Johnson-Henry, K. C., D. J. Mitchell, Y. Avitzur, E. Galindo-Mata, N. L. Jones, and P. M. Sherman. 2004. Probiotics reduce bacterial colonization and gastric inflammation in H. pylori-infected mice. Dig. Dis. Sci. 49:1095-102. [DOI] [PubMed] [Google Scholar]
- 120.Jones, A. C., R. P. Logan, S. Foynes, A. Cockayne, B. W. Wren, and C. W. Penn. 1997. A flagellar sheath protein of Helicobacter pylori is identical to HpaA, a putative N-acetylneuraminyllactose-binding hemagglutinin, but is not an adhesion for AGS cells. J. Bacteriol. 179:5643-5647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Josenhans, C., A. Labigne, and S. Suerbaum. 1995. Comparative ultrastructural and functional studies of Helicobacter pylori and Helicobacter mustelae flagellin mutants: both flagellin subunits, FlaA and FlaB, are necessary for full motility in Helicobacter species. J. Bacteriol. 177:3010-3020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Josenhans, C., R. L. Ferrero, A. Labigne, and S. Suerbaum. 1999. Cloning and allelic exchange mutagenesis of two flagellin genes of Helicobacter felis. Mol. Microbiol. 33:350-362. [DOI] [PubMed] [Google Scholar]
- 123.Kaklikkaya, N., O. Ozgur, F. Aydin, and U. Cobanoglu. 2002. Helicobacter heilmannii as causative agent of chronic active gastritis. Scand. J. Infect. Dis. 34:768-770. [DOI] [PubMed] [Google Scholar]
- 124.Kersulyte, D., H. Chalkauskas, and D. E. Berg. 1999. Emergence of recombinant strains of Helicobacter pylori during human infection. Mol. Microbiol. 31:31-43. [DOI] [PubMed] [Google Scholar]
- 125.Kim, K. M., S. G. Lee, M. G. Park, J. Y. Song, H. L. Kang, W. K. Lee, M. J. Cho, K. H. Rhee, H. S. Youn, and S. C. Baik. 2007. Gamma-glutamyltranspeptidase of Helicobacter pylori induces mitochondria-mediated apoptosis in AGS cells. Biochem. Biophys. Res. Commun. 355:562-567. [DOI] [PubMed] [Google Scholar]
- 126.Kimura, M. 1980. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J. Mol. Evol. 16:111-120. [DOI] [PubMed] [Google Scholar]
- 127.Kivistö, R., J. Linros, H. Rautelin, and M.-L. Hänninen. 2008. Characterization of Helicobacter bizzozeronii cultured from human gastric mucosa, abstr. HP-53. Abstracts 8th International Workshop on Pathogenesis and Host Response in Helicobacter Infections, Helsingör, Denmark.
- 128.Kodama, M., K. Murakami, R. Sato, T. Okimoto, A. Nishizono, and T. Fujioka. 2005. Helicobacter pylori infected animal models are extremely suitable for the investigation of gastric carcinogenesis. World J. Gastroenterol. 11:7063-7071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Krakowka, S., S. S. Ringler, J. Flores, R. J. Kearns, K. A. Eaton, and J. A. Ellis. 2005. Isolation and preliminary characterization of a novel Helicobacter species from swine. Am. J. Vet. Res. 66:938-944. [DOI] [PubMed] [Google Scholar]
- 130.Krakowka, S., M. Rings, and J. A. Ellis. 2005. Experimental induction of bacterial gastritis and gastric ulcer disease in gnotobiotic swine inoculated with porcine Helicobacter-like species. Am. J. Vet. Res. 66:945-952. [DOI] [PubMed] [Google Scholar]
- 131.Krishnamurthy, P., M. Parlow, J. B. Zitzer, N. B. Vakil, H. L. Mobley, M. Levy, S. H. Phadnis, and B. E. Dunn. 1998. Helicobacter pylori containing only cytoplasmic urease is susceptible to acid. Infect. Immun. 66:5060-5066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Kusters, J. G., A. H. M. van Vliet, and E. J. Kuipers. 2006. Pathogenesis of Helicobacter pylori infection. Clin. Microbiol. Rev. 19:449-490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Lavelle, J. P., S. Landas, F. A. Mitros, and J. L. Conklin. 1994. Acute gastritis associated with spiral organisms from cats. Dig. Dis. Sci. 39:744-750. [DOI] [PubMed] [Google Scholar]
- 134.Lee, A., S. L. Hazell, J. O'Rourke, and S. Kouprach. 1988. Isolation of a spiral-shaped bacterium from the cat stomach. Infect. Immun. 56:2843-2850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Lee, A., J. G. Fox, G. Otto, E. Dick-Hegedus, and S. Krakowka. 1991. Transmission of Helicobacter species: a challenge to the dogma of fecal oral spread. Epidemiol. Infect. 107:99-109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Lee, A., M. W. Phillips, J. L. O'Rourke, B. J. Paster, F. E. Dewhirst, G. J. Fraser, J. G. Fox, L. I. Sly, P. J. Romaniuk, T. J. Trust, and S. Kouprach. 1992. Helicobacter muridarum sp. nov., a microaerophilic helical bacterium with a novel ultrastructure isolated from the intestinal mucosa of rodents. Int. J. Syst. Bacteriol. 42:27-36. [DOI] [PubMed] [Google Scholar]
- 137.Lee, A., S. Krakowka, J. G. Fox, G. Otto, K. A. Eaton, and J. C. Murphy. 1992. Role of Helicobacter felis in chronic canine gastritis. Vet. Pathol. 29:487-494. [DOI] [PubMed] [Google Scholar]
- 138.Mackie, J. T., and J. L. O'Rourke. 2003. Gastritis associated with Helicobacter-like organisms in baboons. Vet. Pathol. 40:563-566. [DOI] [PubMed] [Google Scholar]
- 139.Marcus, E. A., and D. R. Scott. 2001. Cell lysis is responsible for the appearance of extracellular urease in Helicobacter pylori. Helicobacter 6:93-99. [DOI] [PubMed] [Google Scholar]
- 140.Marini, R. P., and J. G. Fox. 1999. Animal models of Helicobacter (ferrets), p. 273-284. In O. Zak and M. A. Sande (ed.), Handbook of animal models of infection. Academic Press, London, United Kingdom.
- 141.Marshall, B. J., and J. R. Warren. 1984. Unidentified curved bacilli in the stomach of patients with gastritis and peptic ulceration. Lancet i:1311-1315. [DOI] [PubMed] [Google Scholar]
- 142.Mazzucchelli, L., C. H. Wilder-Smith, C. Ruchti, B. Meyer-Wyss, and H. S. Merki. 1993. Gastrospirillum hominis in asymptomatic, healthy individuals. Dig. Dis. Sci. 38:2087-2089. [DOI] [PubMed] [Google Scholar]
- 143.McNulty, C. A., J. C. Dent, A. Curry, J. S. Uff, G. A. Ford, M. W. Gear, and S. P. Wilkinson. 1989. New spiral bacterium in gastric mucosa. J. Clin. Pathol. 42:585-591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Meining, A., G. Kroher, and M. Stolte. 1998. Animal reservoirs in the transmission of Helicobacter heilmannii. Results of a questionnaire-based study. Scand. J. Gastroenterol. 33:795-798. [DOI] [PubMed] [Google Scholar]
- 145.Melnichouk, S. I., R. M. Friendship, C. E. Dewey, R. J. Bildfell, and N. L. Smart. 1999. Helicobacter-like organisms in the stomach of pigs with and without gastric ulceration. Swine Health Prod. 7:201-205. [Google Scholar]
- 146.Mendes, E. N., D. M. Queiroz, G. A. Rocha, S. B. Moura, V. H. R. Leite, and M. E. F. Fonseca. 1990. Ultrastructure of a spiral micro-organism from pig gastric mucosa (“Gastrospirillum suis”). J. Med. Microbiol. 33:61-66. [DOI] [PubMed] [Google Scholar]
- 147.Mendes, E. N., D. M. Queiroz, G. A. Rocha, A. M. Nogueira, A. C. Carvalho, A. P. Lage, and A. J. Barbosa. 1991. Histopathological study of porcine gastric mucosa with and without a spiral bacterium (“Gastrospirillum suis”). J. Med. Microbiol. 35:345-348. [DOI] [PubMed] [Google Scholar]
- 148.Mention, K., L. Michaud, D. Guimber, E. Martin De Lasalle, P. Vincent, D. Turck, and F. Gottrand. 1999. Characteristics and prevalence of Helicobacter heilmannii infection in children undergoing upper gastrointestinal endoscopy. J. Pediatr. Gastroenterol. Nutr. 29:533-539. [DOI] [PubMed] [Google Scholar]
- 149.Mikkonen, T. P., R. I. Karenlampi, and M. L. Hänninen. 2004. Phylogenetic analysis of gastric and enterohepatic Helicobacter species based on partial HSP60 gene sequences. Int. J. Syst. Evol. Microbiol. 54:753-758. [DOI] [PubMed] [Google Scholar]
- 150.Mine, T., H. Muraoka, T. Saika, and I. Kobayashi. 2005. Characteristics of a clinical isolate of urease-negative Helicobacter pylori and its ability to induce gastric ulcers in Mongolian gerbils. Helicobacter 10:125-133. [DOI] [PubMed] [Google Scholar]
- 151.Mollenhauer-Rektorschek, M., G. Hanauer, G. Sachs, and K. Melchers. 2002. Expression of UreI is required for intragastric transit and colonization of gerbil gastric mucosa by Helicobacter pylori. Res. Microbiol. 153:659-666. [DOI] [PubMed] [Google Scholar]
- 152.Montecucco, C., and R. Rappuoli. 2001. Living dangerously: how Helicobacter pylori survives in the human stomach. Nat. Rev. Mol. Cell Biol. 2:457-466. [DOI] [PubMed] [Google Scholar]
- 153.Morgner, A., E. Bayerdorffer, A. Meining, M. Stolte, and G. Kroher. 1995. Helicobacter heilmannii and gastric cancer. Lancet 346:551-552. [DOI] [PubMed] [Google Scholar]
- 154.Morgner, A., N. Lehn, L. P. Andersen, C. Thiede, M. Bennedsen, K. Trebesius, B. Neubauer, A. Neubauer, M. Stolte, and E. Bayerdorffer. 2000. Helicobacter heilmannii-associated primary gastric low-grade MALT lymphoma: complete remission after curing the infection. Gastroenterology 118:821-828. [DOI] [PubMed] [Google Scholar]
- 155.Mörner, T., C. Bröjer, M.-P. Ryser-Degiorgis, D. Gavier-Widén, H.-O. Nilsson, and T. Wadström. 2008. Detection of gastric Helicobacter species in free-ranging lynx (Lynx lynx) and red foxes (Vulpes vulpes) in Sweden. J. Wildl. Dis. 44:697-700. [DOI] [PubMed] [Google Scholar]
- 156.Murray, M. J. 1994. Gastric ulcers in adult horses. Compendium 16:792-797. [Google Scholar]
- 157.Nakamura, H., H. Yoshiyama, H. Takeuchi, T. Mizote, K. Okita, and T. Nakazawa. 1998. Urease plays an important role in the chemotactic motility of Helicobacter pylori in a viscous environment. Infect. Immun. 66:4832-4837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Nakamura, M., S. Y. Murayama, H. Serizawa, Y. Sekiya, M. Eguchi, S. Takahashi, K. Nishikawa, T. Takahashi, T. Matsumoto, H. Yamada, T. Hibi, K. Tshuchimoto, and H. Matsui. 2007. “Candidatus H. Heilmannii” from a cynomolgus monkey induces gastric mucosa-associated lymphoid tissue lymphomas in C57BL/6 mice. Infect. Immun. 75:1214-1222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Nambiar, P. R., S. Kirchain, and J. G. Fox. 2005. Gastritis-associated adenocarcinoma and intestinal metaplasia in a Syrian hamster naturally infected with Helicobacter species. Vet. Pathol. 42:386-390. [DOI] [PubMed] [Google Scholar]
- 160.Naumann, M., and J. E. Crabtree. 2004. Helicobacter pylori-induced epithelial cell signalling in gastric carcinogenesis. Trends Microbiol. 12:29-36. [DOI] [PubMed] [Google Scholar]
- 161.Neiger, R., C. Dieterich, A. Burnens, A. Waldvogel, I. Corthesy-Theulaz, F. Halter, B. Lauterburg, and A. Schmassmann. 1998. Detection and prevalence of Helicobacter infection in pet cats. J. Clin. Microbiol. 36:634-637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Odenbreit, S. 2005. Adherence properties of Helicobacter pylori: inpact on pathogenesis and adaptation to the host. Int. J. Med. Microbiol. 295:317-324. [DOI] [PubMed] [Google Scholar]
- 163.Ok, M., I. Sen, K. Turgut, and K. Irmak. 2001. Plasma gastrin activity and the diagnosis of bleeding abomasal ulcers in cattle. J. Vet. Med. A 48:563-568. [DOI] [PubMed] [Google Scholar]
- 164.Oliva, M. M., A. J. Lzaenby, and J. A. Perman. 1993. Gastritis associated with Gastrospirillum hominis in children. Comparison with Helicobacter pylori and review of the literature. Modern Pathol. 6:513-515. [PubMed] [Google Scholar]
- 165.O'Rourke, J., A. Lee, and J. G. Fox. 1992. An ultrastructural study of Helicobacter mustelae and evidence of a specific association with gastric mucosa. J. Med. Microbiol. 36:420-427. [DOI] [PubMed] [Google Scholar]
- 166.O'Rourke, J. L., J. V. Solnick, B. A. Neilan, K. Seidel, R. Hayter, L. M. Hansen, and A. Lee. 2004. Description of “Candidatus Helicobacter heilmannii” based on DNA sequence analysis of 16S rRNA and urease genes. Int. J. Syst. Evol. Microbiol. 54:2203-2211. [DOI] [PubMed] [Google Scholar]
- 167.O'Rourke, J. L., M. F. Dixon, A. Jack, A. Enno, and A. Lee. 2004. Gastric B-cell mucosa-associated lymphoid tissue (MALT) lymphoma in an animal model of ‘Helicobacter heilmannii’ infection. J. Pathol. 203:896-903. [DOI] [PubMed] [Google Scholar]
- 168.O'Toole, P. W., J. W. Austin, and T. J. Trust. 1994. Identification and molecular characterization of a major ring-forming surface protein from the gastric pathogen Helicobacter mustelae. Mol. Microbiol. 11:349-361. [DOI] [PubMed] [Google Scholar]
- 169.O'Toole, P. W., M. Kostrzynska, and T. J. Trust. 1994. Non-motile mutants of Helicobacter pylori and Helicobacter mustelae defective in flagellar hook production. Mol. Microbiol. 14:691-703. [DOI] [PubMed] [Google Scholar]
- 170.Ottemann, K. M., and A. C. Lowenthal. 2002. Helicobacter pylori uses motility for initial colonization and to attain robust infection. Infect. Immun. 70:19847-1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Otto, G., S. H. Hazell, J. G. Fox, C. R. Howlett, J. C. Murphy, J. L. O'Rourke, and A. Lee. 1994. Animal and public health implications of gastric colonization of cats by Helicobacter-like organisms. J. Clin. Microbiol. 32:1043-1049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Papasouliotis, K., T. J. Gruffydd-Jones, G. Werrett, P. J. Brown, and G. R. Pearson. 1997. Occurrence of ‘gastric Helicobacter-like organisms’ in cats. Vet. Rec. 140:369-370. [DOI] [PubMed] [Google Scholar]
- 173.Park, J. H., B. J. Lee, Y. S. Lee, and J. H. Park. 2000. Association of tightly spiraled bacterial infection and gastritis in pigs. J. Vet. Med. Sci. 62:725-729. [DOI] [PubMed] [Google Scholar]
- 174.Parsonnet, J., G. D. Friedman, D. P. Vandersteen, Y. Chang, J. H. Vogelman, N. Orentreich, and R. K. Sibley. 1991. Helicobacter pylori infection and the risk of gastric carcinoma. N. Engl. J. Med. 17:1127-1131. [DOI] [PubMed] [Google Scholar]
- 175.Patterson, M. M., P. W. O'Toole, N. T. Forester, B. Noonan, T. J. Trust, S. Xu, N. S. Taylor, R. P. Marini, M. M. Ihrig, and J. G. Fox. 2003. Failure of surface ring mutant strains of Helicobacter mustelae to persistently infect the ferret stomach. Infect. Immun. 71:2350-2355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Patterson, M. M., M. D. Schrenzel, Y. Feng, and J. G. Fox. 2000. Gastritis and intestinal metaplasia in Syrian hamsters infected with Helicobacter aurati and two other microaerobes. Vet. Pathol. 37:589-596. [DOI] [PubMed] [Google Scholar]
- 177.Patterson, M. M., M. D. Schrenzel, Y. Feng, S. Xu, F. E. Dewhirst, B. J. Paster, S. A. Thibodeau, J. Versalovic, and J. G. Fox. 2000. Helicobacter aurati sp. nov., a urease-positive Helicobacter species cultured from gastrointestinal tissues of Syrian hamsters. J. Clin. Microbiol. 38:3722-3728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Phadnis, S. H., M. H. Parlow, M. Levy, D. Ilver, C. M. Caulkins, J. B. Connors, and B. E. Dunn. 1996. Surface localization of Helicobacter pylori urease and a heat shock protein homolog requires bacterial autolysis. Infect. Immun. 64:905-912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Pinto-Santini, D., and N. R. Salama. 2005. The biology of Helicobacter pylori infection, a major risk factor for gastric adenocarcinoma. Cancer Epidemiol. Biomarkers Prev. 14:1853-1858. [DOI] [PubMed] [Google Scholar]
- 180.Pot, R. G. J., J. Stoof, P. J. M. Nuijten, L. A. M. de Haan, P. Loeffen, E. J. Kuipers, A. H. M. van Vliet, and J. G. Kusters. 2007. UreA2B2: a second urease system in the gastric pathogen Helicobacter felis. FEMS Immunol. Med. Microbiol. 50:273-279. [DOI] [PubMed] [Google Scholar]
- 181.Pounder, R. E., and D. Ng. 1995. The prevalence of Helicobacter pylori infection in different countries. Aliment. Pharmacol. Ther. 9:33-39. [PubMed] [Google Scholar]
- 182.Priestnall, S. L., B. Wiinberg, A. Spohr, B. Neuhaus, M. Kuffer, M. Wiedmann, and K. W. Simpson. 2004. Evaluation of “Helicobacter heilmannii” subtypes in the gastric mucosa of cats and dogs. J. Clin. Microbiol. 42:2144-2151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Queiroz, D. M. M., C. Contigli, R. S. Coimbra, A. M. Nogueira, E. N. Mendes, G. A. Rocha, and S. B. Moura. 1992. Spiral bacterium associated with gastric, ileal and caecal mucosa of mice. Lab. Anim. 26:288-294. [DOI] [PubMed] [Google Scholar]
- 184.Queiroz, D. M. M., G. A. Rocha, E. N. Mendes, A. P. Lage, A. C. T. Carvalho, and A. J. A. Barbosa. 1990. A spiral microorganism in the stomach of pigs. Vet. Microbiol. 24:199-204. [DOI] [PubMed] [Google Scholar]
- 185.Queiroz, D. M. M., G. A. Rocha, E. N. Mendes, S. B. Moura, A. M. Rocha De Oliveira, and D. Miranda. 1996. Association between Helicobacter and gastric ulcer disease of the pars oesophagea in swine. Gastroenterology 111:19-27. [DOI] [PubMed] [Google Scholar]
- 186.Recordati, C., V. Gualdi, S. Tosi, V. Facchini, G. Pengo, M. Luini, K. W. Simpson, and E. Scanziani. 2007. Detection of Helicobacter spp DNA in the oral cavity of dogs. Vet. Microbiol. 119:346-351. [DOI] [PubMed] [Google Scholar]
- 187.Reindel, J. F., A. L. Fitzgerald, M. A. Breider, A. W. Gough, C. Yan, J. V. Mysore, and A. Dubois. 1999. An epizootic of lymphoplasmacytic gastritis attributed to Helicobacter pylori infection in cynomolgus monkeys (Macaca fascicularis). Vet. Pathol. 36:1-13. [DOI] [PubMed] [Google Scholar]
- 188.Rhead, J. L., D. P. Letley, M. Mohammadi, N. Hussein, M. A. Mohagheghi, M. Eshagh Hosseini, and J. C. Atherton. 2007. A new Helicobacter pylori vacuolating cytotoxin determinant, the intermediate region, is associated with gastric cancer. Gastroenterology 133:926-936. [DOI] [PubMed] [Google Scholar]
- 189.Robertson, L. D., J. M. Accioly, K. M. Moore, S. J. Driesen, D. W. Pethick, and D. J. Hampson. 2002. Risk factors for gastric ulcers in Australian pigs at slaughter. Prev. Vet. Med. 53:293-303. [DOI] [PubMed] [Google Scholar]
- 190.Roosendaal, R., J. H. Vos, T. Roumen, R. Van Vugt, G. Cattoli, A. Bart, H. L. Klaasen, E. J. Kuipers, C. M. Vandenbroucke-Grauls, and J. G. Kusters. 2000. Slaughter pigs are commonly infected with closely related but distinct gastric ulcerative lesion-inducing gastrospirilla. J. Clin. Microbiol. 38:2661-2664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Saitou, N., and M. Nei. 1987. The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 4:406-425. [DOI] [PubMed] [Google Scholar]
- 192.Sapierzynski, R., M. Fabisiak, M. Kizerwetter-Swida, and A. Cywinska. 2007. Effect of Helicobacter sp infection on the number of antral gastric endocrine cells in swine. Polish J. Vet. Sci. 10:65-70. [PubMed] [Google Scholar]
- 193.Scanziani, E., K. W. Simpson, S. Monestiroli, S. Soldati, D. Strauss-Ayali, and F. Del Piero. 2001. Histological and immunohistochemical detection of different Helicobacter species in the gastric mucosa of cats. J. Vet. Diagn. Investig. 13:3-12. [DOI] [PubMed] [Google Scholar]
- 194.Schildt, J., T. Grimm, M. Radke, and H. Lobeck. 2000. Akute Oberbauchschmerzen und Hämatemesis. Monatsschr. Kinderheilkd. 148:900-901. [Google Scholar]
- 195.Schmees, C., C. Prinz, T. Treptau, R. Rad, L. Hengst, P. Voland, S. Bauer, L. Brenner, R. M. Schmid, and M. Gerhard. 2007. Inhibition of T-cell proliferation by Helicobacter pylori gamma-glutamyl transpeptidase. Gastroenterology 132:1820-1833. [DOI] [PubMed] [Google Scholar]
- 196.Reference deleted.
- 197.Scott, D. R., E. A. Marcus, D. L. Weeks, and G. Sachs. 2002. Mechanisms of acid resistance due to the urease system of Helicobacter pylori. Gastroenterology 123:187-195. [DOI] [PubMed] [Google Scholar]
- 198.Segal, E. D., J. Cha, J. Lo, S. Falkow, and L. S. Tompkins. 1999. Altered states: involvement of phosphorylated CagA in the induction of host cellular growth changes by Helicobacter pylori. Proc. Natl. Acad. Sci. USA 96:14559-14564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Seo, W. J., C. S. Park, Y. J. Cho, K. W. Cha, S. W. Lee, S. T. Lim, Y. H. Sung, and A. R. Baek. 2003. A case of gastric ulcer induced by Helicobacter heilmannii-like organism. Korean J. Gastroenterol. 42:63-66. [PubMed] [Google Scholar]
- 200.Sgouras, D. N., E. G. Panayotopoulou, B. Martinez-Gonzalez, K. Petraki, S. Michopoulos, and A. Mentis. 2005. Lactobacillus johnsonii La1 attenuates Helicobacter pylori-associated gastritis and reduces levels of proinflammatory chemokines in C57BL/6 mice. Clin. Diagn. Lab. Immunol. 12:1378-1386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Shibayama, K., K. Kamachi, N. Nagata, T. Yagi, T. Nada, Y. H. Doi, N. Shibata, K. Yokoyama, K. Yamane, H. Kato, Y. Iinuma, and Y. Arakawa. 2003. A novel apoptosis-inducing protein from Helicobacter pylori. Mol. Microbiol. 47:443-445. [DOI] [PubMed] [Google Scholar]
- 202.Silva, J. C., J. L. Santos, and A. J. Barbosa. 2002. Gastrinaemia, tissue gastrin concentration and G cell density in the antral mucosa of swine with and without gastric ulcer of the pars oesophagea. J. Comp. Pathol. 126:235-237. [DOI] [PubMed] [Google Scholar]
- 203.Simmons, J. H., L. K. Riley, C. L. Besch-Williford, and C. L. Franklin. 2000. Helicobacter mesocricetorum sp. nov., a novel helicobacter isolated from the feces of Syrian hamsters. J. Clin. Microbiol. 38:1811-1817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Simpson, K. W., P. L. McDonough, D. Strauss-Ayali, Y. F. Chang, P. Harpending, and B. A. Valentine. 1999. Helicobacter felis in dogs: effect on gastric structure and function. Vet. Pathol. 36:237-248. [DOI] [PubMed] [Google Scholar]
- 205.Skouloubris, S., J. M. Thiberge, A. Labigne, and H. De Reuse. 1998. The Helicobacter pylori UreI protein is not involved in urease activity but is essential for bacterial survival in vivo. Infect. Immun. 66:4517-4521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Smith, D. F., L. Munson, and H. N. Erb. 1983. Abomasal ulcer disease in adult dairy cattle. Cornell Vet. 73:213-224. [PubMed] [Google Scholar]
- 207.Smoot, D. T., H. L. Mobley, G. R. Chippendale, J. F. Lewison, and J. H. Resau. 1990. Helicobacter pylori urease activity is toxic to human gastric epithelial cells. Infect. Immun. 58:1992-199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Solnick, J. V., C. Sodi Josenhans, S. Suerbaum, L. S. Tompkins, and A. Labigne. 1995. Construction and characterization of an isogenic urease-negative mutant of Helicobacter mustelae. Infect. Immun. 63:3718-3721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Solnick, J. V., D. R. Canfield, S. Yang, and J. Parsonnet. 1999. The rhesus monkey (Macaca mulatta) model of helicobacter pylori: noninvasive detection and derivation of specific pathogen free monkeys. Lab. Anim. Sci. 49:197-201. [PubMed] [Google Scholar]
- 210.Solnick, J. V., D. R. Canfield, L. M. Hansen, and S. Z. Torabian. 2000. Immunization with recombinant Helicobacter pylori urease in specific pathogen free rhesus monkeys (Macaca mulatta). Infect. Immun. 68:2560-2565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Solnick, J. V., and D. B. Schauer. 2001. Emergence of diverse Helicobacter species in the pathogenesis of gastric and enterohepatic diseases. Clin. Microbiol. Rev. 14:59-97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Solnick, J. V., L. M. Hansen, D. R. Canfield, and J. Parsonnet. 2001. Determination of the infectious dose of Helicobacter pylori during primary and secondary infection in rhesus monkeys (Macaca mulatta). Infect. Immun. 69:6887-6892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Solnick, J. V., K. Chang, D. R. Canfield, and J. Parsonnet. 2003. Natural acquisition of Helicobacter pylori infection in newborn rhesus macaques. J. Clin. Microbiol. 41:5511-5516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Solnick, J. V., J. Fong, L. M. Hansen, K. Chang, D. R. Canfield, and J. Parsonnet. 2006. Acquisition of Helicobacter pylori infection in rhesus macaques is most consistent with oral-oral transmission. J. Clin. Microbiol. 44:3799-3803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Stolte, M., and S. Eidt. 1993. Healing gastric MALT lymphomas by eradicating H. pylori? Lancet 342:568. [DOI] [PubMed] [Google Scholar]
- 216.Stolte, M., G. Kroher, A. Meining, A. Morgner, E. Bayerdörffer, and B. Bethke. 1997. A comparison of Helicobacter pylori and H. heilmannii gastritis. Scand. J. Gastroenterol. 32:28-33. [DOI] [PubMed] [Google Scholar]
- 217.Stolte, M., E. Wellens, B. Bethke, M. Ritter, and H. Eidt. 1994. Helicobacter heilmannii (formerly Gastrospirillum hominis) gastritis: an infection transmitted by animals? Scand. J. Gastroenterol. 29:1061-1064. [DOI] [PubMed] [Google Scholar]
- 218.Strauss-Ayali, D., E. Scanziani, D. Deng, and K. W. Simpson. 2001. Helicobacter species infection in cats: evaluation of the humoral immune respons and prevalence of gastric Helicobacter species. Vet. Microbiol. 79:253-265. [DOI] [PubMed] [Google Scholar]
- 219.Suerbaum, S., C. Kraft, F. E. Dewhirst, and J. G. Fox. 2002. Helicobacter nemestrinae ATCC 49396T is a strain of Helicobacter pylori (Marshall et al. 1985) Goodwin et al. 1989, and Helicobacter nemestrinae Bronsdon et al. 1991 is therefore a junior heterotypic synonym of Helicobacter pylori. Int. J. Syst. Evol. Microbiol. 52:437-439. [DOI] [PubMed] [Google Scholar]
- 220.Svec, A., P. Kordas, Z. Pavlis, and J. Novotny. 2000. High prevalence of Helicobacter heilmannii-associated gastritis in a small, predominantly rural area: further evidence in support of a zoonosis? Scand. J. Gastroenterol. 35:925-928. [DOI] [PubMed] [Google Scholar]
- 221.Sykora, J., V. Hejda, J. Varvarovska, F. Stozicky, F. Gottrand, and K. Siala. 2003. Helicobacter heilmannii related gastric ulcer in childhood. J. Pediatr. Gastroenterol. Nutr. 36:410-413. [DOI] [PubMed] [Google Scholar]
- 222.Szeredi, L., G. Palkovics, N. Solymosi, L. Tekes, and J. Mehesfalvi. 2005. Study on the role of gastric Helicobacter infection in gross pathological and histological lesions of the stomach in finishing pigs. Acta Vet. Hung. 53:371-383. [DOI] [PubMed] [Google Scholar]
- 223.Taylor, N. S., A. T. Hasubski, J. G. Fox, and A. Lee. 1992. Haemagglutination profiles of Helicobacter species that cause gastritis in man and animals. J. Med. Microbiol. 37:299-303. [DOI] [PubMed] [Google Scholar]
- 224.Terio, K. A., L. Munson, L. Marker, B. M. Aldridge, and J. V. Solnick. 2005. Comparison of Helicobacter spp. in cheetahs (Acinonyx jubatus) with and without gastritis. J. Clin. Microbiol. 43:229-234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Thompson, J. D., D. G. Higgins, and T. J. Gibson. 1994. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 22:4673-4680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Thomson, M. A., P. Storey, R. Greer, and G. J. Cleghorn. 1994. Canine-human transmission of Gastrospirillum hominis. Lancet 343:1605-1607. [DOI] [PubMed] [Google Scholar]
- 227.Trebesius, K., K. Adler, M. Vieth, M. Stolte, and R. Haas. 2001. Specific detection and prevalence of Helicobacter heilmannii-like organisms in the human gastric mucosa by fluorescent in situ hybridization and partial 16S ribosomal DNA sequencing. J. Clin. Microbiol. 39:1510-1516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Vandamme, P., C. S. Harrington, K. Jalava, S. L. W. On. 2000. Misidentifying helicobacters: the Helicobacter cinaedi example. J. Clin. Microbiol. 38:2261-2266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Van de Bovenkamp, J. H. B., A. M. Korteland-van Male, H. A. Büller, A. W. C. Einerhand, and J. Dekker. 2005. Infection with Helicobacter pylori affects all major secretory cell populations in the human antrum. Dig. Dis. Sci. 50:1078-1086. [DOI] [PubMed] [Google Scholar]
- 230.Van den Bulck, K., A. Decostere, M. Baele, A. Driesen, J. C. Debongnie, A. Burette, M. Stolte, R. Ducatelle, and F. Haesebrouck. 2005. Identification of non-Helicobacter pylori spiral organisms in gastric samples from humans, dogs, and cats. J. Clin. Microbiol. 42:2256-2260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Van den Bulck, K., M. Baele, K. Hermans, R. Ducatelle, F. Haesebrouck, and A. Decostere. 2005. First report on the occurrence of ‘Helicobacter heilmannii’ in the stomach of rabbits. Vet. Res. Commun. 29:271-279. [DOI] [PubMed] [Google Scholar]
- 232.Van den Bulck, K., A. Decostere, I. Gruntar, M. Baele, B. Krt, R. Ducatelle, and F. Haesebrouck. 2005. In vitro antimicrobial susceptibility testing of Helicobacter felis, H. bizzozeronii, and H. salomonis. Antimicrob. Agents Chemother. 49:2297-3000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Van den Bulck, K., A. Decostere, M. Baele, P. Vandamme, J. Mast, R. Ducatelle, and F. Haesebrouck. 2006. Helicobacter cynogastricus sp. nov., a Helicobacter species isolated from the canine gastric mucosa. Int. J. Syst. Evol. Microbiol. 56:1559-1564. [DOI] [PubMed] [Google Scholar]
- 234.Van den Bulck, K., A. Decostere, M. Baele, M. Marechal, R. Ducatelle, and F. Haesebrouck. 2006. Low frequency of Helicobacter species in the stomachs of experimental rabbits. Laboratory Animals. 40:282-287. [DOI] [PubMed] [Google Scholar]
- 235.van Loon, S., A. Bart, E. J. den Hertog, P. G. Nikkels, R. H. Houwen, J. E. De Schryver, and J. H. Oudshoorn. 2003. Helicobacter heilmannii gastritis caused by cat to child transmission. J. Pediatr. Gastroenterol. Nutr. 36:407-409. [DOI] [PubMed] [Google Scholar]
- 236.Weber, A. F., O. Hasa, and J. H. Sautter. 1958. Some observations concerning the presence of spirilla in the fundic glands of dogs and cats. Am. J. Vet. Res. 19:677-680. [PubMed] [Google Scholar]
- 237.Weeks, D. L., S. Eskandari, D. R. Scott, and G. Sachs. 2000. A H+-gated urea channel: the link between Helicobacter pylori urease and gastric colonization. Science 287:482-285. [DOI] [PubMed] [Google Scholar]
- 238.Wegmann, W., M. Aschwanden, N. Schaub, W. Aenishänslin, and K. Gyr. 1991. Gastritis associated with Gastrospirillum hominis—a zoonosis? Schweiz Med. Wochenschr. 121:245-254. [PubMed] [Google Scholar]
- 239.Welchman, D. D., and G. N. Baust. 1987. A survey of abomasal ulceration in veal calves. Vet. Rec. 121:586-590. [PubMed] [Google Scholar]
- 240.Whary, M. T., and J. G. Fox. 2004. Natural and experimental Helicobacter infections. Comp. Med. 54:128-158. [PubMed] [Google Scholar]
- 241.Wiinberg, B., A. Spohr, H. H. Dietz, T. Egelund, A. Greiter-Wilke, S. P. McDonough, J. Olsen, S. Priestnall, Y. F. Chang, and K. W. Simpson. 2005. Quantitative analysis of inflammatory and immune responses in dogs with gastritis and their relationship to Helicobacter spp. infection. J. Vet. Intern. Med. 19:4-14. [DOI] [PubMed] [Google Scholar]
- 242.Williams, C. L., T. Preston, M. Hossack, C. Slater, and K. E. McColl. 1996. Helicobacter pylori utilizes urea for amino acid synthesis. FEMS Immunol. Med. Microbiol. 13:87-94. [DOI] [PubMed] [Google Scholar]
- 243.Yamasaki, K., H. Suematsu, and T. Takahashi. 1998. Comparison of gastric lesions in dogs and cats with and without gastric spiral organisms. J. Am. Vet. Med. Assoc. 212:529-533. [PubMed] [Google Scholar]
- 244.Yang, H., X. Li, Z. Xu, and D. Zhou. 1995. “Helicobacter heilmannii” infection in a patient with gastric cancer. Dig. Dis. Sci. 40:1013-1014. [DOI] [PubMed] [Google Scholar]
- 245.Yang, H., J. A. Goliger, M. Song, and D. Zhou. 1998. High prevalence of Helicobacter heilmannii infection in China. Dig. Dis. Sci. 43:1493. [DOI] [PubMed] [Google Scholar]
- 246.Yoshimura, M., H. Isomoto, S. Shikuwa, M. Osabe, K. Matsunaga, K. Omagari, Y. Mizuta, K. Murase, I. Murata, and S. Kohno. 2002. A case of acute gastric mucosal lesions associated with Helicobacter heilmannii infection. Helicobacter 7:322-326. [DOI] [PubMed] [Google Scholar]
- 247.Yu,J., R. M. Russell, R. N. Salomon, J. C. Murphy, L. S. Palley, and J. G. Fox. 1995. Effect of Helicobacter mustelae infection on ferret gastric epithelial cell proliferation. Carcinogenesis 16:1927-1931. [DOI] [PubMed] [Google Scholar]